VIRAMUNE
-
nevirapine tablet, extended release
Boehringer Ingelheim Pharmaceuticals Inc.
----------
|
||||||||||||||||||||||
FULL PRESCRIBING INFORMATION
WARNING: LIFE-THREATENING (INCLUDING FATAL) HEPATOTOXICITY and SKIN REACTIONS
HEPATOTOXICITY:
Severe, life-threatening, and in some cases fatal hepatotoxicity, particularly in the first 18 weeks, has been reported in patients treated with nevirapine. In some cases, patients presented with non-specific prodromal signs or symptoms of hepatitis and progressed to hepatic failure. These events are often associated with rash. Female gender and higher CD4+ cell counts at initiation of therapy place patients at increased risk; women with CD4+ cell counts greater than 250 cells/mm3, including pregnant women receiving nevirapine in combination with other antiretrovirals for the treatment of HIV-1 infection, are at the greatest risk. However, hepatotoxicity associated with nevirapine use can occur in both genders, all CD4+ cell counts and at any time during treatment. Hepatic failure has also been reported in patients without HIV taking nevirapine for post-exposure prophylaxis (PEP). Use of nevirapine for occupational and non-occupational PEP is contraindicated [see Contraindications (4.2)]. Patients with signs or symptoms of hepatitis, or with increased transaminases combined with rash or other systemic symptoms, must discontinue nevirapine and seek medical evaluation immediately [see Warnings and Precautions (5.1)].
SKIN REACTIONS:
Severe, life-threatening skin reactions, including fatal cases, have occurred in patients treated with nevirapine. These have included cases of Stevens-Johnson syndrome, toxic epidermal necrolysis, and hypersensitivity reactions characterized by rash, constitutional findings, and organ dysfunction. Patients developing signs or symptoms of severe skin reactions or hypersensitivity reactions must discontinue nevirapine and seek medical evaluation immediately. Transaminase levels should be checked immediately for all patients who develop a rash in the first 18 weeks of treatment. The 14-day lead-in period with immediate-release VIRAMUNE 200 mg daily dosing has been observed to decrease the incidence of rash and must be followed [see Warnings and Precautions (5.2)].
MONITORING:
Patients must be monitored intensively during the first 18 weeks of therapy with nevirapine to detect potentially life-threatening hepatotoxicity or skin reactions. Extra vigilance is warranted during the first 6 weeks of therapy, which is the period of greatest risk of these events. Do not restart nevirapine following clinical hepatitis, or transaminase elevations combined with rash or other systemic symptoms, or following severe skin rash or hypersensitivity reactions. In some cases, hepatic injury has progressed despite discontinuation of treatment.
1 INDICATIONS AND USAGE
VIRAMUNE XR is indicated for use in combination with other antiretroviral agents for the treatment of HIV-1 infection in adults. This indication is based on one principal clinical trial (1100.1486) that demonstrated prolonged suppression of HIV-1 RNA through 48-weeks and a supportive trial (1100.1526).
Additional important information regarding the use of VIRAMUNE XR for the treatment of HIV-1 infection:
- Based on serious and life-threatening hepatotoxicity observed in controlled and uncontrolled trials, nevirapine should not be initiated in adult females with CD4+ cell counts greater than 250 cells/mm3 or in adult males with CD4+ cell counts greater than 400 cells/mm3 unless the benefit outweighs the risk [see Boxed Warning and Warnings and Precautions (5.1)].
- The 14-day lead-in period with immediate-release VIRAMUNE 200 mg daily dosing must be strictly followed; it has been demonstrated to reduce the frequency of rash [see Dosage and Administration (2.3) and Warnings and Precautions (5.2)].
- If rash persists beyond the 14-day lead-in period with immediate-release VIRAMUNE, do not begin dosing with VIRAMUNE XR. The lead-in dosing with 200 mg once-daily immediate-release VIRAMUNE should not be continued beyond 28 days, at which point an alternative regimen should be sought.
2 DOSAGE AND ADMINISTRATION
2.1 Adults
Patients not currently taking immediate-release VIRAMUNE
Patients must initiate therapy with one 200 mg tablet of immediate-release VIRAMUNE daily for the first 14 days in combination with other antiretroviral agents (this lead-in period should be used because it has been found to lessen the frequency of rash), followed by one 400 mg tablet of VIRAMUNE XR once daily. Patients must swallow VIRAMUNE XR tablets whole. They must not be chewed, crushed, or divided. For concomitantly administered therapy, the manufacturer's recommended dosage and monitoring should be followed. VIRAMUNE XR can be taken with or without food.
Switching Patients from immediate-release VIRAMUNE to VIRAMUNE XR
Patients already on a regimen of immediate-release VIRAMUNE twice daily in combination with other antiretroviral agents can be switched to VIRAMUNE XR 400 mg once daily in combination with other antiretroviral agents without the 14-day lead-in period of immediate-release VIRAMUNE.
Patients must never take more than one form of nevirapine at the same time.
2.2 Monitoring of Patients
Intensive clinical and laboratory monitoring, including liver enzyme tests, is essential at baseline and during the first 18 weeks of treatment with nevirapine. The optimal frequency of monitoring during this period has not been established. Some experts recommend clinical and laboratory monitoring more often than once per month, and in particular, would include monitoring of liver enzyme tests prior to beginning the 14-day lead-in period with immediate-release VIRAMUNE, prior to initiation of VIRAMUNE XR, and at two weeks after initiation of VIRAMUNE XR therapy. After the initial 18-week period, frequent clinical and laboratory monitoring should continue throughout VIRAMUNE XR treatment [see Warnings and Precautions (5)]. In some cases, hepatic injury has progressed despite discontinuation of treatment.
