celebrex (Celecoxib) capsule
[Pfizer Inc]
Cardiovascular Risk
- CELEBREX may cause an increased risk of serious cardiovascular thrombotic events, myocardial infarction, and stroke, which can be fatal. All NSAIDs may have a similar risk. This risk may increase with duration of use. Patients with cardiovascular disease or risk factors for cardiovascular disease may be at greater risk (see WARNINGS and CLINICAL STUDIES).
- CELEBREX is contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery (see WARNINGS).
Gastrointestinal Risk
- NSAIDs, including CELEBREX, cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events (see WARNINGS).
DESCRIPTION
CELEBREX (celecoxib) is chemically designated as 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl] benzenesulfonamide and is a diaryl-substituted pyrazole. It has the following chemical structure:
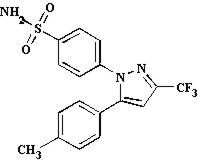
The empirical formula for celecoxib is C17H14F3N3O2S, and the molecular weight is 381.38.
CELEBREX oral capsules contain either 50 mg, 100 mg, 200 mg or 400 mg of celecoxib.
The inactive ingredients in CELEBREX capsules include: croscarmellose sodium, edible inks, gelatin, lactose monohydrate, magnesium stearate, povidone and sodium lauryl sulfate.
CLINICAL PHARMACOLOGY
Mechanism of Action
CELEBREX is a nonsteroidal anti-inflammatory drug that exhibits anti-inflammatory, analgesic, and antipyretic activities in animal models. The mechanism of action of CELEBREX is believed to be due to inhibition of prostaglandin synthesis, primarily via inhibition of cyclooxygenase-2 (COX-2), and at therapeutic concentrations in humans, CELEBREX does not inhibit the cyclooxygenase-1 (COX-1) isoenzyme. In animal colon tumor models, celecoxib reduced the incidence and multiplicity of tumors.
Platelets
In clinical trials using normal volunteers, CELEBREX at single doses up to 800 mg and multiple doses of 600 mg twice daily for up to 7 days duration (higher than recommended therapeutic doses) had no effect on reduction of platelet aggregation or increase in bleeding time. Because of its lack of platelet effects, CELEBREX is not a substitute for aspirin for cardiovascular prophylaxis. It is not known if there are any effects of CELEBREX on platelets that may contribute to the increased risk of serious cardiovascular thrombotic adverse events associated with the use of CELEBREX.
Fluid Retention
Inhibition of PGE2 synthesis may lead to sodium and water retention through increased reabsorption in the renal medullary thick ascending loop of Henle and perhaps other segments of the distal nephron. In the collecting ducts, PGE2 appears to inhibit water reabsorption by counteracting the action of antidiuretic hormone.
Pharmacokinetics
Absorption
Peak plasma levels of celecoxib occur approximately 3 hrs after an oral dose. Under fasting conditions, both peak plasma levels (Cmax) and area under the curve (AUC) are roughly dose proportional up to 200 mg BID; at higher doses there are less than proportional increases in Cmax and AUC (see Food Effects). Absolute bioavailability studies have not been conducted. With multiple dosing, steady state conditions are reached on or before Day 5.
The pharmacokinetic parameters of celecoxib in a group of healthy subjects are shown in Table 1.
| Mean (%CV) PK Parameter Values | ||||
|---|---|---|---|---|
| Cmax, ng/mL | Tmax, hr | Effective t1/2, hr | Vss/F, L | CL/F, L/hr |
|
||||
| 705 (38) | 2.8 (37) | 11.2 (31) | 429 (34) | 27.7 (28) |
Food Effects
When CELEBREX capsules were taken with a high fat meal, peak plasma levels were delayed for about 1 to 2 hours with an increase in total absorption (AUC) of 10% to 20%. Under fasting conditions, at doses above 200 mg, there is less than a proportional increase in Cmax and AUC, which is thought to be due to the low solubility of the drug in aqueous media. Coadministration of CELEBREX with an aluminum- and magnesium-containing antacid resulted in a reduction in plasma celecoxib concentrations with a decrease of 37% in Cmax and 10% in AUC. CELEBREX, at doses up to 200 mg BID can be administered without regard to timing of meals. Higher doses (400 mg BID) should be administered with food to improve absorption.
In healthy adult volunteers, the overall systemic exposure (AUC) of celecoxib was equivalent when celecoxib was administered as intact capsule or capsule contents sprinkled on applesauce. There were no significant alterations in Cmax, Tmax or T1/2 after administration of capsule contents on applesauce.
Distribution
In healthy subjects, celecoxib is highly protein bound (~97%) within the clinical dose range. In vitro studies indicate that celecoxib binds primarily to albumin and, to a lesser extent, α1-acid glycoprotein. The apparent volume of distribution at steady state (Vss/F) is approximately 400 L, suggesting extensive distribution into the tissues. Celecoxib is not preferentially bound to red blood cells.
Metabolism
Celecoxib metabolism is primarily mediated via cytochrome P450 2C9. Three metabolites, a primary alcohol, the corresponding carboxylic acid and its glucuronide conjugate, have been identified in human plasma. These metabolites are inactive as COX-1 or COX-2 inhibitors. Patients who are known or suspected to be P450 2C9 poor metabolizers based on a previous history should be administered celecoxib with caution as they may have abnormally high plasma levels due to reduced metabolic clearance.
Excretion
Celecoxib is eliminated predominantly by hepatic metabolism with little (<3%) unchanged drug recovered in the urine and feces. Following a single oral dose of radiolabeled drug, approximately 57% of the dose was excreted in the feces and 27% was excreted into the urine. The primary metabolite in both urine and feces was the carboxylic acid metabolite (73% of dose) with low amounts of the glucuronide also appearing in the urine. It appears that the low solubility of the drug prolongs the absorption process making terminal half-life (t1/2) determinations more variable. The effective half-life is approximately 11 hours under fasted conditions. The apparent plasma clearance (CL/F) is about 500 mL/min.
Special Populations
Geriatric
At steady state, elderly subjects (over 65 years old) had a 40% higher Cmax and a 50% higher AUC compared to the young subjects. In elderly females, celecoxib Cmax and AUC are higher than those for elderly males, but these increases are predominantly due to lower body weight in elderly females. Dose adjustment in the elderly is not generally necessary. However, for patients of less than 50 kg in body weight, initiate therapy at the lowest recommended dose.
Pediatric
The steady state pharmacokinetics of celecoxib administered as an investigational oral suspension was evaluated in 152 juvenile rheumatoid arthritis (JRA) patients 2 years to 17 years of age weighing ≥10 kg with pauciarticular or polyarticular course JRA and in patients with systemic onset JRA. Population pharmacokinetic analysis indicated that the oral clearance (unadjusted for body weight) of celecoxib increases less than proportionally to increasing weight, with 10 kg and 25 kg patients predicted to have 40% and 24% lower clearance, respectively, compared with a 70 kg adult RA patient.
Twice-daily administration of 50 mg capsules to JRA patients weighing ≥12 to ≤25 kg and 100 mg capsules to JRA patients weighing >25 kg should achieve plasma concentrations similar to those observed in a clinical trial that demonstrated the non-inferiority of celecoxib to naproxen 7.5 mg/kg twice daily (see DOSAGE AND ADMINISTRATION). Celecoxib has not been studied in JRA patients under the age of 2 years, in patients with body weight less than 10 kg (22 lbs), or beyond 24 weeks.
Race
Meta-analysis of pharmacokinetic studies has suggested an approximately 40% higher AUC of celecoxib in Blacks compared to Caucasians. The cause and clinical significance of this finding is unknown.
Hepatic Insufficiency
A pharmacokinetic study in subjects with mild (Child-Pugh Class A) and moderate (Child-Pugh Class B) hepatic impairment has shown that steady-state celecoxib AUC is increased about 40% and 180%, respectively, above that seen in healthy control subjects. Therefore, the daily recommended dose of CELEBREX capsules should be reduced by approximately 50% in patients with moderate (Child-Pugh Class B) hepatic impairment. Patients with severe hepatic impairment (Child-Pugh Class C) have not been studied. The use of CELEBREX in patients with severe hepatic impairment is not recommended (see DOSAGE AND ADMINISTRATION).
Renal Insufficiency
In a cross-study comparison, celecoxib AUC was approximately 40% lower in patients with chronic renal insufficiency (GFR 35–60 mL/min) than that seen in subjects with normal renal function. No significant relationship was found between GFR and celecoxib clearance. Patients with severe renal insufficiency have not been studied. Similar to other NSAIDs, CELEBREX is not recommended in patients with severe renal insufficiency (see WARNINGS – Advanced Renal Disease).
