videx (DIDANOSINE) powder, for solution
[Bristol-Myers Squibb]
VIDEX® (didanosine) Pediatric Powder for Oral Solution
(Patient Information Leaflet Included)
WARNING
FATAL AND NONFATAL PANCREATITIS HAVE OCCURRED DURING THERAPY WITH VIDEX USED ALONE OR IN COMBINATION REGIMENS IN BOTH TREATMENT-NAIVE AND TREATMENT-EXPERIENCED PATIENTS, REGARDLESS OF DEGREE OF IMMUNOSUPPRESSION. VIDEX SHOULD BE SUSPENDED IN PATIENTS WITH SUSPECTED PANCREATITIS AND DISCONTINUED IN PATIENTS WITH CONFIRMED PANCREATITIS (SEE WARNINGS).
LACTIC ACIDOSIS AND SEVERE HEPATOMEGALY WITH STEATOSIS, INCLUDING FATAL CASES, HAVE BEEN REPORTED WITH THE USE OF NUCLEOSIDE ANALOGUES ALONE OR IN COMBINATION, INCLUDING DIDANOSINE AND OTHER ANTIRETROVIRALS. FATAL LACTIC ACIDOSIS HAS BEEN REPORTED IN PREGNANT WOMEN WHO RECEIVED THE COMBINATION OF DIDANOSINE AND STAVUDINE WITH OTHER ANTIRETROVIRAL AGENTS. THE COMBINATION OF DIDANOSINE AND STAVUDINE SHOULD BE USED WITH CAUTION DURING PREGNANCY AND IS RECOMMENDED ONLY IF THE POTENTIAL BENEFIT CLEARLY OUTWEIGHS THE POTENTIAL RISK (SEE WARNINGS AND PRECAUTIONS: PREGNANCY, REPRODUCTION, AND FERTILITY).
DESCRIPTION
VIDEX® (didanosine) is a brand name for didanosine (ddI), a synthetic purine nucleoside analogue active against the Human Immunodeficiency Virus (HIV).
VIDEX Pediatric Powder for Oral Solution is supplied for oral administration in 4- or 8-ounce glass bottles containing 2 or 4 grams of didanosine, respectively.
Didanosine is also available as an enteric-coated formulation (VIDEX® EC Delayed-Release Capsules). Please consult the prescribing information for VIDEX EC (didanosine).
The chemical name for didanosine is 2′,3′-dideoxyinosine. The structural formula is:
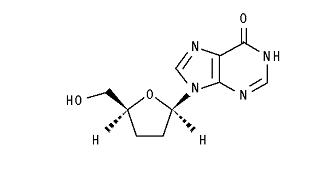
Didanosine is a white crystalline powder with the molecular formula C10H12N4O3 and a molecular weight of 236.2. The aqueous solubility of didanosine at 25° C and pH of approximately 6 is 27.3 mg/mL. Didanosine is unstable in acidic solutions. For example, at pH <3 and 37° C, 10% of didanosine decomposes to hypoxanthine in less than 2 minutes.
MICROBIOLOGY
Mechanism of Action
Didanosine is a synthetic nucleoside analogue of the naturally occurring nucleoside deoxyadenosine in which the 3′-hydroxyl group is replaced by hydrogen. Intracellularly, didanosine is converted by cellular enzymes to the active metabolite, dideoxyadenosine 5′-triphosphate. Dideoxyadenosine 5′-triphosphate inhibits the activity of HIV-1 reverse transcriptase both by competing with the natural substrate, deoxyadenosine 5′-triphosphate, and by its incorporation into viral DNA causing termination of viral DNA chain elongation.
HIV Susceptibility in Cell Culture
The anti-HIV-1 activity of didanosine was evaluated in a variety of HIV-1 infected lymphoblastic cell lines and monocyte/macrophage cell cultures. The concentration of drug necessary to inhibit viral replication by 50% (EC50) ranged from 2.5 to 10 µM (1 µM = 0.24 µg/mL) in lymphoblastic cell lines and 0.01 to 0.1 µM in monocyte/macrophage cell cultures.
Resistance
HIV-1 isolates with reduced sensitivity to didanosine have been selected in cell culture and were also obtained from patients treated with didanosine. Genetic analysis of isolates from didanosine-treated patients showed mutations in the reverse transcriptase gene that resulted in the amino acid substitutions K65R, L74V, and M184V. The L74V mutation was most frequently observed in clinical isolates. Phenotypic analysis of HIV-1 isolates from 60 patients (some with prior zidovudine treatment) receiving 6 to 24 months of didanosine monotherapy showed that isolates from 10 of 60 patients exhibited an average of a 10-fold decrease in susceptibility to didanosine in cell culture compared to baseline isolates. Clinical isolates that exhibited a decrease in didanosine susceptibility harbored one or more didanosine-associated mutations. The clinical relevance of genotypic and phenotypic changes associated with didanosine therapy has not been established.
Cross-resistance
HIV-1 isolates from 2 of 39 patients receiving combination therapy for up to 2 years with zidovudine and didanosine exhibited decreased susceptibility to zidovudine, didanosine, zalcitabine, stavudine, and lamivudine in cell culture. These isolates harbored five mutations (A62V, V75I, F77L, F116Y, and Q151M) in the reverse transcriptase gene. In data from clinical studies, the presence of thymidine analogue mutations (M41L, D67N, L210W, T215Y, K219Q) has been shown to decrease the response to didanosine.
CLINICAL PHARMACOLOGY
Animal Toxicology
Evidence of a dose-limiting skeletal muscle toxicity has been observed in mice and rats (but not in dogs) following long-term (greater than 90 days) dosing with didanosine at doses that were approximately 1.2 to 12 times the estimated human exposure. The relationship of this finding to the potential of VIDEX (didanosine) to cause myopathy in humans is unclear. However, human myopathy has been associated with administration of VIDEX and other nucleosideanalogues.
Pharmacokinetics
The pharmacokinetic parameters of didanosine are summarized in Table 1. Didanosine is rapidly absorbed, with peak plasma concentrations generally observed from 0.25 to 1.50 hours following oral dosing. Increases in plasma didanosine concentrations were dose proportional over the range of 50 to 400 mg. Steady-state pharmacokinetic parameters did not differ significantly from values obtained after a single dose. Binding of didanosine to plasma proteins in vitro was low (<5%). Based on data from in vitro and animal studies, it is presumed that the metabolism of didanosine in man occurs by the same pathways responsible for the elimination of endogenous purines.
| Pediatric Patientsb | ||||||
|---|---|---|---|---|---|---|
| Parameter | Adult Patientsa | n | 8 months to 19 years | n | 2 weeks to 4 months | n |
| CSF = cerebrospinal fluid, ND = not determined. | ||||||
| a Parameter units for adults were converted to the same units in pediatric patients to facilitate comparisons among populations: mean adult body weight = 70 kg and mean adult body surface area = 1.73 m2. | ||||||
| b In 1-day old infants (n=10), the mean ± SD apparent oral clearance was 1523 ± 1176 mL/min/m2 and half-life was 2.0 ± 0.7 h. | ||||||
| c Following IV administration. | ||||||
| d Following IV administration in adults and IV or oral administration in pediatric patients. | ||||||
| e Mean ± SE. | ||||||
| f Following oral administration. | ||||||
| g Apparent oral clearance estimate was determined as the ratio of the mean systemic clearance and the mean oral bioavailability estimate. | ||||||
| Oral bioavailability (%) | 42 ± 12 | 6 | 25 ± 20 | 46 | ND | |
| Apparent volume of distributionc (L/m2) | 43.70 ± 8.90 | 6 | 28 ± 15 | 49 | ND | |
| CSF-plasma ratiod | 21 ± 0.03%e | 5 | 46% (range 12-85%) | 7 | ND | |
| Systemic clearancec
(mL/min/m2) | 526 ± 64.7 | 6 | 516 ± 184 | 49 | ND | |
| Renal clearancef (mL/min/m2) | 223 ± 85.0 | 6 | 240 ± 90 | 15 | ND | |
| Apparent oral clearanceg (mL/min/m2) | 1252 ± 154 | 6 | 2064 ± 736 | 48 | 1353 ± 759 | 41 |
| Elimination half-lifef (h) | 1.5 ± 0.4 | 6 | 0.8 ± 0.3 | 60 | 1.2 ± 0.3 | 21 |
| Urinary recovery of didanosinef (%) | 18 ± 8 | 6 | 18 ± 10 | 15 | ND | |
Effect of Food on Absorption of Didanosine: Didanosine peak plasma concentrations (CMAX) and area under the plasma concentration time curve (AUC) were decreased by approximately 55% when VIDEX tablets were administered up to 2 hours after a meal. Administration of VIDEX tablets up to 30 minutes before a meal did not result in any significant changes in bioavailability. VIDEX should be taken on an empty stomach, at least 30 minutes before or 2 hours after eating. (See DOSAGE AND ADMINISTRATION.)