Patients already on a regimen of immediate-release VIRAMUNE twice daily who switch to VIRAMUNE XR once daily should continue with their ongoing clinical and laboratory monitoring.
2.3 Dosage Adjustment
Patients with Rash
Discontinue nevirapine if a patient experiences severe rash or any rash accompanied by constitutional findings [see Boxed Warning, Warnings and Precautions (5.2), and Patient Counseling Information (17.1)]. Do not initiate therapy with VIRAMUNE XR if a patient experiences mild to moderate rash without constitutional symptoms during the 14-day lead-in period of immediate-release VIRAMUNE (200 mg once daily) until the rash has resolved [see Warnings and Precautions (5.2) and Patient Counseling Information (17.1)]. The total duration of the once daily lead-in dosing period should not exceed 28 days at which point an alternative regimen should be sought.
Patients with Hepatic Events
If a clinical (symptomatic) hepatic event occurs, permanently discontinue nevirapine. Do not restart nevirapine after recovery [see Warnings and Precautions (5.1)].
Patients with Dose Interruption
For patients who interrupt VIRAMUNE XR dosing for more than 7 days restart the recommended lead-in dosing with immediate-release VIRAMUNE, using one 200 mg tablet daily for the first 14 days.
Patients with Renal Impairment
Patients with CrCL greater than or equal to 20 mL/min and not requiring dialysis do not require an adjustment in dosing. An additional 200 mg dose of immediate-release VIRAMUNE following each dialysis treatment is indicated in patients requiring dialysis. Nevirapine metabolites may accumulate in patients receiving dialysis; however, the clinical significance of this accumulation is not known [see Clinical Pharmacology (12.3)]. VIRAMUNE XR has not been studied in patients with renal dysfunction.
3 DOSAGE FORMS AND STRENGTHS
Tablets: 400 mg, yellow, oval, biconvex, tablets debossed with "V04" on one side and the Boehringer Ingelheim logo on the other side.
4 CONTRAINDICATIONS
4.1 Hepatic Impairment
VIRAMUNE XR is contraindicated in patients with moderate or severe (Child-Pugh Class B or C, respectively) hepatic impairment [see Warnings and Precautions (5.1) and Use in Specific Populations (8.7)].
4.2 Post-Exposure Prophylaxis
VIRAMUNE XR is contraindicated for use as part of occupational and non-occupational post-exposure prophylaxis (PEP) regimens [see Warnings and Precautions (5.1)].
5 WARNINGS AND PRECAUTIONS
The most serious adverse reactions associated with nevirapine are hepatitis/hepatic failure, Stevens-Johnson syndrome, toxic epidermal necrolysis, and hypersensitivity reactions. Hepatitis/hepatic failure may be associated with signs of hypersensitivity which can include severe rash or rash accompanied by fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial edema, eosinophilia, granulocytopenia, lymphadenopathy, or renal dysfunction.
The first 18 weeks of therapy with nevirapine are a critical period during which intensive clinical and laboratory monitoring of patients is required to detect potentially life-threatening hepatic events and skin reactions. The optimal frequency of monitoring during this time period has not been established. Some experts recommend clinical and laboratory monitoring more often than once per month, and in particular, include monitoring of liver enzyme tests prior to beginning the 14-day lead-in period with immediate-release VIRAMUNE, prior to initiation of VIRAMUNE XR (during the lead-in period), and at two weeks after initiation of VIRAMUNE XR therapy. After the initial 18-week period, frequent clinical and laboratory monitoring should continue throughout VIRAMUNE XR treatment. In addition, the 14-day lead-in period with VIRAMUNE 200 mg daily dosing has been demonstrated to reduce the frequency of rash [see Dosage and Administration (2.1)].
For patients already on a regimen of immediate-release VIRAMUNE twice daily who switch to VIRAMUNE XR therapy, continue with their ongoing clinical and laboratory monitoring.
5.1 Hepatotoxicity and Hepatic Impairment
Severe, life-threatening, and in some cases fatal hepatotoxicity, including fulminant and cholestatic hepatitis, hepatic necrosis and hepatic failure, have been reported in patients treated with nevirapine.
The risk of symptomatic hepatic events regardless of severity is greatest in the first 6 weeks of therapy. The risk continued to be greater in the nevirapine groups in controlled clinical trials through 18 weeks of treatment. However, hepatic events may occur at any time during treatment. In some cases, patients presented with non-specific, prodromal signs or symptoms of fatigue, malaise, anorexia, nausea, jaundice, liver tenderness or hepatomegaly, with or without initially abnormal serum transaminase levels. Rash was observed in approximately half of the patients with symptomatic hepatic adverse events. Fever and flu-like symptoms accompanied some of these hepatic events. Some events, particularly those with rash and other symptoms, have progressed to hepatic failure with transaminase elevation, with or without hyperbilirubinemia, hepatic encephalopathy, prolonged partial thromboplastin time, or eosinophilia. Rhabdomyolysis has been observed in some patients experiencing skin and/or liver reactions associated with nevirapine use. Patients with signs or symptoms of hepatitis must be advised to discontinue nevirapine and immediately seek medical evaluation, which should include liver enzyme tests.