Drug Interactions
Also see PRECAUTIONS – Drug Interactions.
General
Significant interactions may occur when celecoxib is administered together with drugs that inhibit P450 2C9. In vitro studies indicate that celecoxib is not an inhibitor of cytochrome P450 2C9, 2C19 or 3A4.
Clinical studies with celecoxib have identified potentially significant interactions with fluconazole and lithium. Experience with nonsteroidal anti-inflammatory drugs (NSAIDs) suggests the potential for interactions with furosemide and ACE inhibitors. The effects of celecoxib on the pharmacokinetics and/or pharmacodynamics of glyburide, ketoconazole, methotrexate, phenytoin, and tolbutamide have been studied in vivo and clinically important interactions have not been found.
CLINICAL STUDIES
Osteoarthritis (OA)
CELEBREX has demonstrated significant reduction in joint pain compared to placebo. CELEBREX was evaluated for treatment of the signs and the symptoms of OA of the knee and hip in placebo- and active-controlled clinical trials of up to 12 weeks duration. In patients with OA, treatment with CELEBREX 100 mg BID or 200 mg QD resulted in improvement in WOMAC (Western Ontario and McMaster Universities) osteoarthritis index, a composite of pain, stiffness, and functional measures in OA. In three 12-week studies of pain accompanying OA flare, CELEBREX doses of 100 mg BID and 200 mg BID provided significant reduction of pain within 24–48 hours of initiation of dosing. At doses of 100 mg BID or 200 mg BID the effectiveness of CELEBREX was shown to be similar to that of naproxen 500 mg BID. Doses of 200 mg BID provided no additional benefit above that seen with 100 mg BID. A total daily dose of 200 mg has been shown to be equally effective whether administered as 100 mg BID or 200 mg QD.
Rheumatoid Arthritis (RA)
CELEBREX has demonstrated significant reduction in joint tenderness/pain and joint swelling compared to placebo. CELEBREX was evaluated for treatment of the signs and symptoms of RA in placebo- and active-controlled clinical trials of up to 24 weeks in duration. CELEBREX was shown to be superior to placebo in these studies, using the ACR20 Responder Index, a composite of clinical, laboratory, and functional measures in RA. CELEBREX doses of 100 mg BID and 200 mg BID were similar in effectiveness and both were comparable to naproxen 500 mg BID.
Although CELEBREX 100 mg BID and 200 mg BID provided similar overall effectiveness, some patients derived additional benefit from the 200 mg BID dose. Doses of 400 mg BID provided no additional benefit above that seen with 100–200 mg BID.
Juvenile Rheumatoid Arthritis (JRA)
In a 12-week, randomized, double-blind active-controlled, parallel-group, multicenter, non-inferiority study, patients from 2 years to 17 years of age with pauciarticular, polyarticular course JRA or systemic onset JRA (with currently inactive systemic features), received one of the following treatments: celecoxib 3 mg/kg (to a maximum of 150 mg) twice daily; celecoxib 6 mg/kg (to a maximum of 300 mg) twice daily; or naproxen 7.5 mg/kg (to a maximum of 500 mg) twice daily. The response rates were based upon the JRA Definition of Improvement greater than or equal to 30% (JRA DOI 30) criterion, which is a composite of clinical, laboratory, and functional measures of JRA. The JRA DOI 30 response rates at week 12 were 69%, 80% and 67% in the celecoxib 3 mg/kg BID, celecoxib 6 mg/kg BID, and naproxen 7.5 mg/kg BID treatment groups, respectively.
The efficacy and safety of CELEBREX for JRA have not been studied beyond six months. The long-term cardiovascular toxicity in children exposed to CELEBREX has not been evaluated and it is unknown if the long-term risk may be similar to that seen in adults exposed to CELEBREX or other COX-2 selective and non-selective NSAIDS. (see Boxed Warning, WARNINGS, and PRECAUTIONS)
Analgesia, including primary dysmenorrhea
In acute analgesic models of post-oral surgery pain, post-orthopedic surgical pain, and primary dysmenorrhea, CELEBREX relieved pain that was rated by patients as moderate to severe. Single doses (see DOSAGE AND ADMINISTRATION) of CELEBREX provided pain relief within 60 minutes.
Ankylosing Spondylitis (AS)
CELEBREX was evaluated in AS patients in two placebo- and active-controlled clinical trials of 6 and 12 weeks duration. CELEBREX at doses of 100 mg BID, 200 mg QD and 400 mg QD was shown to be statistically superior to placebo in these studies for all three co-primary efficacy measures assessing global pain intensity (Visual Analogue Scale), global disease activity (Visual Analogue Scale) and functional impairment (Bath Ankylosing Spondylitis Functional Index). In the 12-week study, there was no difference in the extent of improvement between the 200 mg and 400 mg celecoxib doses in a comparison of mean change from baseline, but there was a greater percentage of patients who responded to celecoxib 400 mg, 53%, than to celecoxib 200 mg, 44%, using the Assessment in Ankylosing Spondylitis response criteria (ASAS 20). The ASAS 20 defines a responder as improvement from baseline of at least 20% and an absolute improvement of at least 10 mm, on a 0 to 100 mm scale, in at least three of the four following domains: patient global, pain, Bath Ankylosing Spondylitis Functional Index, and inflammation. The responder analysis also demonstrated no change in the responder rates beyond 6 weeks.
Familial Adenomatous Polyposis (FAP)
CELEBREX was evaluated to reduce the number of adenomatous colorectal polyps. A randomized double-blind placebo-controlled study was conducted in patients with FAP. The study population included 58 patients with a prior subtotal or total colectomy and 25 patients with an intact colon. Thirteen patients had the attenuated FAP phenotype.
One area in the rectum and up to four areas in the colon were identified at baseline for specific follow-up, and polyps were counted at baseline and following six months of treatment. The mean reduction in the number of colorectal polyps was 28% for CELEBREX 400 mg BID, 12% for CELEBREX 100 mg BID and 5% for placebo. The reduction in polyps observed with CELEBREX 400 mg BID was statistically superior to placebo at the six-month timepoint (p=0.003). (See Figure 1.)
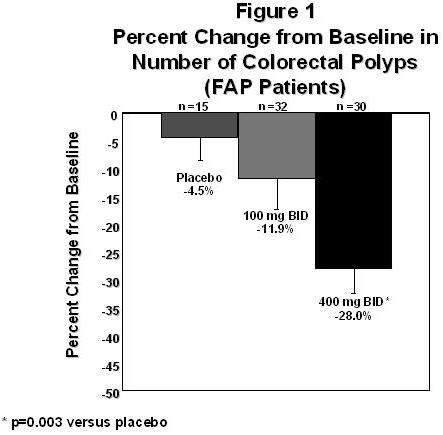
Special Studies
Celecoxib Long-Term Arthritis Safety Study (CLASS)
The Celecoxib Long-Term Arthritis Safety Study (CLASS) was a prospective long-term safety outcome study conducted postmarketing in approximately 5,800 OA patients and 2,200 RA patients. Patients received CELEBREX 400 mg BID (4-fold and 2-fold the recommended OA and RA doses, respectively, and the approved dose for FAP), ibuprofen 800 mg TID or diclofenac 75 mg BID (common therapeutic doses). Median exposures for CELEBREX (n = 3,987) and diclofenac (n = 1,996) were 9 months while ibuprofen (n = 1,985) was 6 months. The primary endpoint of this outcome study was the incidence of complicated ulcers (gastrointestinal bleeding, perforation or obstruction). Patients were allowed to take concomitant low-dose (≤ 325 mg/day) aspirin (ASA) for cardiovascular prophylaxis (ASA subgroups: CELEBREX, n = 882; diclofenac, n = 445; ibuprofen, n = 412). Differences in the incidence of complicated ulcers between CELEBREX and the combined group of ibuprofen and diclofenac were not statistically significant.
Those patients on CELEBREX and concomitant low-dose ASA (N=882) experienced 4-fold higher rates of complicated ulcers compared to those not on ASA (N=3105). The Kaplan Meier rate for complicated ulcers at 9 months was 1.12% versus 0.32% for those on low dose ASA and those not on ASA, respectively (see WARNINGS – Gastrointestinal (GI) Effects – Risk of GI Ulceration, Bleeding and Perforation).