Special Populations
Renal Insufficiency: It is recommended that the VIDEX (didanosine) dose be modified in patients with reduced creatinine clearance and in patients receiving maintenance hemodialysis (see DOSAGE AND ADMINISTRATION). Data from two studies in adults indicated that the apparent oral clearance of didanosine decreased and the terminal elimination half-life increased as creatinine clearance decreased (see Table 2). Following oral administration, didanosine was not detectable in peritoneal dialysate fluid (n=6); recovery in hemodialysate (n=5) ranged from 0.6% to 7.4% of the dose over a 3-4 hour dialysis period. The absolute bioavailability of didanosine was not affected in patients requiring dialysis.
| Creatinine Clearance (mL/min) | |||||
|---|---|---|---|---|---|
| Parameter | ≥90 (n=12) | 60-90 (n=6) | 30-59 (n=6) | 10-29 (n=3) | Dialysis Patients (n=11) |
| ND = not determined due to anuria. | |||||
| CLcr = creatinine clearance. | |||||
| CL/F = apparent oral clearance. | |||||
| CLR = renal clearance. | |||||
| CLcr (mL/min) | 112 ± 22 | 68 ± 8 | 46 ± 8 | 13 ± 5 | ND |
| CL/F (mL/min) | 2164 ± 638 | 1566 ± 833 | 1023 ± 378 | 628 ± 104 | 543 ± 174 |
| CLR (mL/min) | 458 ± 164 | 247 ± 153 | 100 ± 44 | 20 ± 8 | <10 |
| T½ (h) | 1.42 ± 0.33 | 1.59 ± 0.13 | 1.75 ± 0.43 | 2.0 ± 0.3 | 4.1 ± 1.2 |
Pediatric Patients: The pharmacokinetics of didanosine have been evaluated in HIV-exposed and -infected pediatric patients from birth to 19 years of age (see Table 1). Overall, the pharmacokinetics of didanosine in pediatric patients are similar to those of didanosine in adults. Didanosine plasma concentrations appear to increase in proportion to oral doses ranging from 25 to 120 mg/m2 in pediatric patients less than 5 months old and from 80 to 180 mg/m2 in children above 8 months old. For information on controlled clinical studies in pediatric patients, see INDICATIONS AND USAGE: Clinical Studies and PRECAUTIONS: Pediatric Use.
Geriatric Patients: Didanosine pharmacokinetics have not been studied in patients over 65 years of age.
Gender: The effects of gender on didanosine pharmacokinetics have not been studied.
Drug Interactions: Ribavirin has been shown in vitro to increase intracellular triphosphate levels of didanosine, which could cause or worsen clinical toxicities (see PRECAUTIONS: Drug Interactions).
Tables 3 and 4 summarize the effects on AUC and CMAX, with a 90% or 95% confidence interval (CI) when available, following coadministration of VIDEX (didanosine) with a variety of drugs. For most of the listed drugs, no clinically significant pharmacokinetic interactions were observed. Clinical recommendations based on drug interaction studies for drugs in bold font are included in PRECAUTIONS: Drug Interactions and DOSAGE AND ADMINISTRATION (for tenofovir).
| ↑ indicates increase. | ||||
| ↓ indicates decrease. | ||||
| ↔ indicates no change, or mean increase or decrease of <10%. | ||||
| a HIV-infected patients. | ||||
| b 90% CI. | ||||
| c Parallel-group design; entries are subjects receiving combination and control regimens, respectively. | ||||
| d tenofovir disoproxil fumarate. | ||||
| e For results of drug interaction studies between the enteric-coated formulation of didanosine (VIDEX EC) and tenofovir, see the complete prescribing information for VIDEX EC. | ||||
| f patients <60 kg with creatinine clearance ≥60 mL/min. | ||||
| NA Not available. | ||||
| Drugs With Clinical Recommendations Regarding Coadministration(see PRECAUTIONS: Drug Interactions) | ||||
| Drug | Didanosine Dosage | n | AUC of Didanosine (95% CI) | CMAX of
Didanosine (95% CI) |
| allopurinol
renally impaired, 300 mg/day | 200 mg single dose | 2 | ↑ 312% | ↑ 232% |
| healthy volunteer, 300 mg/day for 7 days | 400 mg single dose | 14 | ↑ 113% | ↑ 69% |
| ciprofloxacin, 750 mg q12h for 3 days, 2 h before didanosine | 200 mg q12h for 3 days | 8a | ↓ 16% | ↓ 28% |
| ganciclovir,1000 mg q8h, 2 h after didanosine | 200 mg q12h | 12 | ↑ 111% | NA |
| indinavir, 800 mg single dose simultaneous 1 h before didanosine | 200 mg single dose 200 mg single dose | 16 16 | ↔ ↓ 17% (-27, - 7%)b | ↔ ↓ 13% (-28, 5%)b |
| ketoconazole, 200 mg/day for 4 days, 2 h before didanosine | 375 mg q12h for 4 days | 12a | ↔ | ↓ 12% |
| methadone, chronic maintenance dose | 200 mg single dose | 16, 10c | ↓ 57% | ↓ 66% |
| tenofovir,d,e 300
mg once daily, 1 h after didanosine | 250f or 400 mg once daily for 7 days | 14 | ↑ 44% (31, 59%)b | ↑ 28% (11, 48%)b |
| No Clinically Significant Interaction Observed | ||||
| Drug | Didanosine Dosage | n | AUC of Didanosine (95% CI) | CMAX of
Didanosine (95% CI) |
| loperamide, 4 mg q6h for 1 day | 300 mg single dose | 12a | ↔ | ↓ 23% |
| metoclopramide, 10 mg single dose | 300 mg single dose | 12a | ↔ | ↑ 13% |
| ranitidine, 150 mg single dose, 2 h before didanosine | 375 mg single dose | 12a | ↑ 14% | ↑ 13% |
| rifabutin, 300 or 600 mg/day for 12 days | 167 or 250 mg q12h for 12 days | 11 | ↑ 13% (-1, 27%) | ↑ 17% (-4, 38%) |
| ritonavir, 600 mg q12h for 4 days | 200 mg q12h for 4 days | 12 | ↓ 13% (0, 23%) | ↓ 16% (5, 26%) |
| stavudine, 40 mg q12h for 4 days | 100 mg q12h for 4 days | 10 | ↔ | ↔ |
| sulfamethoxazole, 1000 mg single dose | 200 mg single dose | 8a | ↔ | ↔ |
| trimethoprim, 200 mg single dose | 200 mg single dose | 8a | ↔ | ↑ 17% (-23, 77%) |
| zidovudine, 200 mg q8h for 3 days | 200 mg q12h for 3 days | 6a | ↔ | ↔ |
| ↑ indicates increase. | ||||
| ↓ indicates decrease. | ||||
| ↔ indicates no change, or mean increase or decrease of <10%. | ||||
| a HIV-infected patients. | ||||
| b tenofovir disoproxil fumarate. | ||||
| c patients <60 kg with creatinine clearance ≥60 mL/min. | ||||
| NA Not available. | ||||
| Drugs With Clinical Recommendations Regarding Coadministration (see PRECAUTIONS: Drug Interactions) | ||||
| Drug | Didanosine Dosage | n | AUC of Coadministered Drug (95% CI) | CMAX of Coadministered Drug (95% CI) |