Transaminases should be checked immediately if a patient experiences signs or symptoms suggestive of hepatitis and/or hypersensitivity reaction. Transaminases should also be checked immediately for all patients who develop a rash in the first 18 weeks of treatment. Physicians and patients should be vigilant for the appearance of signs or symptoms of hepatitis, such as fatigue, malaise, anorexia, nausea, jaundice, bilirubinuria, acholic stools, liver tenderness, or hepatomegaly. The diagnosis of hepatotoxicity should be considered in this setting, even if transaminases are initially normal or alternative diagnoses are possible [see Boxed Warning, Dosage and Administration (2.2), and Patient Counseling Information (17.1)].
If clinical hepatitis or transaminase elevations combined with rash or other systemic symptoms occur, permanently discontinue nevirapine. Do not restart nevirapine after recovery. In some cases, hepatic injury progresses despite discontinuation of treatment.
The patients at greatest risk of hepatic events, including potentially fatal events, are women with high CD4+ cell counts. In a retrospective analysis of pooled clinical trials with immediate-release VIRAMUNE, during the first 6 weeks of treatment women had a 3-fold higher risk than men for symptomatic, often rash-associated, hepatic events (6% vs 2%). Patients with higher CD4+ cell counts at initiation of nevirapine therapy are at higher risk for symptomatic hepatic events. Women with CD4+ cell counts greater than 250 cells/mm3 had a 12-fold higher risk of symptomatic hepatic adverse events compared to women with CD4+ cell counts less than 250 cells/mm3 (11% vs 1%). An increased risk was observed in men with CD4+ cell counts greater than 400 cells/mm3 (6% vs 1% for men with CD4+ cell counts less than 400 cells/mm3). However, all patients, regardless of gender, CD4+ cell count, or antiretroviral treatment history, should be monitored for hepatotoxicity since symptomatic hepatic adverse events have been reported at all CD4+ cell counts. Co-infection with hepatitis B or C and/or increased transaminase elevations at the start of therapy with nevirapine are associated with a greater risk of later symptomatic events (6 weeks or more after starting nevirapine) and asymptomatic increases in AST or ALT.
In addition, serious hepatotoxicity (including liver failure requiring transplantation in one instance) has been reported in HIV-1 uninfected individuals receiving multiple doses of immediate-release VIRAMUNE in the setting of post-exposure prophylaxis (PEP), an unapproved use. Use of VIRAMUNE XR for occupational and non-occupational PEP is contraindicated [see Contraindications (4.2)].
Increased nevirapine trough concentrations have been observed in some patients with hepatic fibrosis or cirrhosis. Therefore, carefully monitor patients with either hepatic fibrosis or cirrhosis for evidence of drug-induced toxicity. Do not administer nevirapine to patients with moderate or severe (Child-Pugh Class B or C, respectively) hepatic impairment [see Contraindications (4.1), Use in Specific Populations (8.7), and Clinical Pharmacology (12.3)]. VIRAMUNE XR has not been evaluated in subjects with hepatic impairment.
5.2 Skin Reactions
Severe and life-threatening skin reactions, including fatal cases, have been reported in patients taking nevirapine. These have occurred most frequently during the first 6 weeks of therapy. These have included cases of Stevens-Johnson syndrome, toxic epidermal necrolysis, and hypersensitivity reactions characterized by rash, constitutional findings, and organ dysfunction including hepatic failure. Rhabdomyolysis has been observed in some patients experiencing skin and/or liver reactions associated with nevirapine use.
Patients developing signs or symptoms of severe skin reactions or hypersensitivity reactions (including, but not limited to, severe rash or rash accompanied by fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial edema, and/or hepatitis, eosinophilia, granulocytopenia, lymphadenopathy, and renal dysfunction) must permanently discontinue nevirapine and seek medical evaluation immediately [see Boxed Warning and Patient Counseling Information (17.1)]. Do not restart nevirapine following severe skin rash, skin rash combined with increased transaminases or other symptoms, or hypersensitivity reaction.
If patients present with a suspected nevirapine-associated rash, measure transaminases immediately. Permanently discontinue nevirapine in patients with rash-associated transaminase elevations [see Warnings and Precautions (5.1)].
Patients must initiate therapy with one 200 mg tablet of immediate-release VIRAMUNE daily for the first 14 days. This lead-in period has been shown to reduce the frequency of rash. Discontinue nevirapine if a patient experiences severe rash or any rash accompanied by constitutional findings. Do not initiate VIRAMUNE XR if a patient experiencing a mild to moderate rash without constitutional symptoms during the 14-day immediate-release VIRAMUNE lead-in period of 200 mg/day until the rash has resolved. The total duration of the immediate-release VIRAMUNE lead-in dosing period must not exceed 28 days at which point an alternative regimen should be sought [see Dosage and Administration (2.3)]. Patients must be monitored closely if isolated rash of any severity occurs. Delay in stopping nevirapine treatment after the onset of rash may result in a more serious reaction.
Women appear to be at higher risk than men of developing rash with nevirapine.
In a clinical trial of immediate-release VIRAMUNE, concomitant prednisone use (40 mg/day for the first 14 days of nevirapine administration) was associated with an increase in incidence and severity of rash during the first 6 weeks of nevirapine therapy. Therefore, use of prednisone to prevent nevirapine-associated rash is not recommended.
5.3 Resistance
VIRAMUNE XR must not be used as a single agent to treat HIV-1 or added on as a sole agent to a failing regimen. Resistant virus emerges rapidly when nevirapine is administered as monotherapy. The choice of new antiretroviral agents to be used in combination with nevirapine should take into consideration the potential for cross resistance. When discontinuing an antiretroviral regimen containing VIRAMUNE XR, the long half-life of nevirapine should be taken into account; if antiretrovirals with shorter half-lives than nevirapine are stopped concurrently, low plasma concentrations of nevirapine alone may persist for a week or longer and virus resistance may subsequently develop [see Clinical Pharmacology (12.4)].