The estimated cumulative rates at 9 months of complicated and symptomatic ulcers for patients treated with CELEBREX 400 mg BID are described in Table 2. Table 2 also displays results for patients less than or greater than 65 years of age. The difference in rates between CELEBREX alone and CELEBREX with ASA groups may be due to the higher risk for GI events in ASA users.
| Complicated and Symptomatic Ulcer Rates | |
|---|---|
| All Patients | |
| Celebrex alone (n=3105) | 0.78 |
| Celebrex with ASA (n=882) | 2.19 |
| Patients <65 Years | |
| Celebrex alone (n=2025) | 0.47 |
| Celebrex with ASA (n=403) | 1.26 |
| Patients ≥65 Years | |
| Celebrex alone (n=1080) | 1.40 |
| Celebrex with ASA (n=479) | 3.06 |
In a small number of patients with a history of ulcer disease, the complicated and symptomatic ulcer rates in patients taking CELEBREX alone or CELEBREX with ASA were, respectively, 2.56% (n=243) and 6.85% (n=91) at 48 weeks. These results are to be expected in patients with a prior history of ulcer disease (see WARNINGS – Gastrointestinal (GI) Effects – Risk of GI Ulceration, Bleeding, and Perforation and ADVERSE REACTIONS – Safety Data from CLASS Study – Hematological Events).
Cardiovascular safety outcomes were also evaluated in the CLASS trial. Kaplan-Meier cumulative rates for investigator-reported serious cardiovascular thromboembolic adverse events (including MI, pulmonary embolism, deep venous thrombosis, unstable angina, transient ischemic attacks, and ischemic cerebrovascular accidents) demonstrated no differences between the CELEBREX, diclofenac, or ibuprofen treatment groups. The cumulative rates in all patients at nine months for CELEBREX, diclofenac, and ibuprofen were 1.2%, 1.4%, and 1.1%, respectively. The cumulative rates in non-ASA users at nine months in each of the three treatment groups were less than 1%. The cumulative rates for myocardial infarction in non-ASA users at nine months in each of the three treatment groups were less than 0.2%. There was no placebo group in the CLASS trial, which limits the ability to determine whether the three drugs tested had no increased risk of CV events or if they all increased the risk to a similar degree.
Adenomatous Polyp Prevention Studies
Cardiovascular safety was evaluated in two randomized, double-blind, placebo-controlled, three-year studies involving patients with Sporadic Adenomatous Polyps treated with CELEBREX. The first of these studies was the APC (Prevention of Sporadic Colorectal Adenomas with Celecoxib) study, which compared CELEBREX 400 mg twice daily (N=671) and CELEBREX 200 mg twice daily (N=685) to placebo (N=679). Preliminary safety information from this trial demonstrated a dose-related increase in serious cardiovascular events (mainly myocardial infarction [MI]) at CELEBREX doses of 200 mg and 400 mg twice daily compared to placebo). The cumulative rates of serious cardiovascular thrombotic events began to differ between the CELEBREX treatment groups and placebo after approximately one year of treatment. There were 2.8 to 3.1 years of follow-up in the APC trial except those patients who died earlier. The relative risk (RR) for the composite endpoint of cardiovascular death, MI, or stroke was 3.4 (95% CI 1.4 – 8.5) for the higher dose and 2.5 (95% CI 1.0 – 6.4) for the lower dose of CELEBREX compared to placebo. The absolute risk for the composite endpoint was 3.0% for the higher dose of CELEBREX, 2.2% for the lower dose of CELEBREX, and 0.9% for placebo.
The second long-term study, PreSAP (Prevention of Colorectal Sporadic Adenomatous Polyps) compared CELEBREX 400 mg once daily to placebo. Preliminary safety information from this trial demonstrated no increased cardiovascular risk for the composite endpoint of cardiovascular death, MI or stroke. The reason for the differing results for CV events in the APC and PreSAP trials is not known.
Clinical trials of other COX-2 selective and nonselective NSAIDs of up to three-years duration have shown an increased risk of serious cardiovascular thrombotic events, myocardial infarction, and stroke, which can be fatal. As a result, all NSAIDs are considered potentially associated with this risk.
Endoscopic Studies
The correlation between findings of short-term endoscopic studies with CELEBREX and the relative incidence of clinically significant serious upper GI events with long-term use has not been established.
A randomized, double-blind study in 430 RA patients was conducted in which an endoscopic examination was performed at 6 months. The incidence of endoscopic ulcers in patients taking CELEBREX 200 mg twice daily was 4% vs. 15% for patients taking diclofenac SR 75 mg twice daily. However, CELEBREX was not statistically different than diclofenac for clinically relevant GI outcomes in the CLASS trial (see Special Studies - CLASS).
The incidence of endoscopic ulcers was studied in two 12-week, placebo-controlled studies in 2157 OA and RA patients in whom baseline endoscopies revealed no ulcers. There was no dose relationship for the incidence of gastroduodenal ulcers and the dose of CELEBREX (50 mg to 400 mg twice daily). The incidence for naproxen 500 mg twice daily was 16.2 and 17.6% in the two studies, for placebo was 2.0 and 2.3%, and for all doses of CELEBREX the incidence ranged between 2.7%–5.9%. There have been no large, clinical outcome studies to compare clinically relevant GI outcomes with CELEBREX and naproxen.
In the endoscopic studies, approximately 11% of patients were taking aspirin (≤ 325 mg/day). In the CELEBREX groups, the endoscopic ulcer rate appeared to be higher in aspirin users than in non-users. However, the increased rate of ulcers in these aspirin users was less than the endoscopic ulcer rates observed in the active comparator groups, with or without aspirin.
Serious clinically significant upper GI bleeding has been observed in patients receiving CELEBREX in controlled and open-labeled trials (see Special Studies - CLASS and WARNINGS – Gastrointestinal (GI) Effects – Risk of GI Ulceration, Bleeding and Perforation).
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of CELEBREX and other treatment options before deciding to use CELEBREX. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS).
CELEBREX is indicated:
- 1)
- For relief of the signs and symptoms of osteoarthritis.
- 2)
- For relief of the signs and symptoms of rheumatoid arthritis in adults.
- 3)
- For relief of the signs and symptoms of juvenile rheumatoid arthritis in patients 2 years and older (see CLINICAL STUDIES and ADVERSE REACTIONS - Adverse Events from JRA Study).
- 4)
- For the relief of signs and symptoms of ankylosing spondylitis.
- 5)
- For the management of acute pain in adults (see CLINICAL STUDIES).
- 6)
- For the treatment of primary dysmenorrhea.
- 7)
- To reduce the number of adenomatous colorectal polyps in familial adenomatous polyposis (FAP), as an adjunct to usual care (e.g., endoscopic surveillance, surgery). It is not known whether there is a clinical benefit from a reduction in the number of colorectal polyps in FAP patients. It is also not known whether the effects of CELEBREX treatment will persist after CELEBREX is discontinued. The efficacy and safety of CELEBREX treatment in patients with FAP beyond six months have not been studied (see CLINICAL STUDIES, WARNINGS and PRECAUTIONS sections).
CONTRAINDICATIONS
CELEBREX is contraindicated in patients with known hypersensitivity to celecoxib.
CELEBREX should not be given to patients who have demonstrated allergic-type reactions to sulfonamides.
CELEBREX should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see WARNINGS — Anaphylactoid Reactions, and PRECAUTIONS — Preexisting Asthma).
CELEBREX is contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery (see WARNINGS)
WARNINGS
Cardiovascular Effects
Cardiovascular Thrombotic Events
Chronic use of CELEBREX may cause an increased risk of serious adverse cardiovascular thrombotic events, myocardial infarction, and stroke, which can be fatal. In the APC trial, the relative risk for the composite endpoint of cardiovascular death, MI, or stroke was 3.4 (95% CI 1.4 – 8.5) for CELEBREX 400 mg twice daily and 2.5 (95% CI 1.0 – 6.4) for the CELEBREX 200 mg twice daily compared to placebo (see Special Studies – Adenomatous Polyp Studies).
All NSAIDs, both COX-2 selective and nonselective, may have a similar risk. Patients with known CV disease or risk factors for CV disease may be at greater risk. To minimize the potential risk for an adverse CV event in patients treated with CELEBREX, the lowest effective dose should be used for the shortest duration possible. Physicians and patients should remain alert for the development of such events, even in the absence of previous CV symptoms. Patients should be informed about the signs and/or symptoms of serious CV toxicity and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and CELEBREX does increase the risk of serious GI events (see GI WARNINGS - Risk of GI Ulceration, Bleeding, and Perforation).
Two large, controlled, clinical trials of a different COX-2 selective NSAID for the treatment of pain in the first 10–14 days following CABG surgery found an increased incidence of myocardial infarction and stroke (see CONTRAINDICATIONS).