| ciprofloxacin
750 mg q12h for 3 days, 2 h before didanosine | 200 mg q12h for 3 days | 8a | ↓ 26% | ↓ 16% |
| 750 mg single dose | buffered placebo tablet | 12 | ↓ 98% | ↓ 93% |
| delavirdine, 400 mg single dose simultaneous 1 h before didanosine | 125 or 200 mg q12h 125 or 200 mg q12h | 12a
12a | ↓ 32% ↑ 20% | ↓ 53% ↑ 18% |
| ganciclovir, 1000 mg q8h, 2 h after didanosine | 200 mg q12h | 12a | ↓ 21% | NA |
| indinavir, 800 mg single dose simultaneous 1 h before didanosine | 200 mg single dose 200 mg single dose | 16 16 | ↓ 84% ↓ 11% | ↓ 82% ↓ 4% |
| ketoconazole, 200 mg/day for 4 days, 2 h before didanosine | 375 mg q12h for 4 days | 12a | ↓ 14% | ↓ 20% |
| nelfinavir, 750 mg single dose, 1 h after didanosine | 200 mg single dose | 10a | ↑ 12% | ↔ |
| No Clinically Significant Interaction Observed | ||||
| Drug | Didanosine Dosage | n | AUC of Coadministered Drug (95% CI) | CMAX of Coadministered Drug (95% CI) |
| dapsone, 100 mg single dose | 200 mg q12h for 14 days | 6a | ↔ | ↔ |
| ranitidine, 150 mg single dose, 2 h before didanosine | 375 mg single dose | 12a | ↓ 16% | ↔ |
| ritonavir, 600 mg q12h for 4 days | 200 mg q12h for 4 days | 12 | ↔ | ↔ |
| stavudine, 40 mg q12h for 4 days | 100 mg q12h for 4 days | 10a | ↔ | ↑ 17% |
| sulfamethoxazole, 1000 mg single dose | 200 mg single dose | 8a | ↓ 11% (-17, -4%) | ↓ 12% (-28, 8%) |
| tenofovir,b 300 mg once daily 1 h after didanosine | 250c or 400 mg once daily for 7 days | 14 | ↔ | ↔ |
| trimethoprim, 200 mg single dose | 200 mg single dose | 8a | ↑ 10% (-9, 34%) | ↓ 22% (-59, 49%) |
| zidovudine, 200 mg q8h for 3 days | 200 mg q12h for 3 days | 6a | ↓ 10% (-27, 11%) | ↓ 16.5% (-53, 47%) |
INDICATIONS AND USAGE
VIDEX (didanosine) in combination with other antiretroviral agents is indicated for the treatment of HIV-1 infection (see Clinical Studies).
Clinical Studies
Combination Therapy
START 2 was a multicenter, randomized, open-label study comparing VIDEX (200 mg twice daily)/stavudine/indinavir to zidovudine/lamivudine/indinavir in 205 treatment-naive patients. Both regimens resulted in a similar magnitude of suppression of HIV RNA levels and increases in CD4 cell counts through 48 weeks.
Study A1454-148 was a randomized, open-label, multicenter study comparing treatment with VIDEX (400 mg once daily) plus stavudine (40 mg twice daily) and nelfinavir (750 mg three times daily) versus zidovudine (300 mg twice daily) plus lamivudine (150 mg twice daily) and nelfinavir (750 mg three times daily) in 756 treatment-naive patients, with a median CD4 cell count of 340 cells/mm3 (range 80 to 1568 cells/mm3) and a median plasma HIV-1 RNA of 4.69 log10 copies/mL (range 2.6 to 5.9 log10 copies/mL) at baseline. Median CD4 cell count increases at 48 weeks were 188 cells/mm3 in both treatment groups. Treatment response and outcomes through 48 weeks are shown in Figure 1 and Table 5.
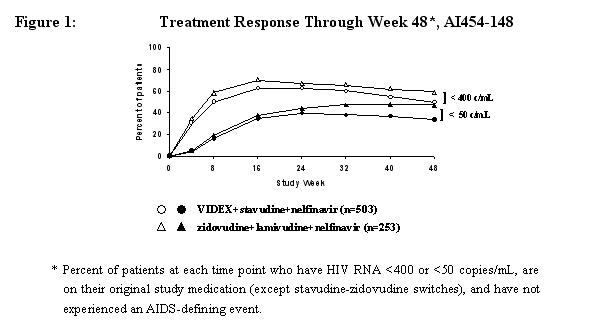
| Percent of Patients with HIV RNA <400 copies/mL (<50 copies/mL) | ||
|---|---|---|
| Week 48 Status | VIDEX/stavudine/nelfinavir n=503 | lamivudine/zidovudine/nelfinavir n=253 |
| * p <0.05 for the differences between treatment groups, by Cochran-Mantel-Haenszel test. | ||
| a Patients achieved virologic response [two consecutive viral loads <400 (<50) copies/mL] and maintained it to Week 48. | ||
| b Includes viral rebound and failing to achieve confirmed <400 (<50) copies/mL by Week 48. | ||
| c Includes lost to follow-up, noncompliance, withdrawal, and pregnancy. | ||
| Respondera | 50* (34*) | 59 (47) |
| Virologic failureb | 36 (57) | 32 (48) |
| Death or disease progression | <1 (<1) | 1 (<1) |
| Discontinued due to adverse events | 4 (2) | 2 (<1) |
| Discontinued due to other reasonsc | 6 (3) | 4 (2) |
| Never initiated treatment | 4 (4) | 2 (2) |
Monotherapy
The efficacy of VIDEX was demonstrated in two randomized, double-blind studies comparing VIDEX, given on a twice-daily schedule, to zidovudine, given three times daily, in 617 (ACTG 116A, conducted 1989-1992) and 913 (ACTG 116B/117, conducted 1989-1991) patients with symptomatic HIV infection or AIDS who were treated for more than one year. In treatment-naive patients (ACTG 116A), the rate of HIV disease progression or death was similar between the treatment groups; mortality rates were 26% for patients receiving VIDEX and 21% for patients receiving zidovudine. Of the patients who had received previous zidovudine treatment (ACTG 116B/117), those treated with VIDEX had a lower rate of HIV disease progression or death (32%) compared to those treated with zidovudine (41%); however, survival rates were similar between the treatment groups.
Efficacy in pediatric patients was demonstrated in a randomized, double-blind, controlled study (ACTG 152, conducted 1991-1995) involving 831 patients 3 months to 18 years of age treated for more than 1.5 years with zidovudine (180 mg/m2 q6h), VIDEX (120 mg/m2 q12h), or zidovudine (120 mg/m2 q6h) plus VIDEX (90 mg/m2 q12h). Patients treated with VIDEX or VIDEX plus zidovudine had lower rates of HIV disease progression or death compared with those treated with zidovudine alone.
Studies have demonstrated that the clinical benefit of monotherapy with antiretrovirals, including VIDEX, was time limited.
CONTRAINDICATION
VIDEX (didanosine) is contraindicated in patients with previously demonstrated clinically significant hypersensitivity to any of the components of the formulations.
WARNINGS
1. Pancreatitis
FATAL AND NONFATAL PANCREATITIS HAVE OCCURRED DURING THERAPY WITH VIDEX USED ALONE OR IN COMBINATION REGIMENS IN BOTH TREATMENT-NAIVE AND TREATMENT-EXPERIENCED PATIENTS, REGARDLESS OF DEGREE OF IMMUNOSUPPRESSION. VIDEX SHOULD BE SUSPENDED IN PATIENTS WITH SIGNS OR SYMPTOMS OF PANCREATITIS AND DISCONTINUED IN PATIENTS WITH CONFIRMED PANCREATITIS. PATIENTS TREATED WITH VIDEX IN COMBINATION WITH STAVUDINE, WITH OR WITHOUT HYDROXYUREA, MAY BE AT INCREASED RISK FOR PANCREATITIS.