5.4 Drug Interactions
See Table 3 for listings of established and potential drug interactions [see Drug Interactions (7)].
Concomitant use of St. John's wort (Hypericum perforatum) or St. John's wort-containing products and nevirapine is not recommended. Co-administration of St. John's wort with non-nucleoside reverse transcriptase inhibitors (NNRTIs), including nevirapine, is expected to substantially decrease NNRTI concentrations and may result in sub-optimal levels of nevirapine and lead to loss of virologic response and possible resistance to nevirapine or to the class of NNRTIs.
Co-administration of nevirapine and efavirenz is not recommended as this combination has been associated with an increase in adverse reactions and no improvement in efficacy.
5.5 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including nevirapine. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jiroveci pneumonia (PCP), or tuberculosis), which may necessitate further evaluation and treatment.
5.6 Fat Redistribution
Redistribution/accumulation of body fat including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The most serious adverse reactions associated with nevirapine are hepatitis, hepatic failure, Stevens-Johnson syndrome, toxic epidermal necrolysis, and hypersensitivity reactions. Hepatitis/hepatic failure may be isolated or associated with signs of hypersensitivity which may include severe rash or rash accompanied by fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial edema, eosinophilia, granulocytopenia, lymphadenopathy, or renal dysfunction [see Boxed Warning and Warnings and Precautions (5.1, 5.2)].
The most common clinical toxicity of nevirapine is rash, which can be severe or life-threatening [see Boxed Warning and Warnings and Precautions (5.2)]. Rash occurs most frequently within the first 6 weeks of therapy. Rashes are usually mild to moderate, maculopapular erythematous cutaneous eruptions, with or without pruritus, located on the trunk, face and extremities.
The safety database in VIRAMUNE XR clinical trials contains data from 800 subjects treated with VIRAMUNE XR and 654 subjects treated with immediate release VIRAMUNE.
Trial 1100.1486 (VERxVE)
In Trial 1100.1486 (VERxVE) treatment-naïve subjects received a lead-in dose of immediate-release VIRAMUNE 200 mg once daily for 14 days (n=1068) and then were randomized to receive either immediate-release VIRAMUNE 200 mg twice daily (n=506) or VIRAMUNE XR 400 mg once daily (n=505). All subjects received tenofovir + emtricitabine as background therapy. Subjects were enrolled with CD4+ counts less than 250 cells/mm3 for women and less than 400 cells/mm3 for men [see Indications and Usage (1)]. Data on potential symptoms of hepatic events were prospectively collected in this trial. The safety data include all subject visits up to the time of the last subject’s completion of the 48 week primary endpoint in the trial (mean observation period 61 weeks).
After the lead-in period, the incidence of any hepatic event was 9% in the immediate-release VIRAMUNE group and 6% in the VIRAMUNE XR group; the incidence of symptomatic hepatic events (anorexia, jaundice, vomiting) was 3% and 2%, respectively. The incidence of GRADE 3 or 4 ALT/AST elevation was 7% in the immediate-release VIRAMUNE group and 6% in the VIRAMUNE XR group. Overall, there was a comparable incidence of symptomatic hepatic events among men and women enrolled in VERxVE.
Severe or life-threatening rash considered to be related to nevirapine treatment occurred in 1% of subjects during the lead-in phase with immediate-release VIRAMUNE, and in 1% of subjects in either treatment group during the randomization phase. In addition, five cases of Stevens-Johnson syndrome were reported in the trial, all of which occurred within the first 30 days of nevirapine treatment.
No Grade 2 or above adverse reactions judged to be related to treatment by the investigator occurred in more than 2% of subjects during the 14-day lead-in with immediate-release VIRAMUNE (200 mg once daily), with the exception of rash which occurred in 4% of subjects.
Adverse reactions of at least moderate intensity (Grades 2 or above) and considered to be related to treatment by the investigator in at least 2% of treatment-naive subjects receiving either immediate-release VIRAMUNE or VIRAMUNE XR after randomization in Trial 1100.1486 are shown in Table 1.
| Adverse Drug Reaction (%) | VIRAMUNE Immediate-Release N=506 | VIRAMUNE XR N=505 |
| 1 Includes adverse drug reactions considered by the investigator to be at least possibly, probably, or definitely related to the drug. 2 Mean observation period 61 weeks. 3 Rash includes terms rash, rash maculo-papular, erythema nodosum, rash erythematous, rash papular, skin reaction, Stevens-Johnson syndrome, drug rash with eosinophilia and systemic symptoms (DRESS), and allergic dermatitis. 4 Clinical hepatitis includes terms hepatitis, hepatotoxicity, hepatitis acute, liver disorder, hepatitis toxic, hepatic failure, jaundice. |
||
| Rash3 | 3% | 3% |
| Clinical hepatitis4 | 3% | 2% |
Laboratory Abnormalities
Liver enzyme test abnormalities (AST, ALT) were observed in subjects receiving VIRAMUNE XR. Asymptomatic elevations in GGT occur frequently but are not a contraindication to continue therapy with nevirapine in the absence of elevations in other liver enzyme tests. Selected laboratory abnormalities that occurred in trial 1100.1486 are shown in Table 2.