Hypertension
As with all NSAIDS, CELEBREX can lead to the onset of new hypertension or worsening of pre-existing hypertension, either of which may contribute to the increased incidence of CV events. Patients taking thiazides or loop diuretics may have impaired response to these therapies when taking NSAIDs. NSAIDs, including CELEBREX, should be used with caution in patients with hypertension. Blood pressure should be monitored closely during the initiation of therapy with CELEBREX and throughout the course of therapy. The rates of hypertension from the CLASS trial in the CELEBREX, ibuprofen and diclofenac treated patients were 2.4%, 4.2% and 2.5%, respectively (see Special Studies - CLASS).
Congestive Heart Failure and Edema
Fluid retention and edema have been observed in some patients taking NSAIDs, including CELEBREX (see ADVERSE REACTIONS). In the CLASS study (see Special Studies – CLASS), the Kaplan-Meier cumulative rates at 9 months of peripheral edema in patients on CELEBREX 400 mg twice daily (4-fold and 2-fold the recommended OA and RA doses, respectively, and the approved dose for FAP), ibuprofen 800 mg three times daily and diclofenac 75 mg twice daily were 4.5%, 6.9% and 4.7%, respectively. CELEBREX should be used with caution in patients with fluid retention or heart failure.
Gastrointestinal (GI) Effects — Risk of GI Ulceration, Bleeding, and Perforation
NSAIDs, including CELEBREX, can cause serious gastrointestinal events including bleeding, ulceration, and perforation of the stomach, small intestine or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs. Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Complicated and symptomatic ulcer rates were 0.78% at nine months for all patients in the CLASS trial, and 2.19% for the subgroup on low dose ASA. Patients 65 years of age and older had an incidence of 1.40% at nine months, 3.06% when also taking ASA (see Special Studies - CLASS). With longer duration of use of NSAIDs, there is a trend for increasing the likelihood of developing a serious GI event at some time during the course of therapy. However, even short-term therapy is not without risk.
NSAIDs should be prescribed with extreme caution in patients with a prior history of ulcer disease or gastrointestinal bleeding. Patients with a prior history of peptic ulcer disease and/or gastrointestinal bleeding who use NSAIDs have a greater than 10-fold increased risk for developing a GI bleed compared to patients with neither of these risk factors. Other factors that increase the risk of GI bleeding in patients treated with NSAIDs include concomitant use of oral corticosteroids or anticoagulants, longer duration of NSAID therapy, smoking, use of alcohol, older age, and poor general health status. Most spontaneous reports of fatal GI events are in elderly or debilitated patients and therefore special care should be taken in treating this population.
To minimize the potential risk for an adverse GI event, the lowest effective dose should be used for the shortest possible duration. Physicians and patients should remain alert for signs and symptoms of GI ulceration and bleeding during CELEBREX therapy and promptly initiate additional evaluation and treatment if a serious GI adverse event is suspected. For high-risk patients, alternate therapies that do not involve NSAIDs should be considered.
Renal Effects
Long-term administration of NSAIDs has resulted in renal papillary necrosis and other renal injury. Renal toxicity has also been seen in patients in whom renal prostaglandins have a compensatory role in the maintenance of renal perfusion. In these patients, administration of an NSAID may cause a dose-dependent reduction in prostaglandin formation and, secondarily, in renal blood flow, which may precipitate overt renal decompensation. Patients at greatest risk of this reaction are those with impaired renal function, heart failure, liver dysfunction, those taking diuretics and ACE inhibitors, angiotensin II receptor antagonists, and the elderly. Discontinuation of NSAID therapy is usually followed by recovery to the pretreatment state. Clinical trials with CELEBREX have shown renal effects similar to those observed with comparator NSAIDs.
Advanced Renal Disease
No information is available from controlled clinical studies regarding the use of CELEBREX in patients with advanced renal disease. Therefore, treatment with CELEBREX is not recommended in these patients with advanced renal disease. If CELEBREX therapy must be initiated, close monitoring of the patient's renal function is advisable.
Anaphylactoid Reactions
As with NSAIDs in general, anaphylactoid reactions have occurred in patients without known prior exposure to CELEBREX. In post-marketing experience, rare cases of anaphylactic reactions and angioedema have been reported in patients receiving CELEBREX. CELEBREX should not be given to patients with the aspirin triad. This symptom complex typically occurs in asthmatic patients who experience rhinitis with or without nasal polyps, or who exhibit severe, potentially fatal bronchospasm after taking aspirin or other NSAIDs (see CONTRAINDICATIONS and PRECAUTIONS — Preexisting Asthma). Emergency help should be sought in cases where an anaphylactoid reaction occurs.
Skin Reactions
CELEBREX is a sulfonamide and can cause serious skin adverse events such as exfoliative dermatitis, Stevens Johnson syndrome (SJS), and toxic epidermal necrolysis (TENS), which can be fatal. These serious events can occur without warning and in patients without prior known sulfa allergy. Patients should be informed about the signs and symptoms of serious skin manifestations and use of the drug should be discontinued at the first appearance of skin rash or any other sign of hypersensitivity.
Pregnancy
In late pregnancy CELEBREX should be avoided because it may cause premature closure of the ductus arteriosus (see PRECAUTIONS – Pregnancy).
Familial Adenomatous Polyposis (FAP)
Treatment with CELEBREX in FAP has not been shown to reduce the risk of gastrointestinal cancer or the need for prophylactic colectomy or other FAP-related surgeries. Therefore, the usual care of FAP patients should not be altered because of the concurrent administration of CELEBREX. In particular, the frequency of routine endoscopic surveillance should not be decreased and prophylactic colectomy or other FAP-related surgeries should not be delayed.
PRECAUTIONS
General
CELEBREX cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to exacerbation of corticosteroid-responsive illness. Patients on prolonged corticosteroid therapy should have their therapy tapered slowly if a decision is made to discontinue corticosteroids.
The concomitant use of CELEBREX with any dose of a non-aspirin NSAID should be avoided.
The pharmacological activity of CELEBREX in reducing inflammation, and possibly fever, may diminish the utility of these diagnostic signs in detecting infectious complications of presumed noninfectious, painful conditions.
Hepatic Effects
Borderline elevations of one or more liver associated enzymes may occur in up to 15% of patients taking NSAIDs, and notable elevations of ALT or AST (approximately 3 or more times the upper limit of normal) have been reported in approximately 1% of patients in clinical trials with NSAIDs. These laboratory abnormalities may progress, may remain unchanged, or may be transient with continuing therapy. Rare cases of severe hepatic reactions, including jaundice and fatal fulminant hepatitis, liver necrosis and hepatic failure (some with fatal outcome) have been reported with NSAIDs, including CELEBREX (see ADVERSE REACTIONS – post-marketing experience). In controlled clinical trials of CELEBREX, the incidence of borderline elevations (greater than or equal to 1.2 times and less than 3 times the upper limit of normal) of liver associated enzymes was 6% for CELEBREX and 5% for placebo, and approximately 0.2% of patients taking CELEBREX and 0.3% of patients taking placebo had notable elevations of ALT and AST.
A patient with symptoms and/or signs suggesting liver dysfunction, or in whom an abnormal liver test has occurred, should be monitored carefully for evidence of the development of a more severe hepatic reaction while on therapy with CELEBREX. If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), CELEBREX should be discontinued.
Hematological Effects
Anemia is sometimes seen in patients receiving CELEBREX. In controlled clinical trials the incidence of anemia was 0.6% with CELEBREX and 0.4% with placebo. Patients on long-term treatment with CELEBREX should have their hemoglobin or hematocrit checked if they exhibit any signs or symptoms of anemia or blood loss. CELEBREX does not generally affect platelet counts, prothrombin time (PT), or partial thromboplastin time (PTT), and does not inhibit platelet aggregation at indicated dosages (see CLINICAL PHARMACOLOGY—Platelets).
Systemic Onset Juvenile Rheumatoid Arthritis
CELEBREX should be used only with caution in pediatric patients with systemic onset JRA due to the risk for serious adverse reactions including disseminated intravascular coagulation.
Preexisting Asthma
Patients with asthma may have aspirin-sensitive asthma. The use of aspirin in patients with aspirin-sensitive asthma has been associated with severe bronchospasm, which can be fatal. Since cross reactivity, including bronchospasm, between aspirin and other nonsteroidal anti-inflammatory drugs has been reported in such aspirin-sensitive patients, CELEBREX should not be administered to patients with this form of aspirin sensitivity and should be used with caution in patients with preexisting asthma.
Information for Patients
Patients should be informed of the following information before initiating therapy with CELEBREX and periodically during the course of ongoing therapy. Patients should also be encouraged to read the NSAID Medication Guide that accompanies each prescription dispensed.