When treatment with life-sustaining drugs known to cause pancreatic toxicity is required, suspension of VIDEX (didanosine) therapy is recommended. In patients with risk factors for pancreatitis, VIDEX should be used with extreme caution and only if clearly indicated. Patients with advanced HIV infection, especially the elderly, are at increased risk of pancreatitis and should be followed closely. Patients with renal impairment may be at greater risk for pancreatitis if treated without dose adjustment.
The frequency of pancreatitis is dose related. In phase 3 studies, incidence ranged from 1% to 10% with doses higher than are currently recommended and 1% to 7% with recommended dose.
In pediatric phase 1 studies, pancreatitis occurred in 3% (2/60) of patients treated at entry doses below 300 mg/m2/day and in 13% (5/38) of patients treated at higher doses. In study ACTG 152, pancreatitis occurred in none of the 281 pediatric patients who received didanosine 120 mg/m2 q12h and in <1% of the 274 pediatric patients who received didanosine 90 mg/m2 q12h in combination with zidovudine. VIDEX (didanosine) use should be suspended in pediatric patients with signs or symptoms of pancreatitis and discontinued in pediatric patients with confirmed pancreatitis.
2. Lactic Acidosis/Severe Hepatomegaly with Steatosis
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogues alone or in combination, including didanosine and other antiretrovirals. A majority of these cases have been in women. Obesity and prolonged nucleoside exposure may be risk factors. Fatal lactic acidosis has been reported in pregnant women who received the combination of didanosine and stavudine with other antiretroviral agents. The combination of didanosine and stavudine should be used with caution during pregnancy and is recommended only if the potential benefit clearly outweighs the potential risk (see PRECAUTIONS: Pregnancy, Reproduction, and Fertility). Particular caution should be exercised when administering VIDEX to any patient with known risk factors for liver disease; however, cases have also been reported in patients with no known risk factors. Treatment with VIDEX should be suspended in any patient who develops clinical or laboratory findings suggestive of symptomatic hyperlactatemia, lactic acidosis, or pronounced hepatotoxicity (which may include hepatomegaly and steatosis even in the absence of marked transaminase elevations).
3. Retinal Changes and Optic Neuritis
Retinal changes and optic neuritis have been reported in adult and pediatric patients. Periodic retinal examinations should be considered for patients receiving VIDEX. (See ADVERSE REACTIONS.)
4. Hepatic Impairment and Toxicity
It is unknown if hepatic impairment significantly affects didanosine pharmacokinetics. Therefore, these patients should be monitored closely for evidence of didanosine toxicity. The safety and efficacy of VIDEX have notbeen established in HIV-infected patients with significant underlying liver disease. During combination antiretroviral therapy, patients with preexisting liver dysfunction, including chronic active hepatitis, have an increased frequency of liver function abnormalities, including severe and potentially fatal hepatic adverse events, and should be monitored according to standard practice. If there is evidence of worsening liver disease in such patients, interruption or discontinuation of treatment must be considered.
Hepatotoxicity and hepatic failure resulting in death were reported during postmarketing surveillance in HIV-infected patients treated with hydroxyurea and other antiretroviral agents. Fatal hepatic events were reported most often in patients treated with the combination of hydroxyurea, didanosine, and stavudine. This combination should be avoided.
PRECAUTIONS
Frequency of Dosing
The preferred dosing frequency of VIDEX is twice daily because there is more evidence to support the effectiveness of this dosing frequency. Once-daily dosing should be considered only for adult patients whose management requires once-daily dosing of VIDEX (see INDICATIONS AND USAGE: Clinical Studies).
VIDEX should be taken on an empty stomach, at least 30 minutes before or 2 hours after eating.
Peripheral Neuropathy
Peripheral neuropathy, manifested by numbness, tingling, or pain in the hands or feet, has been reported in patients receiving VIDEX therapy. Peripheral neuropathy has occurred more frequently in patients with advanced HIV disease, in patients with a history of neuropathy, or in patients being treated with neurotoxic drug therapy, including stavudine. Peripheral neuropathy, which was severe in some cases, has been reported in HIV-infected patients receiving hydroxyurea in combination with antiretroviral agents, including didanosine, with or without stavudine (see ADVERSE REACTIONS).
Fat Redistribution
Redistribution/accumulation of body fat including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and “cushingoid appearance” have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including VIDEX. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jiroveci pneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
General
Patients with Renal Impairment: Patients with renal impairment (creatinine clearance <60 mL/min) may be at greater risk of toxicity from VIDEX due to decreased drug clearance (see CLINICAL PHARMACOLOGY). A dose reduction is recommended in these patients (see DOSAGE AND ADMINISTRATION).
Hyperuricemia: VIDEX has been associated with asymptomatic hyperuricemia; treatment suspension may be necessary if clinical measures aimed at reducing uric acid levels fail.
Information for Patients (see Patient Information Leaflet)
Patients should be informed that a serious toxicity of VIDEX used alone and in combination regimens is pancreatitis, which may be fatal.
Patients should be informed that the preferred dosing frequency of VIDEX is twice daily because there is more evidence to support the effectiveness of this dosing frequency. Once-daily dosing should be considered only for adult patients whose management requires once-daily dosing of VIDEX.
Patients should also be aware that peripheral neuropathy, manifested by numbness, tingling, or pain in hands or feet, may develop during therapy with VIDEX. Patients should be counseled that peripheral neuropathy occurs with greatest frequency in patients with advanced HIV disease or a history of peripheral neuropathy, and that dose modification and/or discontinuation of VIDEX may be required if toxicity develops.
Patients should be informed that when VIDEX is used in combination with other agents with similar toxicities, the incidence of adverse events may be higher than when VIDEX is used alone. These patients should be followed closely.
Patients should be cautioned about the use of medications or other substances, including alcohol, that may exacerbate VIDEX toxicities.
VIDEX (didanosine) is not a cure for HIV infection, and patients may continue to develop HIV-associated illnesses, including opportunistic infection. Therefore, patients should remain under the care of a physician when using VIDEX. Patients should be advised that VIDEX therapy has not been shown to reduce the risk of transmission of HIV to others through sexual contact or blood contamination. Patients should be informed that the long-term effects of VIDEX are unknown at this time.
Patients should be informed that redistribution or accumulation of body fat may occur in patients receiving antiretroviral therapy and that the cause and long-term health effects of these conditions are not known at this time.
Drug Interactions (see also CLINICAL PHARMACOLOGY: Drug Interactions)
Drug interactions that have been established based on drug interaction studies are listed with the pharmacokinetic results in CLINICAL PHARMACOLOGY: Drug Interactions (Tables 3 and 4). The clinical recommendations based on the results of these studies are listed in Table 6.
| ↑ indicates increase. | ||
| ↓ indicates decrease. | ||
| Coadministration Not Recommended Based on Drug Interaction Studies (see CLINICAL PHARMACOLOGY: Drug Interactionsfor Magnitude of Interaction) | ||
| Drug | Effect | Clinical Comment |
| allopurinol | ↑ didanosine concentration | Coadministration not recommended. |
| Alteration in Dose or Regimen Recommended Based on Drug Interaction Studies(see CLINICAL PHARMACOLOGY: Drug Interactions for Magnitude of Interaction) | ||
| Drug | Effect | Clinical Comment |
| ciprofloxacin | ↓ ciprofloxacin concentration | Administer VIDEX at least 2 hours after or 6 hours before ciprofloxacin. |
| delavirdine | ↓ delavirdine concentration | Administer VIDEX 1 hour after delavirdine. |
| ganciclovir | ↑ didanosine concentration | Appropriate doses for this combination, with respect to efficacy and safety, have not been established. |
| indinavir | ↓ indinavir concentration | Administer VIDEX 1 hour after indinavir. |
| methadone | ↓ didanosine concentration | Appropriate doses for this combination, with respect to efficacy and safety, have not been established. |
| nelfinavir | No interaction 1 hour after didanosine | Administer nelfinavir 1 hour after VIDEX. |
| tenofovir disoproxil fumarate | ↑ didanosine concentration | A dose reduction of VIDEX to 250 mg (adults weighing ≥60 kg with creatinine clearance ≥60 mL/min) or 200 mg (adults weighing<60 kg with creatinine clearance ≥60 mL/min) once daily is recommended. VIDEX and tenofovir may be taken together in the fasted state. Alternatively, if tenofovir is taken with food, VIDEX should be taken on an empty stomach (at least 30 minutes before food or 2 hours after food). Patients should be monitored for didanosine-associated toxicities and clinical response (see below). |
Coadministration of VIDEX with drugs that are known to cause pancreatitis may increase the risk of this toxicity (see WARNINGS: Pancreatitis). Because VIDEX is mixed with an antacid before administration, interactions may be anticipated with drugs whose absorption can be affected by the level of acidity in the stomach and with drugs that have been demonstrated to interact with antacids containing magnesium, calcium, or aluminum. Predicted drug interactions with VIDEX are listed in Table 7.