| Laboratory Parameter (unit) | Limit | VIRAMUNE Immediate-Release N=506 (%) | VIRAMUNE XR N=505 (%) |
| Chemistry
| |||
| SGPT/ALT (U/L) | |||
| Grade 2 | 2.6-5.0 x ULN | 52 (10) | 44 (9) |
| Grade 3 | 5.1-10.0 x ULN | 18 (4) | 14 (3) |
| Grade 4 | >10.0 x ULN | 18 (4) | 10 (2) |
|
SGOT/AST (U/L) | |||
| Grade 2 | 2.6-5.0 x ULN | 43 (9) | 31 (6) |
| Grade 3 | 5.1-10.0 x ULN | 11 (2) | 13 (3) |
| Grade 4 | >10.0 x ULN | 10 (2) | 7 (1) |
|
Total Bilirubin (mg/dL) | |||
| Grade 2 | 1.6-2.5 x ULN | 2 (<1) | 4 (1) |
| Grade 3 | 2.6-5.0 x ULN | 3 (1) | 3 (1) |
| Grade 4 | >5.0 x ULN | 1 (<1) | 5 (1) |
|
Alkaline Phosphatase (U/L) | |||
| Grade 2 | 2.6-5.0 x ULN | 18 (4) | 11 (2) |
| Grade 3 | 5.1-10.0 x ULN | 1 (<1) | 4 (1) |
| Grade 4 | >10.0 x ULN | 0 | 0 |
| Hematology
| |||
| Neutrophils | |||
| Grade 2 | 750-999/mm3 | 33 (7) | 22 (4) |
| Grade 3 | 500-749/mm3 | 13 (3) | 8 (2) |
| Grade 4 | <500/mm3 | 3 (1) | 2 (<1) |
| Lipids
| |||
| LDL (mg/dL) | |||
| Grade 2 | 160-190 mg/dL | 66 (13) | 66 (13) |
| Grade 3 | >190 mg/dL | 21 (4) | 20 (4) |
|
Cholesterol (mg/dL) | |||
| Grade 2 | 240-300 mg/dL | 81 (16) | 83 (16) |
| Grade 3 | >300 mg/dL | 14 (4) | 9 (2) |
|
Triglycerides (mg/dL) | |||
| Grade 2 | 500-750 mg/dL | 10 (2) | 15 (3) |
| Grade 3 | 751-1,200 mg/dL | 9 (2) | 4 (1) |
| Grade 4 | >1,200 mg/dL | 1 (<1) | 2 (<1) |
Trial 1100.1526 (TRANxITION)
In Trial 1100.1526 (TRANxITION) subjects on immediate-release VIRAMUNE 200 mg twice daily for at least 18 weeks were randomized to either receive VIRAMUNE XR 400 mg once daily (n=295) or remain on their immediate-release VIRAMUNE treatment (n=148). There were no adverse reactions of Grades 2 or above and considered to be related to treatment by the investigator that occurred in more than 1% of subjects (24 week analysis).
6.2 Post-Marketing Experience
The following adverse reactions have been identified during post-approval use of immediate-release VIRAMUNE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Body as a Whole: fever, somnolence, drug withdrawal [see Drug Interactions (7)], redistribution/accumulation of body fat [see Warnings and Precautions (5.6)]
- Gastrointestinal: vomiting
- Liver and Biliary: jaundice, fulminant and cholestatic hepatitis, hepatic necrosis, hepatic failure
- Hematology: anemia, eosinophilia, neutropenia
- Investigations: decreased serum phosphorus
- Musculoskeletal: arthralgia, rhabdomyolysis associated with skin and/or liver reactions
- Neurologic: paraesthesia
- Skin and Appendages: allergic reactions including anaphylaxis, angioedema, bullous eruptions, ulcerative stomatitis and urticaria have all been reported. In addition, hypersensitivity syndrome and hypersensitivity reactions with rash associated with constitutional findings such as fever, blistering, oral lesions, conjunctivitis, facial edema, muscle or joint aches, general malaise, fatigue, or significant hepatic abnormalities [see Warnings and Precautions (5.1)] plus one or more of the following: hepatitis, eosinophilia, granulocytopenia, lymphadenopathy, and/or renal dysfunction have been reported.
7 DRUG INTERACTIONS
Nevirapine is principally metabolized by the liver via the cytochrome P450 isoenzymes, 3A and 2B6. Nevirapine is known to be an inducer of these enzymes. As a result, drugs that are metabolized by these enzyme systems may have lower than expected plasma levels when co-administered with nevirapine.
The results of drug interactions studies with immediate-release VIRAMUNE are expected to also apply to VIRAMUNE XR. The specific pharmacokinetic changes that occur with co-administration of nevirapine and other drugs are listed in Clinical Pharmacology, Table 4. Clinical comments about possible dosage modifications based on established drug interactions are listed in Table 3. The data in Tables 3 and 4 are based on the results of drug interaction studies conducted in HIV-1 seropositive subjects unless otherwise indicated. In addition to established drug interactions, there may be potential pharmacokinetic interactions between nevirapine and other drug classes that are metabolized by the cytochrome P450 system. These potential drug interactions are also listed in Table 3. Although specific drug interaction studies in HIV-1 seropositive subjects have not been conducted for some classes of drugs listed in Table 3, additional clinical monitoring may be warranted when co-administering these drugs.