- CELEBREX, like other NSAIDs, may cause serious CV side effects such as MI or stroke, which may result in hospitalization and even death. Although serious CV events can occur without warning symptoms, patients should be alert for the signs and symptoms of chest pain, shortness of breath, weakness, slurring of speech, and should ask for medical advice if they observe any of these signs or symptoms. Patients should be apprised of the importance of this follow-up (see WARNINGS - Cardiovascular Effects).
- CELEBREX, like other NSAIDs, can cause gastrointestinal discomfort and, rarely, more serious side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when they observe any signs or symptoms that are indicative of these disorders, including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow-up (see WARNINGS — Gastrointestinal (GI) Effects – Risk of Gastrointestinal Ulceration, Bleeding, and Perforation).
- Patients should be advised to stop the drug immediately if they develop any type of rash and contact their physicians as soon as possible. CELEBREX is a sulfonamide and can cause serious skin side effects such as exfoliative dermatitis, SJS, and TENS, which may result in hospitalizations and even death. These reactions can occur with all NSAIDs, even non-sulfonamides. Although serious skin reactions may occur without warning, patients should be alert for the signs and symptoms of skin rash and blisters, fever, or other signs of hypersensitivity such as itching, and should ask for medical advice when observing any indicative signs or symptoms. Patients with prior history of sulfa allergy should not take CELEBREX.
- Patients should promptly report signs or symptoms of unexplained weight gain or edema to their physicians.
- Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). Patients should be instructed that they should stop therapy and seek immediate medical therapy if these signs and symptoms occur.
- Patients should be informed of the signs and symptoms of an anaphylactoid reaction (e.g. difficulty breathing, swelling of the face or throat). Patients should be instructed to seek immediate emergency assistance if they develop any of these signs and symptoms (see WARNINGS – Anaphylactoid Reactions).
- Patients should be informed that in late pregnancy CELEBREX should be avoided because it may cause premature closure of the ductus arteriosus.
- Patients with familial adenomatous polyposis (FAP) should be informed that CELEBREX has not been shown to reduce colorectal, duodenal or other FAP-related cancers, or the need for endoscopic surveillance, prophylactic or other FAP-related surgery. Therefore, all patients with FAP should be instructed to continue their usual care while receiving CELEBREX.
Laboratory Tests
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. Patients on long-term treatment with NSAIDs, should have a CBC and a chemistry profile checked periodically. If abnormal liver tests or renal tests persist or worsen, CELEBREX should be discontinued.
In controlled clinical trials, elevated BUN occurred more frequently in patients receiving CELEBREX compared with patients on placebo. This laboratory abnormality was also seen in patients who received comparator NSAIDs in these studies. The clinical significance of this abnormality has not been established.
Drug Interactions
General
Celecoxib metabolism is predominantly mediated via cytochrome P450 2C9 in the liver. Co-administration of celecoxib with drugs that are known to inhibit 2C9 should be done with caution.
In vitro studies indicate that celecoxib, although not a substrate, is an inhibitor of cytochrome P450 2D6. Therefore, there is a potential for an in vivo drug interaction with drugs that are metabolized by P450 2D6.
ACE-inhibitors and Angiotensin II Antagonists
Reports suggest that NSAIDs may diminish the antihypertensive effect of Angiotensin Converting Enzyme (ACE) inhibitors and angiotensin II antagonists. This interaction should be given consideration in patients taking CELEBREX concomitantly with ACE-inhibitors and angiotensin II antagonists.
Aspirin
CELEBREX can be used with low-dose aspirin. However, concomitant administration of aspirin with CELEBREX increases the rate of GI ulceration or other complications, compared to use of CELEBREX alone (see CLINICAL STUDIES — Special Studies — CLASS, WARNINGS – Gastrointestinal (GI) Effects – Risk of GI Ulceration, Bleeding, and Perforation, and WARNINGS – Cardiovascular Effects).
Because of its lack of platelet effects, CELEBREX is not a substitute for aspirin for cardiovascular prophylaxis.
Fluconazole
Concomitant administration of fluconazole at 200 mg QD resulted in a two-fold increase in celecoxib plasma concentration. This increase is due to the inhibition of celecoxib metabolism via P450 2C9 by fluconazole (see Pharmacokinetics — Metabolism). CELEBREX should be introduced at the lowest recommended dose in patients receiving fluconazole.
Furosemide
Clinical studies, as well as post marketing observations, have shown that NSAIDs can reduce the natriuretic effect of furosemide and thiazides in some patients. This response has been attributed to inhibition of renal prostaglandin synthesis.
Lithium
In a study conducted in healthy subjects, mean steady-state lithium plasma levels increased approximately 17% in subjects receiving lithium 450 mg BID with CELEBREX 200 mg BID as compared to subjects receiving lithium alone. Patients on lithium treatment should be closely monitored when CELEBREX is introduced or withdrawn.
Methotrexate
In an interaction study of rheumatoid arthritis patients taking methotrexate, CELEBREX did not have a significant effect on the pharmacokinetics of methotrexate.
Warfarin
Anticoagulant activity should be monitored, particularly in the first few days, after initiating or changing CELEBREX therapy in patients receiving warfarin or similar agents, since these patients are at an increased risk of bleeding complications. The effect of celecoxib on the anticoagulant effect of warfarin was studied in a group of healthy subjects receiving daily doses of 2–5 mg of warfarin. In these subjects, celecoxib did not alter the anticoagulant effect of warfarin as determined by prothrombin time. However, in post-marketing experience, serious bleeding events, some of which were fatal, have been reported, predominantly in the elderly, in association with increases in prothrombin time in patients receiving CELEBREX concurrently with warfarin.
Animal Toxicology
An increase in the incidence of background findings of spermatocele with or without secondary changes such as epididymal hypospermia as well as minimal to slight dilation of the seminiferous tubules was seen in the juvenile rat. These reproductive findings while apparently treatment-related did not increase in incidence or severity with dose and may indicate an exacerbation of a spontaneous condition. Similar reproductive findings were not observed in studies of juvenile or adult dogs or in adult rats treated with celecoxib. The clinical significance of this observation is unknown.
Carcinogenesis, mutagenesis, impairment of fertility
Celecoxib was not carcinogenic in rats given oral doses up to 200 mg/kg for males and 10 mg/kg for females (approximately 2- to 4-fold the human exposure as measured by the AUC0–24 at 200 mg BID) or in mice given oral doses up to 25 mg/kg for males and 50 mg/kg for females (approximately equal to human exposure as measured by the AUC0–24 at 200 mg BID) for two years.
Celecoxib was not mutagenic in an Ames test and a mutation assay in Chinese hamster ovary (CHO) cells, nor clastogenic in a chromosome aberration assay in CHO cells and an in vivo micronucleus test in rat bone marrow.
Celecoxib did not impair male and female fertility in rats at oral doses up to 600 mg/kg/day (approximately 11-fold human exposure at 200 mg BID based on the AUC0–24).
Pregnancy
Teratogenic effects
Pregnancy Category C
Celecoxib at oral doses ≥150 mg/kg/day (approximately 2-fold human exposure at 200 mg BID as measured by AUC0–24), caused an increased incidence of ventricular septal defects, a rare event, and fetal alterations, such as ribs fused, sternebrae fused and sternebrae misshapen when rabbits were treated throughout organogenesis. A dose-dependent increase in diaphragmatic hernias was observed when rats were given celecoxib at oral doses ≥30 mg/kg/day (approximately 6-fold human exposure based on the AUC0–24 at 200 mg BID) throughout organogenesis. There are no studies in pregnant women. CELEBREX should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic effects
Celecoxib produced pre-implantation and post-implantation losses and reduced embryo/fetal survival in rats at oral dosages ≥50 mg/kg/day (approximately 6-fold human exposure based on the AUC0–24 at 200 mg BID). These changes are expected with inhibition of prostaglandin synthesis and are not the result of permanent alteration of female reproductive function, nor are they expected at clinical exposures. No studies have been conducted to evaluate the effect of celecoxib on the closure of the ductus arteriosus in humans. Therefore, use of CELEBREX during the third trimester of pregnancy should be avoided.
Labor and delivery
Celecoxib produced no evidence of delayed labor or parturition at oral doses up to 100 mg/kg in rats (approximately 7-fold human exposure as measured by the AUC0–24 at 200 mg BID). The effects of CELEBREX on labor and delivery in pregnant women are unknown.