| ↑ indicates increase. | ||
| ↓ indicates decrease. | ||
| a Only if other drugs are not available and if clearly indicated. If treatment with life-sustaining drugs that cause pancreatic toxicity is required, suspension of VIDEX is recommended (see WARNINGS: Pancreatitis). | ||
| b See PRECAUTIONS: Peripheral Neuropathy. | ||
| Use with Caution or Not Recommended, Risk of Adverse Reactions May Be Increased | ||
| Drug or Drug Class | Effect | Clinical Comment |
| Drugs that may cause pancreatic toxicity | ↑ risk of pancreatitis | Use only with extreme caution.a |
| Neurotoxic drugs | ↑ risk of neuropathy | Use with caution.b |
| Antacids containing magnesium or aluminum | ↑ side effects associated with antacid components | Use caution with VIDEX Pediatric Powder for Oral Solution. |
| Ribavirin | ↑ risk of toxicity | Ribavirin has been shown in vitro to increase intracellular triphosphate levels of didanosine. Coadministration is not recommended (see below). |
| Use with Caution, Plasma Concentrations May Be Decreased by Coadministration with VIDEX | ||
| Drug Class | Effect | Clinical Comment |
| Azole antifungals | ↓ ketoconazole or itraconazole concentration | Administer drugs such as ketoconazole or itraconazole at least 2 hours before VIDEX. |
| Quinolone antibiotics (see also ciprofloxacin in Table 6) | ↓ quinolone concentration | Consult package insert of the quinolone. |
| Tetracycline antibiotics | ↓ antibiotic concentration | Consult package insert of the tetracycline. |
Nucleoside/nucleotide analogues
Tenofovir disoproxil fumarate. Exposure to didanosine is increased when coadministered with tenofovir (see Table 3). Increased exposure may cause or worsen didanosine-related clinical toxicities, including pancreatitis, symptomatic hyperlactatemia/lactic acidosis, and peripheral neuropathy. Coadministration of tenofovir with VIDEX should be undertaken with caution, and patients should be monitored closely for didanosine-related toxicities and clinical response. VIDEX should be suspendedif signs or symptoms of pancreatitis, symptomatic hyperlactatemia, or lactic acidosis develop (see WARNINGS). A dose reduction of VIDEX (didanosine) to 250 mg (adults weighing ≥60 kg with creatinine clearance ≥60 mL/min) or 200 mg (adults weighing <60 kg with creatinine clearance ≥60 mL/min) once daily is recommended. VIDEX (didanosine) and tenofovir may be taken together in the fasted state. Alternatively, if tenofovir is taken with food, VIDEX should be taken on an empty stomach, at least 30 minutes before food or 2 hours after food (see DOSAGE AND ADMINISTRATION). (The dosing recommendation for coadministration of VIDEX EC and tenofovir disoproxil fumarate with respect to meal consumption differs from that of VIDEX. See the complete prescribing information for VIDEX EC.)
Ribavirin. Exposure to the active metabolite of didanosine (dideoxyadenosine 5′-triphosphate) is increased when didanosine is coadministered with ribavirin (see Table 7). Fatal hepatic failure, as well as peripheral neuropathy, pancreatitis, and symptomatic hyperlactatemia/lactic acidosis have been reported in patients receiving both didanosine and ribavirin. Coadministration of didanosine and ribavirin is not recommended.
Carcinogenesis and Mutagenesis
Lifetime carcinogenicity studies were conducted in mice and rats for 22 and 24 months, respectively. In the mouse study, initial doses of 120, 800, and 1200 mg/kg/day for each sex were lowered after 8 months to 120, 210, and 210 mg/kg/day for females and 120, 300, and 600 mg/kg/day for males. The two higher doses exceeded the maximally tolerated dose in females and the high dose exceeded the maximally tolerated dose in males. The low dose in females represented 0.68-fold maximum human exposure and the intermediate dose in males represented 1.7-fold maximum human exposure based on relative AUC comparisons. In the rat study, initial doses were 100, 250, and 1000 mg/kg/day, and the high dose was lowered to 500 mg/kg/day after 18 months. The upper dose in male and female rats represented 3-fold maximum human exposure.
Didanosine induced no significant increase in neoplastic lesions in mice or rats at maximally tolerated doses.
Didanosine was positive in the following genetic toxicology assays: 1) the Escherichia coli tester strain WP2 uvrA bacterial mutagenicity assay; 2) the L5178Y/TK+/- mouse lymphoma mammalian cell gene mutation assay; 3) the in vitro chromosomal aberrations assay in cultured human peripheral lymphocytes; 4) the in vitro chromosomal aberrations assay in Chinese Hamster Lung cells; and 5) the BALB/c 3T3 in vitro transformation assay. No evidence of mutagenicity was observed in an Ames Salmonella bacterial mutagenicity assay or in rat and mouse in vivo micronucleus assays.
Pregnancy, Reproduction, and Fertility
Pregnancy Category B. Reproduction studies have been performed in rats and rabbits at doses up to 12 and 14.2 times the estimated human exposure (based upon plasma levels), respectively, and have revealed no evidence of impaired fertility or harm to the fetus due to didanosine. At approximately 12 times the estimated human exposure, didanosine was slightly toxic to female rats and their pups during mid and late lactation. These rats showed reduced food intake and body weight gains but the physical and functional development of the offspring was not impaired and there were no major changes in the F2 generation. A study in rats showed that didanosine and/or its metabolites are transferred to the fetus through the placenta. Animal reproduction studies are not always predictive of human response.
There are no adequate and well-controlled studies of didanosine in pregnant women. Didanosine should be used during pregnancy only if the potential benefit justifies the potential risk.
Fatal lactic acidosis has been reported in pregnant women who received the combination of didanosine and stavudine with other antiretroviral agents. It is unclear if pregnancy augments the risk of lactic acidosis/hepatic steatosis syndrome reported in nonpregnant individuals receiving nucleoside analogues (see WARNINGS: Lactic Acidosis/Severe Hepatomegaly with Steatosis). The combination of didanosine and stavudine should be used with caution during pregnancy and is recommended only if the potential benefit clearly outweighs the potential risk. Health care providers caring for HIV-infected pregnant women receiving didanosine should be alert for early diagnosis of lactic acidosis/hepatic steatosis syndrome.
Antiretroviral Pregnancy Registry: To monitor maternal-fetal outcomes of pregnant women exposed to didanosine and other antiretroviral agents, an Antiretroviral Pregnancy Registry has been established. Physicians are encouraged to register patients by calling 1-800-258-4263.
Nursing Mothers
The Centers for Disease Control and Prevention recommend that HIV-infected mothers not breast-feed their infants to avoid risking postnatal transmission of HIV. A study in rats showed that following oral administration, didanosine and/or its metabolites were excreted into the milk of lactating rats. It is not known if didanosine is excreted in human milk. Because of both the potential for HIV transmission and the potential for serious adverse reactions in nursing infants, mothers should be instructed not to breast-feed if they are receiving VIDEX.