The in vitro interaction between nevirapine and the antithrombotic agent warfarin is complex. As a result, when giving these drugs concomitantly, plasma warfarin levels may change with the potential for increases in coagulation time. When warfarin is co-administered with nevirapine, anticoagulation levels should be monitored frequently.
| Drug Name | Effect on Concentration of Nevirapine or Concomitant Drug | Clinical Comment |
| Atazanavir/Ritonavir | ↓ Atazanavir ↑ Nevirapine | Do not co-administer nevirapine with atazanavir because nevirapine substantially decreases atazanavir exposure. |
| Clarithromycin | ↓ Clarithromycin ↑ 14-OH clarithromycin | Clarithromycin exposure was significantly decreased by nevirapine; however, 14-OH metabolite concentrations were increased. Because clarithromycin active metabolite has reduced activity against Mycobacterium avium-intracellulare complex, overall activity against this pathogen may be altered. Alternatives to clarithromycin, such as azithromycin, should be considered. |
| Efavirenz | ↓ Efavirenz | There has been no determination of appropriate doses for the safe and effective use of this combination [see Warnings and Precautions (5.4)]. |
| Ethinyl estradiol and Norethindrone | ↓ Ethinyl estradiol ↓ Norethindrone | Oral contraceptives and other hormonal methods of birth control should not be used as the sole method of contraception in women taking nevirapine, since nevirapine may lower the plasma levels of these medications. An alternative or additional method of contraception is recommended. |
| Fluconazole | ↑Nevirapine | Because of the risk of increased exposure to nevirapine, caution should be used in concomitant administration, and patients should be monitored closely for nevirapine-associated adverse events. |
| Fosamprenavir | ↓Amprenavir ↑Nevirapine | Co-administration of nevirapine and fosamprenavir without ritonavir is not recommended. |
| Fosamprenavir/Ritonavir | ↓Amprenavir ↑Nevirapine | No dosing adjustments are required when nevirapine is co-administered with 700/100 mg of fosamprenavir/ritonavir twice daily. |
| Indinavir | ↓ Indinavir | Appropriate doses for this combination are not established, but an increase in the dosage of indinavir may be required. |
| Ketoconazole | ↓ Ketoconazole | Nevirapine and ketoconazole should not be administered concomitantly because decreases in ketoconazole plasma concentrations may reduce the efficacy of the drug. |
| Lopinavir/Ritonavir | ↓Lopinavir | Lopinavir/ritonavir 400/100 mg tablets can be used twice-daily in combination with nevirapine with no dose adjustment in antiretroviral-naïve patients. A dose increase of lopinavir/ritonavir tablets to 600/150 mg (3 tablets) twice daily may be considered when used in combination with nevirapine in treatment-experienced patients where decreased susceptibility to lopinavir is clinically suspected (by treatment history or laboratory evidence). A dose increase of lopinavir/ritonavir oral solution to 533/133 mg twice daily with food is recommended in combination with nevirapine. In children 6 months to 12 years of age, consideration should be given to increasing the dose of lopinavir/ritonavir to 13/3.25 mg/kg for those 7 to <15 kg; 11/2.75 mg/kg for those 15 to 45 kg; and up to a maximum dose of 533/133 mg for those >45 kg twice daily when used in combination with nevirapine, particularly for patients in whom reduced susceptibility to lopinavir/ritonavir is suspected. |
| Methadone | ↓ Methadone | Methadone levels were decreased; increased dosages may be required to prevent symptoms of opiate withdrawal. Methadone-maintained patients beginning nevirapine therapy should be monitored for evidence of withdrawal and methadone dose should be adjusted accordingly. |
| Nelfinavir | ↓Nelfinavir M8 Metabolite ↓Nelfinavir Cmin | The appropriate dose for nelfinavir in combination with nevirapine, with respect to safety and efficacy, has not been established. |
| Rifabutin | ↑Rifabutin | Rifabutin and its metabolite concentrations were moderately increased. Due to high intersubject variability, however, some patients may experience large increases in rifabutin exposure and may be at higher risk for rifabutin toxicity. Therefore, caution should be used in concomitant administration. |
| Rifampin | ↓ Nevirapine | Nevirapine and rifampin should not be administered concomitantly because decreases in nevirapine plasma concentrations may reduce the efficacy of the drug. Physicians needing to treat patients co-infected with tuberculosis and using a nevirapine-containing regimen may use rifabutin instead. |
| Saquinavir/ritonavir | The interaction between nevirapine and saquinavir/ritonavir has not been evaluated | The appropriate doses of the combination of nevirapine and saquinavir/ritonavir with respect to safety and efficacy have not been established. |
|
Potential Drug Interactions: | ||
| Drug Class | Examples of Drugs | |
| Antiarrhythmics | Amiodarone, disopyramide, lidocaine | Plasma concentrations may be decreased. |
| Anticonvulsants | Carbamazepine, clonazepam, ethosuximide | Plasma concentrations may be decreased. |
| Antifungals | Itraconazole | Plasma concentrations of some azole antifungals may be decreased. Nevirapine and itraconazole should not be administered concomitantly due to a potential decrease in itraconazole plasma concentrations. |
| Calcium channel blockers | Diltiazem, nifedipine, verapamil | Plasma concentrations may be decreased. |
| Cancer chemotherapy | Cyclophosphamide | Plasma concentrations may be decreased. |
| Ergot alkaloids | Ergotamine | Plasma concentrations may be decreased. |
| Immunosuppressants | Cyclosporin, tacrolimus, sirolimus | Plasma concentrations may be decreased. |
| Motility agents | Cisapride | Plasma concentrations may be decreased. |
| Opiate agonists | Fentanyl | Plasma concentrations may be decreased. |
| Antithrombotics | Warfarin | Plasma concentrations may be increased. Potential effect on anticoagulation. Monitoring of anticoagulation levels is recommended. |
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects, Pregnancy Category B.