Nursing mothers
Celecoxib is excreted in the milk of lactating rats at concentrations similar to those in plasma. Limited data from one subject indicate that celecoxib is also excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from CELEBREX, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
CELEBREX is approved for relief of the signs and symptoms of Juvenile Rheumatoid Arthritis in patients 2 years and older. Safety and efficacy have not been studied beyond six months in children. The long-term cardiovascular toxicity in children exposed to CELEBREX has not been evaluated and it is unknown if long-term risks may be similar to that seen in adults exposed to CELEBREX or other COX-2 selective and non-selective NSAIDS. (see Boxed Warning, WARNINGS, and CLINICAL STUDIES )
The use of celecoxib in patients 2 years to 17 years of age with pauciarticular, polyarticular course JRA or in patients with systemic onset JRA was studied in a 12-week, double-blind, active controlled, pharmacokinetic, safety and efficacy study, with a 12-week open-label extension. Celecoxib has not been studied in patients under the age of 2 years, in patients with body weight less than 10 kg (22 lbs), and in patients with active systemic features. Patients with systemic onset JRA (without active systemic features) appear to be at risk for the development of abnormal coagulation laboratory tests. In some patients with systemic onset JRA, both celecoxib and naproxen were associated with mild prolongation of activated partial thromboplastin time (APTT) but not prothrombin time (PT). NSAIDs including celecoxib should be used only with caution in patients with systemic onset JRA, due to the risk of disseminated intravascular coagulation. Patients with systemic onset JRA should be monitored for the development of abnormal coagulation tests. (see CLINICAL PHARMACOLOGY – Pediatric, CLINICAL STUDIES – JRA, PRECAUTIONS – Systemic Onset JRA, PRECAUTIONS - Animal Toxicology, ADVERSE REACTIONS - Adverse events from JRA studies, and DOSAGE and ADMINISTRATION - JRA).
Geriatric Use
Of the total number of patients who received CELEBREX in clinical trials, more than 3,300 were 65–74 years of age, while approximately 1,300 additional patients were 75 years and over. No substantial differences in effectiveness were observed between these subjects and younger subjects. In clinical studies comparing renal function as measured by the GFR, BUN and creatinine, and platelet function as measured by bleeding time and platelet aggregation, the results were not different between elderly and young volunteers. However, as with other NSAIDs, including those that selectively inhibit COX-2, there have been more spontaneous post-marketing reports of fatal GI events and acute renal failure in the elderly than in younger patients (see WARNINGS – Gastrointestinal (GI) Effects – Risk of GI Ulceration, Bleeding, and Perforation).
ADVERSE REACTIONS
Of the CELEBREX treated patients in the premarketing controlled clinical trials, approximately 4,250 were patients with OA, approximately 2,100 were patients with RA, and approximately 1,050 were patients with post-surgical pain. More than 8,500 patients have received a total daily dose of CELEBREX of 200 mg (100 mg BID or 200 mg QD) or more, including more than 400 treated at 800 mg (400 mg BID). Approximately 3,900 patients have received CELEBREX at these doses for 6 months or more; approximately 2,300 of these have received it for 1 year or more and 124 of these have received it for 2 years or more.
Adverse events from CELEBREX premarketing controlled arthritis trials
Table 3 lists all adverse events, regardless of causality, occurring in ≥2% of patients receiving CELEBREX from 12 controlled studies conducted in patients with OA or RA that included a placebo and/or a positive control group. Since these 12 trials were of different durations, and patients in the trials may not have been exposed for the same duration of time, these percentages do not capture cumulative rates of occurrence.
| Adverse Events Occurring in ≥2% of CELEBREX Patients From CELEBREX Premarketing Controlled Arthritis Trials | |||||
|---|---|---|---|---|---|
| Celebrex (100–200 mg BID or 200 mg QD) | Placebo | Naproxen 500 mg BID | Diclofenac 75 mg BID | Ibuprofen 800 mg TID |
|
| (n=4146) | (n=1864) | (n=1366) | (n=387) | (n=345) | |
| Gastrointestinal | |||||
| Abdominal pain | 4.1% | 2.8% | 7.7% | 9.0% | 9.0% |
| Diarrhea | 5.6% | 3.8% | 5.3% | 9.3% | 5.8% |
| Dyspepsia | 8.8% | 6.2% | 12.2% | 10.9% | 12.8% |
| Flatulence | 2.2% | 1.0% | 3.6% | 4.1% | 3.5% |
| Nausea | 3.5% | 4.2% | 6.0% | 3.4% | 6.7% |
| Body as a whole | |||||
| Back pain | 2.8% | 3.6% | 2.2% | 2.6% | 0.9% |
| Peripheral edema | 2.1% | 1.1% | 2.1% | 1.0% | 3.5% |
| Injury-accidental | 2.9% | 2.3% | 3.0% | 2.6% | 3.2% |
| Central and peripheral nervous system | |||||
| Dizziness | 2.0% | 1.7% | 2.6% | 1.3% | 2.3% |
| Headache | 15.8% | 20.2% | 14.5% | 15.5% | 15.4% |
| Psychiatric | |||||
| Insomnia | 2.3% | 2.3% | 2.9% | 1.3% | 1.4% |
| Respiratory | |||||
| Pharyngitis | 2.3% | 1.1% | 1.7% | 1.6% | 2.6% |
| Rhinitis | 2.0% | 1.3% | 2.4% | 2.3% | 0.6% |
| Sinusitis | 5.0% | 4.3% | 4.0% | 5.4% | 5.8% |
| Upper respiratory tract infection | 8.1% | 6.7% | 9.9% | 9.8% | 9.9% |
| Skin | |||||
| Rash | 2.2% | 2.1% | 2.1% | 1.3% | 1.2% |
In placebo- or active-controlled clinical trials, the discontinuation rate due to adverse events was 7.1% for patients receiving CELEBREX and 6.1% for patients receiving placebo. Among the most common reasons for discontinuation due to adverse events in the CELEBREX treatment groups were dyspepsia and abdominal pain (cited as reasons for discontinuation in 0.8% and 0.7% of CELEBREX patients, respectively). Among patients receiving placebo, 0.6% discontinued due to dyspepsia and 0.6% withdrew due to abdominal pain.
The following adverse events occurred in 0.1 – 1.9% of patients regardless of causality.
| CELEBREX | ||
|---|---|---|
| (100 – 200 mg BID or 200 mg QD) | ||
| Gastrointestinal: | Constipation, diverticulitis, dysphagia, eructation, esophagitis, gastritis, gastroenteritis, gastroesophageal reflux, hemorrhoids, hiatal hernia, melena, dry mouth, stomatitis, tenesmus, tooth disorder, vomiting | |
| Cardiovascular: | Aggravated hypertension, angina pectoris, coronary artery disorder, myocardial infarction | |
| General: | Allergy aggravated, allergic reaction, asthenia, chest pain, cyst NOS, edema generalized, face edema, fatigue, fever, hot flushes, influenza-like symptoms, pain, peripheral pain | |
| Resistance mechanism disorders: | Herpes simplex, herpes zoster, infection bacterial, infection fungal, infection soft tissue, infection viral, moniliasis, moniliasis genital, otitis media | |
| Central, peripheral nervous system: | Leg cramps, hypertonia, hypoesthesia, migraine, neuralgia, neuropathy, paresthesia, vertigo | |
| Female reproductive: | Breast fibroadenosis, breast neoplasm, breast pain, dysmenorrhea, menstrual disorder, vaginal hemorrhage, vaginitis | |
| Male reproductive: | Prostatic disorder | |
| Hearing and vestibular: | Deafness, ear abnormality, earache, tinnitus | |
| Heart rate and rhythm: | Palpitation, tachycardia | |
| Liver and biliary system: | Hepatic function abnormal, SGOT increased, SGPT increased | |
| Metabolic and nutritional: | BUN increased, CPK increased, diabetes mellitus, hypercholesterolemia, hyperglycemia, hypokalemia, NPN increase, creatinine increased, alkaline phosphatase increased, weight increase | |
| Musculoskeletal: | Arthralgia, arthrosis, bone disorder, fracture accidental, myalgia, neck stiffness, synovitis, tendinitis | |
| Platelets (bleeding or clotting): | Ecchymosis, epistaxis, thrombocythemia | |
| Psychiatric: | Anorexia, anxiety, appetite increased, depression, nervousness, somnolence | |
| Hemic: | Anemia | |
| Respiratory: | Bronchitis, bronchospasm, bronchospasm aggravated, coughing, dyspnea, laryngitis, pneumonia | |
| Skin and appendages: | Alopecia, dermatitis, nail disorder, photosensitivity reaction, pruritus, rash erythematous, rash maculopapular, skin disorder, skin dry, sweating increased, urticaria | |
| Application site disorders: | Cellulitis, dermatitis contact, injection site reaction, skin nodule | |
| Special senses: | Taste perversion | |
| Urinary system: | Albuminuria, cystitis, dysuria, hematuria, micturition frequency, renal calculus, urinary incontinence, urinary tract infection | |
| Vision: | Blurred vision, cataract, conjunctivitis, eye pain, glaucoma | |
Other serious adverse reactions which occur rarely (estimated <0.1%), regardless of causality
The following serious adverse events have occurred rarely in patients taking CELEBREX. Cases reported only in the post-marketing experience are indicated in italics.