Pediatric Use
Use of VIDEX in pediatric patients from 2 weeks of age through adolescence is supported by evidence from adequate and well-controlled studies of VIDEX in adults and pediatric patients (see INDICATIONS AND USAGE: Clinical Studies, CLINICAL PHARMACOLOGY, ADVERSE REACTIONS, and DOSAGE AND ADMINISTRATION).
Dosing recommendations for VIDEX in patients less than 2 weeks of age cannot be made because the pharmacokinetics of didanosine in these children are too variable to determine an appropriate dose.
Geriatric Use
In an Expanded Access Program for patients with advanced HIV infection, patients aged 65 years and older had a higher frequency of pancreatitis (10%) than younger patients (5%) (see WARNINGS). Clinical studies of didanosine did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently than younger subjects. Didanosine is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection. In addition, renal function should be monitored and dosage adjustments should be made accordingly (see DOSAGE AND ADMINISTRATION: Dose Adjustment).
ADVERSE REACTIONS
A SERIOUS TOXICITY OF VIDEX (didanosine) IS PANCREATITIS, WHICH MAY BE FATAL (see WARNINGS). OTHER IMPORTANT TOXICITIES INCLUDE LACTIC ACIDOSIS/SEVERE HEPATOMEGALY WITH STEATOSIS; RETINAL CHANGES AND OPTIC NEURITIS; AND PERIPHERAL NEUROPATHY (see WARNINGSand PRECAUTIONS).
When VIDEX is used in combination with other agents with similar toxicities, the incidence of these toxicities may be higher than when VIDEX is used alone. Thus, patients treated with VIDEX in combination with stavudine, with or without hydroxyurea, may be at increased risk for pancreatitis and hepatotoxicity, which may be fatal, and severe peripheral neuropathy (see WARNINGS and PRECAUTIONS).
Adults: Selected clinical adverse events that occurred in adult patients in clinical studies with VIDEX are provided in Tables 8 and 9.
| Percent of Patients | ||||
|---|---|---|---|---|
| ACTG 116A | ACTG 116B/117 | |||
| Adverse Events | VIDEX n=197 | zidovudine n=212 | VIDEX n=298 | zidovudine n=304 |
| Diarrhea | 19 | 15 | 28 | 21 |
| Peripheral Neurologic Symptoms/Neuropathy | 17 | 14 | 20 | 12 |
| Rash/Pruritus | 7 | 8 | 9 | 5 |
| Abdominal Pain | 13 | 8 | 7 | 8 |
| Pancreatitis | 7 | 3 | 6 | 2 |
| Percent of Patientsa | ||||
|---|---|---|---|---|
| a Percentages based on treated subjects. | ||||
| b Median duration of treatment 48 weeks. | ||||
| * This event was not observed in this study arm. | ||||
| AI454-148b | START 2b | |||
|
Adverse Events | VIDEX + stavudine + nelfinavir n=482 | zidovudine + lamivudine + nelfinavir n=248 | VIDEX + stavudine + indinavir n=102 | zidovudine + lamivudine + indinavir n=103 |
| Diarrhea | 70 | 60 | 45 | 39 |
| Nausea | 28 | 40 | 53 | 67 |
| Headache | 21 | 30 | 46 | 37 |
| Peripheral Neurologic Symptoms/Neuropathy | 26 | 6 | 21 | 10 |
| Rash | 13 | 16 | 30 | 18 |
| Vomiting | 12 | 14 | 30 | 35 |
| Pancreatitis (see below) | 1 | * | <1 | * |
Pancreatitis resulting in death was observed in one patient who received VIDEX (didanosine) plus stavudine plus nelfinavir in Study Al454-148 and in one patient who received VIDEX plus stavudine plus indinavir in the START 2 study. In addition, pancreatitis resulting in death was observed in 2 of 68 patients who received VIDEX plus stavudine plus indinavir plus hydroxyurea in an ACTG clinical trial (see WARNINGS).
The frequency of pancreatitis is dose related. In phase 3 studies, incidence ranged from 1% to 10% with doses higher than are currently recommended and from 1% to 7% with recommended dose.
Selected laboratory abnormalities in clinical studies with VIDEX are shown in Tables 10-12.
| ULN = upper limit of normal. | ||||
| Percent of Patients | ||||
| ACTG 116A | ACTG 116B/117 | |||
| Parameter | VIDEX n=197 | zidovudine n=212 | VIDEX n=298 | zidovudine n=304 |
| SGOT (AST) (>5 x ULN) | 9 | 4 | 7 | 6 |
| SGPT (ALT) (>5 x ULN) | 9 | 6 | 6 | 6 |
| Alkaline phosphatase (>5 x ULN) | 4 | 1 | 1 | 1 |
| Amylase (≥1.4 x ULN) | 17 | 12 | 15 | 5 |
| Uric acid (>12 mg/dL) | 3 | 1 | 2 | 1 |
| Percent of Patientsa | ||||
|---|---|---|---|---|
| ULN = upper limit of normal. | ||||
| NC = Not Collected. | ||||
| a Percentages based on treated subjects. | ||||
| b Median duration of treatment 48 weeks. | ||||
| AI454-148b | START 2b | |||
| Parameter | VIDEX + stavudine + nelfinavir n=482 | zidovudine + lamivudine + nelfinavir n=248 | VIDEX + stavudine + indinavir n=102 | zidovudine + lamivudine + indinavir n=103 |
| Bilirubin (>2.6 x ULN) | <1 | <1 | 16 | 8 |
| SGOT (AST) (>5 x ULN) | 3 | 2 | 7 | 7 |
| SGPT (ALT) (>5 x ULN) | 3 | 3 | 8 | 5 |
| GGT (>5 x ULN) | NC | NC | 5 | 2 |
| Lipase (>2 x ULN) | 7 | 2 | 5 | 5 |
| Amylase (>2 x ULN) | NC | NC | 8 | 2 |
| Percent of Patientsa | ||||
|---|---|---|---|---|
| AI454-148b | START 2b | |||
| Parameter | VIDEX + stavudine + nelfinavir n=482 | zidovudine + lamivudine + nelfinavir n=248 | VIDEX + stavudine + indinavir n=102 | zidovudine + lamivudine + indinavir n=103 |
| NC = Not Collected. | ||||
| a Percentages based on treated subjects. | ||||
| b Median duration of treatment 48 weeks. | ||||
| Bilirubin | 7 | 3 | 68 | 55 |
| SGOT (AST) | 42 | 23 | 53 | 20 |
| SGPT (ALT) | 37 | 24 | 50 | 18 |
| GGT | NC | NC | 28 | 12 |
| Lipase | 17 | 11 | 26 | 19 |
| Amylase | NC | NC | 31 | 17 |
Observed During Clinical Practice: The following events have been identified during postapproval use of VIDEX (didanosine). Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to their seriousness, frequency of reporting, causal connection to VIDEX, or a combination of these factors.
- Body as a Whole – alopecia, anaphylactoid reaction, asthenia, chills/fever, pain, and redistribution/accumulation of body fat (see PRECAUTIONS: Fat Redistribution).
-
Digestive Disorders – anorexia, dyspepsia, and flatulence. -
Exocrine Gland Disorders – pancreatitis (including fatal cases) (see WARNINGS), sialoadenitis, parotid gland enlargement, dry mouth, and dry eyes. -
Hematologic Disorders – anemia, leukopenia, and thrombocytopenia. -
Liver – symptomatic hyperlactatemia/lactic acidosis and hepatic steatosis (see WARNINGS); hepatitis and liver failure. -
Metabolic Disorders – diabetes mellitus, hypoglycemia, and hyperglycemia. -
Musculoskeletal Disorders – myalgia (with or without increases in creatine kinase), rhabdomyolysis including acute renal failure and hemodialysis, arthralgia, and myopathy. -
Ophthalmologic Disorders – Retinal depigmentation and optic neuritis (see WARNINGS).
Pediatric Patients: In clinical trials, 743 pediatric patients between 2 weeks and 18 years of age have been treated with VIDEX. Adverse events and laboratory abnormalities reported to occur in these patients were generally consistent with the safety profile of didanosine in adults.