No observable teratogenicity was detected in reproductive studies performed in pregnant rats and rabbits. The maternal and developmental no-observable-effect level dosages produced systemic exposures approximately equivalent to or approximately 50% higher in rats and rabbits, respectively, than those seen at the recommended daily human dose (based on AUC). In rats, decreased fetal body weights were observed due to administration of a maternally toxic dose (exposures approximately 50% higher than that seen at the recommended human clinical dose).
There are no adequate and well-controlled studies of nevirapine in pregnant women. The Antiretroviral Pregnancy Registry, which has been surveying pregnancy outcomes since January 1989, has not found an increased risk of birth defects following first trimester exposures to nevirapine. The prevalence of birth defects after any trimester exposure to nevirapine is comparable to the prevalence observed in the general population.
Severe hepatic events, including fatalities, have been reported in pregnant women receiving chronic nevirapine therapy as part of combination treatment of HIV-1 infection. Regardless of pregnancy status, women with CD4+ cell counts greater than 250 cells/mm3 should not initiate nevirapine unless the benefit outweighs the risk. It is unclear if pregnancy augments the risk observed in non-pregnant women [see Boxed Warning].
VIRAMUNE XR should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
8.3 Nursing Mothers
The Centers for Disease Control and Prevention recommend that HIV-1 infected mothers not breastfeed their infants to avoid risking postnatal transmission of HIV-1. Nevirapine is excreted in breast milk. Because of both the potential for HIV-1 transmission and the potential for serious adverse reactions in nursing infants, mothers should be instructed not to breastfeed if they are receiving VIRAMUNE XR.
8.4 Pediatric Use
The safety and efficacy of VIRAMUNE XR in pediatric patients have not been established. Please consult the complete prescribing information for immediate-release VIRAMUNE tablets and VIRAMUNE Oral Suspension for dosage and administration of nevirapine to pediatric patients.
8.5 Geriatric Use
Clinical studies of VIRAMUNE XR did not include sufficient numbers of subjects aged 65 and older to determine whether elderly subjects respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
8.6 Renal Impairment
In subjects with renal impairment (mild, moderate or severe), there were no significant changes in the pharmacokinetics of nevirapine. Nevirapine is extensively metabolized by the liver and nevirapine metabolites are extensively eliminated by the kidney. Nevirapine metabolites may accumulate in patients receiving dialysis; however, the clinical significance of this accumulation is not known. No adjustment in nevirapine dosing is required in patients with CrCL greater than or equal to 20 mL/min. In patients undergoing chronic hemodialysis, an additional dose of immediate-release VIRAMUNE (200 mg) following each dialysis treatment is indicated [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)]. VIRAMUNE XR has not been studied in patients with renal dysfunction.
8.7 Hepatic Impairment
Because increased nevirapine levels and nevirapine accumulation may be observed in patients with serious liver disease, do not administer nevirapine to patients with moderate or severe (Child-Pugh Class B or C, respectively) hepatic impairment [see Contraindications (4), Warnings and Precautions (5.1), and Clinical Pharmacology (12.3)]. VIRAMUNE XR has not been evaluated in subjects with hepatic impairment.
10 OVERDOSAGE
There is no known antidote for nevirapine overdosage. Cases of immediate-release VIRAMUNE overdose at doses ranging from 800 to 1800 mg per day for up to 15 days have been reported. Patients have experienced events including edema, erythema nodosum, fatigue, fever, headache, insomnia, nausea, pulmonary infiltrates, rash, vertigo, vomiting and weight decrease. All events subsided following discontinuation of immediate-release VIRAMUNE.
11 DESCRIPTION
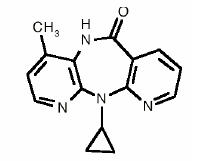
VIRAMUNE XR is the brand name for nevirapine extended-release tablets. Nevirapine is a non-nucleoside reverse transcriptase inhibitor (NNRTI) with activity against Human Immunodeficiency Virus Type 1 (HIV-1). Nevirapine is structurally a member of the dipyridodiazepinone chemical class of compounds.
The chemical name of nevirapine is 11-cyclopropyl-5,11-dihydro-4-methyl-6H-dipyrido [3,2-b:2',3'-e][1,4] diazepin-6-one. Nevirapine is a white to off-white crystalline powder with the molecular weight of 266.30 and the molecular formula C15H14N4O. Nevirapine has the following structural formula:
VIRAMUNE XR Tablets are for oral administration. Each tablet contains 400 mg of nevirapine and the inactive ingredients lactose monohydrate, hypromellose, iron oxide, and magnesium stearate.
12 CLINICAL PHARMACOLOGY
12.3 Pharmacokinetics
Adults
Absorption and Bioavailability
The single-dose pharmacokinetics of VIRAMUNE XR was studied in 17 healthy volunteers. Nevirapine was absorbed with a median tmax of approximately 24 hrs. The mean Cmax and AUC0-∞ of nevirapine were 2060 ng/mL and 161,000 ng*hr/mL, respectively. The bioavailability of 400 mg of VIRAMUNE XR, relative to 400 mg of immediate-release VIRAMUNE, was approximately 75%.
The multiple-dose pharmacokinetics of VIRAMUNE XR was studied in 24 HIV-1 infected subjects who switched from chronic VIRAMUNE IR to VIRAMUNE XR. The mean nevirapine AUC0-24,ss and Cmin,ss after 19 days of VIRAMUNE XR dosing under fasted conditions were 82,000 ng*hr/mL and 2920 ng/mL, respectively. When VIRAMUNE XR was administered under fed conditions, the mean nevirapine AUC0-24,ss and Cmin,ss were 96,700 ng*hr/mL and 3150 ng/mL, respectively. The bioavailability of 400 mg of VIRAMUNE XR, relative to 400 mg of immediate-release VIRAMUNE, under fasted and fed conditions, was 80% and 94%, respectively. The difference in the bioavailability of nevirapine, when VIRAMUNE XR is dosed under fasted or fed conditions, is not considered clinically relevant. VIRAMUNE XR can be taken with or without food.