| Cardiovascular: | Syncope, congestive heart failure, ventricular fibrillation, pulmonary embolism, cerebrovascular accident, peripheral gangrene, thrombophlebitis, vasculitis, deep venous thrombosis |
| Gastrointestinal: | Intestinal obstruction, intestinal perforation, gastrointestinal bleeding, colitis with bleeding, esophageal perforation, pancreatitis, ileus |
| Liver and biliary system: | Cholelithiasis, hepatitis, jaundice, liver failure |
| Hemic and lymphatic: | Thrombocytopenia, agranulocytosis, aplastic anemia, pancytopenia, leukopenia |
| Metabolic: | Hypoglycemia, hyponatremia |
| Nervous system: | Ataxia, suicide, aseptic meningitis, ageusia, anosmia, fatal intracranial hemorrhage (see PRECAUTIONS – Drug Interactions – Warfarin) |
| Renal: | Acute renal failure, interstitial nephritis |
| Skin: | Erythema multiforme, exfoliative dermatitis, Stevens-Johnson syndrome, toxic epidermal necrolysis |
| General: | Sepsis, sudden death, anaphylactoid reaction, angioedema |
Adverse reactions from long-term, placebo-controlled polyp prevention studies
Exposure to CELEBREX in the APC and PreSAP trials was 400 to 800 mg daily for up to 3 years; see Special Studies - Adenomatous Polyp Prevention Studies.
Some adverse reactions occurred in higher percentages of patients than in the arthritis pre-marketing trials (treatment durations up to 12 weeks; see Adverse events from CELEBREX premarketing controlled arthritis trials). The adverse reactions for which these differences in patients treated with CELEBREX were greater as compared to the arthritis pre-marketing trials were as follows:
| CELEBREX (400 – 800 mg daily dose) | Placebo | |
|---|---|---|
| (n=2285) | (n=1303) | |
| Diarrhea | 10.5% | 7.0% |
| Gastroesophageal reflux disease | 4.7% | 3.1% |
| Nausea | 6.8% | 5.3% |
| Vomiting | 3.2% | 2.1% |
| Dyspnea | 2.8% | 1.6% |
| Hypertension | 12.5% | 9.8% |
The following additional adverse reactions occurred in ≥0.1% and <1% of patients taking CELEBREX, at an incidence greater than placebo in the long-term polyp prevention studies and were either not reported during the controlled arthritis pre-marketing trials or occurred with greater frequency in the long-term, placebo-controlled polyp prevention studies:
Nervous system disorders: Cerebral infarction
Eye disorders: Vitreous floaters, conjunctival hemorrhage
Ear and labyrinth disorders: Labyrinthitis
Cardiac disorders: Angina unstable, aortic valve incompetence, sinus bradycardia, ventricular hypertrophy
Vascular disorders: Deep vein thrombosis
Reproductive system and breast disorders: Ovarian cyst
Investigations: Blood potassium increased, blood sodium increased, blood testosterone decreased
Injury, poisoning and procedural complications: Epicondylitis, tendon rupture
Safety Data from CLASS Study
Hematological Events
During this study (see Special Studies – CLASS), the incidence of clinically significant decreases in hemoglobin (>2 g/dL) confirmed by repeat testing was lower in patients on CELEBREX 400 mg BID (4-fold and 2-fold the recommended OA and RA doses, respectively, and the approved dose for FAP) compared to patients on either diclofenac 75 mg BID or ibuprofen 800 mg TID: 0.5%, 1.3% and 1.9%, respectively. The lower incidence of events with CELEBREX was maintained with or without ASA use (see CLINICAL PHARMACOLOGY - Platelets).
Withdrawals/Serious Adverse Events
Kaplan-Meier cumulative rates at 9 months for withdrawals due to adverse events for CELEBREX, diclofenac and ibuprofen were 24%, 29%, and 26%, respectively. Rates for serious adverse events (i.e. those causing hospitalization or felt to be life threatening or otherwise medically significant) regardless of causality were not different across treatment groups, respectively, 8%, 7%, and 8%.
Adverse events from juvenile rheumatoid arthritis study
In a 12-week, double-blind, active-controlled study, 242 JRA patients 2 years to 17 years of age were treated with celecoxib or naproxen; 77 JRA patients were treated with celecoxib 3 mg/kg BID, 82 patients were treated with celecoxib 6 mg/kg BID, and 83 patients were treated with naproxen 7.5 mg/kg BID. The most commonly occurring (≥5%) adverse events in celecoxib treated patients were headache, fever (pyrexia), upper abdominal pain, cough, nasopharyngitis, abdominal pain, nausea, arthralgia, diarrhea and vomiting. The most commonly occurring (≥5%) adverse experiences for naproxen treated patients were headache, nausea, vomiting, fever, upper abdominal pain, diarrhea, cough, abdominal pain, and dizziness (Table 4). Compared with naproxen, celecoxib at doses of 3 and 6 mg/kg BID had no observable deleterious effect on growth and development during the course of the 12-week double-blind study. There was no substantial difference in the number of clinical exacerbations of uveitis or systemic features of JRA among treatment groups.
In a 12-week, open-label extension of the double-blind study described above, 202 JRA patients were treated with celecoxib 6 mg/kg BID. The incidence of adverse events was similar to that observed during the double-blind study; no unexpected adverse events of clinical importance emerged.
| System Organ Class/ Adverse Event Preferred Term | Celecoxib 3 mg/kg BID N=77 | Celecoxib 6 mg/kg BID N=82 | Naproxen 7.5 mg/kg BID N=83 |
|---|---|---|---|
|
|||
| Any Event, % | 64 | 70 | 72 |
| Eye Disorders | 5 | 5 | 5 |
| Gastrointestinal Disorders | 26 | 24 | 36 |
| Abdominal pain NOS | 4 | 7 | 7 |
| Abdominal pain upper | 8 | 6 | 10 |
| Vomiting NOS | 3 | 6 | 11 |
| Diarrhea NOS | 5 | 4 | 8 |
| Nausea | 7 | 4 | 11 |
| General Disorders and Administration Site Conditions | 13 | 11 | 18 |
| Pyrexia | 8 | 9 | 11 |
| Infections and Infestations | 25 | 20 | 27 |
| Nasopharyngitis | 5 | 6 | 5 |
| Injury and Poisoning | 4 | 6 | 5 |
| Investigations* | 3 | 11 | 7 |
| Musculoskeletal, Connective Tissue and Bone Disorders | 8 | 10 | 17 |
| Arthralgia | 3 | 7 | 4 |
| Nervous System Disorders | 17 | 11 | 21 |
| Headache NOS | 13 | 10 | 16 |
| Dizziness (excluding vertigo) | 1 | 1 | 7 |
| Respiratory, Thoracic and Mediastinal Disorders | 8 | 15 | 15 |
| Cough | 7 | 7 | 8 |
| Skin & Subcutaneous Tissue Disorders | 10 | 7 | 18 |
Adverse events from ankylosing spondylitis studies
A total of 378 patients were treated with CELEBREX in placebo- and active- controlled ankylosing spondylitis studies. Doses up to 400 mg QD were studied. The types of adverse events reported in the ankylosing spondylitis studies were similar to those reported in the arthritis studies.
Adverse events from analgesia and dysmenorrhea studies
Approximately 1,700 patients were treated with CELEBREX in analgesia and dysmenorrhea studies. All patients in post-oral surgery pain studies received a single dose of study medication. Doses up to 600 mg/day of CELEBREX were studied in primary dysmenorrhea and post-orthopedic surgery pain studies. The types of adverse events in the analgesia and dysmenorrhea studies were similar to those reported in arthritis studies. The only additional adverse event reported was post-dental extraction alveolar osteitis (dry socket) in the post-oral surgery pain studies.