In pediatric phase 1 studies, pancreatitis occurred in 2 of 60 (3%) patients treated at entry doses below 300 mg/m2/day and in 5 of 38 (13%) patients treated at higher doses. In study ACTG 152, pancreatitis occurred in none of the 281 pediatric patients who received didanosine 120 mg/m2 q12h and in <1% of the 274 pediatric patients who received didanosine 90 mg/m2 q12h in combination with zidovudine (see INDICATIONS AND USAGE: Clinical Studies).
Retinal changes and optic neuritis have been reported in pediatric patients.
OVERDOSAGE
There is no known antidote for VIDEX (didanosine) overdosage. In phase 1 studies, in which VIDEX was initially administered at doses ten times the currently recommended dose, toxicities included: pancreatitis, peripheral neuropathy, diarrhea, hyperuricemia, and hepatic dysfunction. Didanosine is not dialyzable by peritoneal dialysis, although there is some clearance by hemodialysis (see CLINICAL PHARMACOLOGY: Pharmacokinetics).
DOSAGE AND ADMINISTRATION
Dosage
VIDEX should be administered on an empty stomach, at least 30 minutes before or 2 hours after eating.
Adults: The preferred dosing frequency of VIDEX is twice daily because there is more evidence to support the effectiveness of this dosing regimen. Once-daily dosing should be considered only for adult patients whose management requires once-daily dosing of VIDEX (see INDICATIONS AND USAGE: Clinical Studies). The daily dose in adult patients is dependent on weight as outlined in Table 13.
| Recommended VIDEX Dose by Patient Weight | ||
|---|---|---|
| ≥60 kg | <60 kg | |
| Preferred dosing | 200 mg twice daily | 125 mg twice daily |
| Dosing for patients whose management requires once-daily frequency | 400 mg once daily | 250 mg once daily |
Pediatric Patients: The recommended dose of VIDEX (didanosine) in pediatric patients between 2 weeks and 8 months of age is 100 mg/m2 twice daily, and the recommended VIDEX dose for pediatric patients older than 8 months is 120 mg/m2 twice daily.
Dosing recommendations for VIDEX in patients less than 2 weeks of age cannot be made because the pharmacokinetics of didanosine in these children are too variable to determine an appropriate dose. There are no data on once-daily dosing of VIDEX in pediatric patients.
Dose Adjustment
Clinical and laboratory signs suggestive of pancreatitis should prompt dose suspension and careful evaluation of the possibility of pancreatitis. VIDEX use should be discontinued in patients with confirmed pancreatitis (see WARNINGS and PRECAUTIONS: Drug Interactions).
Patients with symptoms of peripheral neuropathy may tolerate a reduced dose of VIDEX after resolution of the symptoms of peripheral neuropathy upon drug discontinuation. If neuropathy recurs after resumption of VIDEX, permanent discontinuation of VIDEX should be considered.
Concomitant Therapy: Tenofovir disoproxil fumarate. A dose reduction of VIDEX to 250 mg (adults weighing ≥60 kg with creatinine clearance ≥60 mL/min) or 200 mg (adults weighing<60 kg with creatinine clearance ≥60 mL/min) once daily is recommended. VIDEX and tenofovir may be taken together in the fasted state. Alternatively, if tenofovir is taken with food, VIDEX should be taken on an empty stomach (at least 30 minutes before food or 2 hours after food). The appropriate dose of VIDEX coadministered with tenofovir in patients with creatinine clearance<60 mL/min has not been established. [See CLINICAL PHARMACOLOGY: Drug Interactions and PRECAUTIONS: Drug Interactions; see the complete prescribing information for VIDEX EC (enteric-coated formulation of didanosine) for results of drug interaction studies of tenofovir with reduced doses of the enteric-coated formulation of didanosine.]
Renal Impairment: In adult patients with impaired renal function, the dose of VIDEX should be adjusted to compensate for the slower rate of elimination. The recommended doses and dosing intervals of VIDEX in adult patients with renal insufficiency are presented in Table 14.
| Recommended VIDEX Dose by Patient Weight | ||
|---|---|---|
| Creatinine Clearance (mL/min) | ≥60 kg | <60 kg |
| a 400 mg once daily (≥60 kg) or 250 mg once daily (<60 kg) for patients whose management requires once-daily frequency of administration. | ||
| ≥60 | 200 mg twice dailya | 125 twice dailya |
| 30-59 | 200 mg once daily or 100 mg twice daily | 150 mg once daily or 75 mg twice daily |
| 10-29 | 150 mg once daily | 100 mg once daily |
| <10 | 100 mg once daily | 75 mg once daily |
Urinary excretion is also a major route of elimination of didanosine in pediatric patients; therefore, the clearance of didanosine may be altered in children with renal impairment. Although there are insufficient data to recommend a specific dose adjustment of VIDEX in this patient population, a reduction in the dose and/or an increase in the interval between doses should be considered.
Patients Requiring Continuous Ambulatory Peritoneal Dialysis (CAPD) or Hemodialysis: For patients requiring CAPD or hemodialysis, follow dosing recommendations for patients with creatinine clearance less than 10 mL/min, shown in Table 14. It is not necessary to administer a supplemental dose of VIDEX following hemodialysis.
Hepatic Impairment and Toxicity: See WARNINGS.
Method of Preparation
VIDEX Pediatric Powder for Oral Solution
Prior to dispensing, the pharmacist must constitute dry powder with Purified Water, USP, to an initial concentration of 20 mg/mL and immediately mix the resulting solution with antacid to a final concentration of 10 mg/mL as follows:
20 mg/mL Initial Solution: Constitute the product to 20 mg/mL by adding 100 mL or 200 mL of Purified Water, USP, to the 2 g or 4 g of VIDEX powder, respectively, in the product bottle.
10 mg/mL Final Admixture: 1. Immediately mix one part of the 20 mg/mL initial solution with one part of Maximum Strength Mylanta® Liquid for a final dispensing concentration of 10 mg VIDEX per mL. For patient home use, the admixture should be dispensed in appropriately sized, flint-glass or plastic (HDPE, PET, or PETG) bottles with child-resistant closures. This admixture is stable for 30 days under refrigeration, 36° to 46° F (2° to 8° C).
2. Instruct the patient to shake the admixture thoroughly prior to use and to store the tightly closed container in the refrigerator, 36° to 46° F (2° to 8° C), up to 30 days.
HOW SUPPLIED
VIDEX (didanosine) Pediatric Powder for Oral Solution is supplied in 4- and 8-ounce glass bottles containing 2 g or 4 g of VIDEX, respectively.
The bottles of powder should be stored at 59° to 86° F (15° to 30° C). The VIDEX admixture may be stored up to 30 days in a refrigerator, 36° to 46° F (2° to 8° C). Discard any unused portion after 30 days.
The NDC numbers for the previously described VIDEX products are:
| NDC NO. | Packaging Information | Product Strength |
|---|---|---|
| VIDEX® Pediatric Powder for Oral Solution | ||
| 0087-6632-41 | One bottle per carton | 2 g/bottle |
| 0087-6633-41 | One bottle per carton | 4 g/bottle |
US Patent Nos: 4,861,759, 5,254,539, 5,616,566, and 5,880,106.
HANDLING AND DISPOSAL
Spill, Leak, and Disposal Procedure
Avoid generating dust during clean-up of powdered products; use wet mop or damp sponge. Clean surface with soap and water as necessary. Containerize larger spills.
There is no single preferred method of disposal of containerized waste. Disposal options include incineration, landfill, or sewer as dictated by specific circumstances and relevant national, state, and local regulations.
______________________
Mylanta® is a registered trademark of Johnson & Johnson-Merck Consumer Pharmaceuticals Company.
Bristol-Myers Squibb Company
Princeton,
NJ 08543 USA
1196181A4
Revised January 2007
PATIENT INFORMATION
VIDEX®
(generic name = didanosine also known as ddI)
VIDEX® (didanosine) Pediatric Powder for Oral Solution
What is VIDEX?