Adverse events from the controlled trial in familial adenomatous polyposis
The adverse event profile reported for the 83 patients with familial adenomatous polyposis enrolled in the randomized, controlled clinical trial was similar to that reported for patients in the arthritis controlled trials. Intestinal anastomotic ulceration was the only new adverse event reported in the FAP trial, regardless of causality, and was observed in 3 of 58 patients (one at 100 mg BID, and two at 400 mg BID) who had prior intestinal surgery.
OVERDOSAGE
No overdoses of CELEBREX were reported during clinical trials. Doses up to 2400 mg/day for up to 10 days in 12 patients did not result in serious toxicity. Symptoms following acute NSAID overdoses are usually limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which are generally reversible with supportive care. Gastrointestinal bleeding can occur. Hypertension, acute renal failure, respiratory depression and coma may occur, but are rare. Anaphylactoid reactions have been reported with therapeutic ingestion of NSAIDs, and may occur following an overdose.
Patients should be managed by symptomatic and supportive care following an NSAID overdose. There are no specific antidotes. No information is available regarding the removal of celecoxib by hemodialysis, but based on its high degree of plasma protein binding (>97%) dialysis is unlikely to be useful in overdose. Emesis and/or activated charcoal (60 to 100 g in adults, 1 to 2 g/kg in children) and/or osmotic cathartic may be indicated in patients seen within 4 hours of ingestion with symptoms or following a large overdose. Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of CELEBREX and other treatment options before deciding to use CELEBREX. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS).
For osteoarthritis and rheumatoid arthritis, the lowest dose of CELEBREX should be sought for each patient. These doses can be given without regard to timing of meals.
Osteoarthritis
For relief of the signs and symptoms of osteoarthritis the recommended oral dose is 200 mg per day administered as a single dose or as 100 mg twice per day.
Rheumatoid arthritis
For relief of the signs and symptoms of rheumatoid arthritis the recommended oral dose is 100 to 200 mg twice per day.
Juvenile Rheumatoid Arthritis
| Pediatric Patients (2 years and older) | Dose |
|---|---|
| ≥10 kg to ≤25 kg | 50 mg capsule twice daily |
| >25 kg | 100 mg capsule twice daily |
Method of Administration
For patients who have difficulty swallowing capsules, the contents of a CELEBREX capsule can be added to applesauce. The entire capsule contents are carefully emptied onto a level teaspoon of cool or room temperature applesauce and ingested immediately with water. The sprinkled capsule contents on applesauce are stable for up to 6 hours under refrigerated conditions (2–8° C/ 35–45° F).
Ankylosing Spondylitis (AS)
For the management of the signs and symptoms of AS, the recommended dose of CELEBREX is 200 mg daily single (once per day) or divided (twice per day) doses. If no effect is observed after 6 weeks, a trial of 400 mg daily may be worthwhile. If no effect is observed after 6 weeks on 400 mg daily, a response is not likely and consideration should be given to alternate treatment options.
Management of Acute Pain and Treatment of Primary Dysmenorrhea
The recommended dose of CELEBREX is 400 mg initially, followed by an additional 200 mg dose if needed on the first day. On subsequent days, the recommended dose is 200 mg twice daily as needed.
Familial adenomatous polyposis (FAP)
Usual medical care for FAP patients should be continued while on CELEBREX. To reduce the number of adenomatous colorectal polyps in patients with FAP, the recommended oral dose is 400 mg twice per day to be taken with food.
Special Populations
Hepatic insufficiency
The daily recommended dose of CELEBREX capsules in patients with moderate hepatic impairment (Child-Pugh Class B) should be reduced by approximately 50%. The use of CELEBREX in patients with severe hepatic impairment is not recommended (see CLINICAL PHARMACOLOGY – Special Populations).
HOW SUPPLIED
CELEBREX 50-mg capsules are white, with reverse printed white on red band of body and cap with markings of 7767 on the cap and 50 on the body, supplied as:
NDC Number Size
0025-1515-01 bottle of 60
CELEBREX 100-mg capsules are white, reverse printed white on blue band of body and cap with markings of 7767 on the cap and 100 on the body, supplied as:
NDC Number Size
0025-1520-31 bottle of 100
0025-1520-51 bottle of 500
0025-1520-34 carton of 100 unit dose
CELEBREX 200-mg capsules are white, with reverse printed white on gold band with markings of 7767 on the cap and 200 on the body, supplied as:
NDC Number Size
0025-1525-31 bottle of 100
0025-1525-51 bottle of 500
0025-1525-34 carton of 100 unit dose
CELEBREX 400-mg capsules are white, with reverse printed white on green band with markings of 7767 on the cap and 400 on the body, supplied as:
NDC Number Size
0025-1530-02 bottle of 60
0025-1530-01 carton of 100 unit dose
Store at 25°C (77°F); excursions permitted to 15–30°C (59–86°F) [see USP Controlled Room Temperature].
Rx only

LAB-0036-11
January 2008
Medication Guide for Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
(See the end of this Medication Guide for a list of prescription NSAID medicines.)
What is the most important information I should know about medicines called Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)?
NSAID medicines may increase the chance of a heart attack or stroke that can lead to death. This chance increases:
- with longer use of NSAID medicines
- in people who have heart disease
NSAID medicines should never be used right before or after a heart surgery called a "coronary artery bypass graft (CABG)."
NSAID medicines can cause ulcers and bleeding in the stomach and intestines at any time during treatment. Ulcers and bleeding:
- can happen without warning symptoms
- may cause death
The chance of a person getting an ulcer or bleeding increases with:
- taking medicines called "corticosteroids" and "anticoagulants"
- longer use
- smoking
- drinking alcohol
- older age
- having poor health
NSAID medicines should only be used:
- exactly as prescribed
- at the lowest dose possible for your treatment
- for the shortest time needed
What are Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)?
NSAID medicines are used to treat pain and redness, swelling, and heat (inflammation) from medical conditions such as:
- different types of arthritis
- menstrual cramps and other types of short-term pain
Who should not take a Non-Steroidal Anti-Inflammatory Drug (NSAID)?
Do not take an NSAID medicine:
- if you had an asthma attack, hives, or other allergic reaction with aspirin or any other NSAID medicine
- for pain right before or after heart bypass surgery
Tell your healthcare provider:
- about all of your medical conditions.
- about all of the medicines you take. NSAIDs and some other medicines can interact with each other and cause serious side effects. Keep a list of your medicines to show to your healthcare provider and pharmacist.
- if you are pregnant. NSAID medicines should not be used by pregnant women late in their pregnancy.
- if you are breastfeeding. Talk to your doctor.
What are the possible side effects of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)?
| Serious side effects include: | Other side effects include: |
|
|
Get emergency help right away if you have any of the following symptoms:
|
|
Stop your NSAID medicine and call your healthcare provider right away if you have any of the following symptoms:
|
|
These are not all the side effects with NSAID medicines. Talk to your healthcare provider or pharmacist for more information about NSAID medicines.
Other information about Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
- Aspirin is an NSAID medicine but it does not increase the chance of a heart attack. Aspirin can cause bleeding in the brain, stomach, and intestines. Aspirin can also cause ulcers in the stomach and intestines.
- Some of these NSAID medicines are sold in lower doses without a prescription (over –the –counter). Talk to your healthcare provider before using over –the –counter NSAIDs for more than 10 days.
| Generic Name | Tradename |
|---|---|
|
|
| Celecoxib | Celebrex |
| Diclofenac | Cataflam, Voltaren, Arthrotec (combined with misoprostol) |
| Diflunisal | Dolobid |
| Etodolac | Lodine, Lodine XL |
| Fenoprofen | Nalfon, Nalfon 200 |
| Flurbiprofen | Ansaid |
| Ibuprofen | Motrin, Tab-Profen, Vicoprofen* (combined with hydrocodone), Combunox (combined with oxycodone) |
| Indomethacin | Indocin, Indocin SR, Indo-Lemmon, Indomethagan |
| Ketoprofen | Oruvail |
| Ketorolac | Toradol |
| Mefenamic Acid | Ponstel |
| Meloxicam | Mobic |
| Nabumetone | Relafen |
| Naproxen | Naprosyn, Anaprox, Anaprox DS, EC-Naproxyn, Naprelan, Naprapac (copackaged with lansoprazole) |
| Oxaprozin | Daypro |
| Piroxicam | Feldene |
| Sulindac | Clinoril |
| Tolmetin | Tolectin, Tolectin DS, Tolectin 600 |
This Medication Guide has been approved by the U.S. Food and Drug Administration.
| Celebrex (Celecoxib) | |||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| Celebrex (Celecoxib) | |||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| Celebrex (Celecoxib) | |||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| Celebrex (Celecoxib) | |||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
Revised: 01/2008Pfizer Inc