VIDEX (pronounced VY dex) is a prescription medicine used in combination with other drugs to treat children and adults who are infected with HIV (the human immunodeficiency virus, the virus that causes AIDS). VIDEX belongs to a class of drugs called nucleoside analogues. By reducing the growth of HIV,VIDEX helps your body maintain its supply of CD4 cells, which are important for fighting HIV and other infections.
VIDEX will not cure your HIV infection. At present there is no cure for HIV infection. Even while taking VIDEX, you may continue to have HIV-related illnesses, including infections with other disease-producing organisms. Continue to see your doctor regularly and report any medical problems that occur.
VIDEX does not prevent a patient infected with HIV from passing the virus to other people. To protect others, you must continue to practice safe sex and take precautions to prevent others from coming in contact with your blood and other body fluids.
There is limited information on the effects of long-term use of VIDEX.
Who should not take VIDEX?
Do not take VIDEX if you are allergic to any of its ingredients, including its active ingredient, didanosine and the inactive ingredients. (See Inactive Ingredients at the end of this leaflet.) Tell your doctor if you think you have had an allergic reaction to any of these ingredients.
How should I take VIDEX? How should I store it?
Your doctor will determine your dose based on your body weight,
kidney and liver function, other medicines you are taking, and any side effects
that you may have had with VIDEX or other medicines. Take VIDEX on an
empty stomach - that means at least 30 minutes before or 2 hours after eating.
Do not take VIDEX with food. Try not to miss a dose, but if you do,
take it as soon as possible. If it is almost time for the next dose, skip
the missed dose and continue your regular dosing schedule.
- Your pharmacist will prepare the oral solution. Shake the solution well before each use. Store in the refrigerator. Throw away any unused portion after 30 days.
If you have kidney disease: If your kidneys are not working properly, your doctor will need to do regular tests to check how they are working while you take VIDEX. Your doctor may also lower your dosage of VIDEX.
What should I do if someone takes an overdose of VIDEX?
If someone may have taken an overdose of VIDEX, get medical help right away. Contact their doctor or a poison control center.
What should I avoid while taking VIDEX?
Alcohol. Do not drink alcohol while taking VIDEX since alcohol may increase your risk of pancreatitis (pain and inflammation of the pancreas) or liver damage.
Other medicines. Other medicines, including those
you can buy without a prescription, may interfere with the actions of VIDEX
or may increase the possibility or severity of side effects. Do not
take any medicine, vitamin supplement, or other health preparation without
first checking with your doctor.
- Antacids. Since VIDEX is mixed with an antacid, any side effects related to VIDEX’s ingredients may get worse if you also take an antacid.
- Medicines at the same time you take your VIDEX dose. Some medicines should not be taken at the same time of day that you take VIDEX. Check with your doctor.
Pregnancy. It is not known if VIDEX can harm a human fetus. Also, pregnant women have experienced serious side effects when taking VIDEX in combination with ZERIT (stavudine), also known as d4T, and other HIV medicines. VIDEX should be used during pregnancy only after discussion with your doctor. Tell your doctor if you become pregnant or plan to become pregnant while taking VIDEX.
Nursing. Studies have shown VIDEX is in the breast milk of animals getting the drug. It may also be in human breast milk. The Centers for Disease Control and Prevention (CDC) recommends that HIV-infected mothers not breast-feed. This should reduce the risk of passing HIV infection to their babies and the potential for serious adverse reactions in nursing infants. Therefore, do not nurse a baby while taking VIDEX.
What are the possible side effects of VIDEX?
Pancreatitis. Pancreatitis is a dangerous inflammation of the pancreas that may cause death. Tell your doctor right away if you or a child taking VIDEX develops stomach pain, nausea, or vomiting. These can be signs of pancreatitis. Before starting VIDEX therapy, let your doctor know if you or a child for whom it has been prescribed has ever had pancreatitis. This condition is more likely to happen in people who have had it before. It is also more likely in people with advanced HIV disease. However, it can occur at any stage of HIV disease. It may be more common in patients with kidney problems, those who drink alcohol, and those who are also treated with stavudine or hydroxyurea. If you get pancreatitis, your doctor will tell you to stop taking VIDEX.
Lactic acidosis, severe liver enlargement, and liver failure, including deaths, have been reported among patients taking VIDEX (including pregnant women). Symptoms that may indicate a liver problem are:
- feeling very weak, tired, or uncomfortable,
- unusual or unexpected stomach discomfort,
- feeling cold,
- feeling dizzy or lightheaded,
- suddenly developing a slow or irregular heartbeat.
Lactic acidosis is a medical emergency that must be treated in a hospital.
If you notice any of these symptoms or if your medical condition changes, stop taking VIDEX and call your doctor right away. Women, overweight patients, and those who have been treated for a long time with other medicines used to treat HIV infection are more likely to develop lactic acidosis. Your doctor should check your liver function periodically while you are taking VIDEX. You should be especially careful if you have a history of heavy alcohol use or a liver problem.
Vision changes. VIDEX (didanosine) may affect the nerves in your eyes. Because of this, you should have regular eye examinations. You should also report any changes in vision to your doctor right away. This includes, for example, seeing colors abnormally or blurred vision.
Peripheral neuropathy. This is a problem with the nerves in your hands or feet. The nerve problem may be serious. Tell your doctor right away if you or a child taking VIDEX has continuing numbness, tingling, or pain in the feet or hands.A child may not recognize these symptoms or know to tell you that his or her feet or hands are numb, burning, tingling, or painful. Ask your child’s doctor how to find out if your child is developing peripheral neuropathy.
Before starting VIDEX therapy, let your doctor know if you or a child for whom it has been prescribed has ever had peripheral neuropathy. This condition is more likely to happen in people who have had it before. It is also more likely in patients taking medicines that affect the nerves and in people with advanced HIV disease. However, it can occur at any stage of HIV disease. If you get peripheral neuropathy, your doctor will tell you to stop taking VIDEX. After stopping VIDEX, the symptoms may get worse for a short time and then get better. Once symptoms of peripheral neuropathy go away completely, you and your doctor should decide if starting VIDEX is right for you. If so, you might be started at a lower dose.
Special note about other medicines. If you take VIDEX along with other medicines with similar side effects, you may increase the chance of having these side effects. For example, using VIDEX in combination with other medicines that may cause pancreatitis, peripheral neuropathy, or liver problems (including stavudine and hydroxyurea) may increase your chance of having these side effects.
Other side effects: The most common side effects in adults taking VIDEX are diarrhea, neuropathy (nerve disorders), chills or fever, rash, abdominal pain, weakness, headache, and nausea and vomiting. Children may have similar side effects as adults.
Changes in body fat have been seen in some patients taking antiretroviral therapy. These changes may include increased amount of fat in the upper back and neck (“buffalo hump”), breast, and around the trunk. Loss of fat from the legs, arms, and face may also happen. The cause and long-term health effects of these conditions are not known at this time.
Inactive Ingredients:
- Pediatric Oral Solution: Maximum Strength Mylanta® Liquid.
- __________________
- This medicine was prescribed for your particular condition. Do not use VIDEX for another condition or give it to others. Keep VIDEX and all medicines out of the reach of children. Throw away VIDEX when it is outdated or no longer needed by flushing it down the toilet or pouring it down the sink.
This summary does not include everything there is to know about VIDEX. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information Leaflet. If you have questions or concerns, or want more information about VIDEX, your physician and pharmacist have the complete prescribing information upon which this leaflet is based. You may want to read it and discuss it with your doctor or other healthcare professional. Remember, no written summary can replace careful discussion with your doctor.
Mylanta® is a registered trademark of Johnson & Johnson-Merck Consumer Pharmaceuticals Company.
Bristol-Myers Squibb Company
Princeton,
NJ 08543 USA
This Patient Information Leaflet has been approved
by the U.S. Food and Drug Administration.
1196181A4
Revised
January 2007
| VIDEX (DIDANOSINE) | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| VIDEX (DIDANOSINE) | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
Revised: 01/2008Bristol-Myers Squibb