Distribution
Nevirapine is highly lipophilic and is essentially nonionized at physiologic pH. Following intravenous administration to healthy adults, the apparent volume of distribution (Vdss) of nevirapine was 1.21 ± 0.09 L/kg, suggesting that nevirapine is widely distributed in humans. Nevirapine readily crosses the placenta and is also found in breast milk [see Use In Specific Populations (8.3)]. Nevirapine is about 60% bound to plasma proteins in the plasma concentration range of 1-10 mcg/mL. Nevirapine concentrations in human cerebrospinal fluid (n=6) were 45% (±5%) of the concentrations in plasma; this ratio is approximately equal to the fraction not bound to plasma protein.
Metabolism/Elimination
In vivo trials in humans and in vitro studies with human liver microsomes have shown that nevirapine is extensively biotransformed via cytochrome P450 (oxidative) metabolism to several hydroxylated metabolites. In vitro studies with human liver microsomes suggest that oxidative metabolism of nevirapine is mediated primarily by cytochrome P450 (CYP) isozymes from the CYP3A and CYP2B6 families, although other isozymes may have a secondary role. In a mass balance/excretion trial in eight healthy male volunteers dosed to steady state with immediate-release VIRAMUNE 200 mg given twice daily followed by a single 50 mg dose of 14C-nevirapine, approximately 91.4 ± 10.5% of the radiolabeled dose was recovered, with urine (81.3 ± 11.1%) representing the primary route of excretion compared to feces (10.1 ± 1.5%). Greater than 80% of the radioactivity in urine was made up of glucuronide conjugates of hydroxylated metabolites. Thus cytochrome P450 metabolism, glucuronide conjugation, and urinary excretion of glucuronidated metabolites represent the primary route of nevirapine biotransformation and elimination in humans. Only a small fraction (less than 5%) of the radioactivity in urine (representing less than 3% of the total dose) was made up of parent compound; therefore, renal excretion plays a minor role in elimination of the parent compound.
Nevirapine is an inducer of hepatic cytochrome P450 (CYP) metabolic enzymes 3A and 2B6. Nevirapine induces CYP3A and CYP2B6 by approximately 20-25%, as indicated by erythromycin breath test results and urine metabolites. Autoinduction of CYP3A and CYP2B6 mediated metabolism leads to an approximately 1.5- to 2-fold increase in the apparent oral clearance of nevirapine as treatment continues from a single dose to two-to-four weeks of dosing with 200-400 mg/day of immediate-release VIRAMUNE. Autoinduction also results in a corresponding decrease in the terminal phase half-life of nevirapine in plasma, from approximately 45 hours (single dose) to approximately 25-30 hours following multiple dosing with 200-400 mg/day.
Specific Populations
Renal Impairment
HIV-1 seronegative adults with mild (CrCL 50-79 mL/min; n=7), moderate (CrCL 30-49 mL/min; n=6), or severe (CrCL less than 30 mL/min; n=4) renal impairment received a single 200 mg dose of immediate-release VIRAMUNE in a pharmacokinetic trial. These subjects did not require dialysis. The trial included six additional subjects with renal failure requiring dialysis.
In subjects with renal impairment (mild, moderate or severe), there were no significant changes in the pharmacokinetics of nevirapine. However, subjects requiring dialysis exhibited a 44% reduction in nevirapine AUC over a one-week exposure period. There was also evidence of accumulation of nevirapine hydroxy-metabolites in plasma in subjects requiring dialysis. An additional 200 mg dose of immediate-release VIRAMUNE following each dialysis treatment is indicated [see Dosage and Administration (2.3) and Use in Specific Populations (8.6)]. VIRAMUNE XR has not been studied in patients with renal dysfunction.
Hepatic Impairment
In a steady-state trial comparing 46 subjects with mild (n=17; expansion of some portal areas; Ishak Score 1-2), moderate (n=20; expansion of most portal areas with occasional portal-to-portal and portal-to-central bridging; Ishak Score 3-4), or severe (n=9; marked bridging with occasional cirrhosis without decompensation indicating Child-Pugh A; Ishak Score 5-6) fibrosis as a measure of hepatic impairment, the multiple dose pharmacokinetic disposition of nevirapine and its five oxidative metabolites were not altered. However, approximately 15% of these subjects with hepatic fibrosis had nevirapine trough concentrations above 9,000 mcg/mL (2-fold the usual mean trough). Therefore, patients with hepatic impairment should be monitored carefully for evidence of drug-induced toxicity [see Warnings and Precautions (5.1)]. The subjects studied were receiving antiretroviral therapy containing immediate-release VIRAMUNE 200 mg twice daily for at least 6 weeks prior to pharmacokinetic sampling, with a median duration of therapy of 3.4 years.
In a pharmacokinetic trial where HIV-1 negative cirrhotic subjects with mild (Child-Pugh A; n=6) or moderate (Child-Pugh B; n=4) hepatic impairment received a single 200 mg dose of immediate-release VIRAMUNE, a significant increase in the AUC of nevirapine was observed in one subject with Child-Pugh B and ascites suggesting that patients with worsening hepatic function and ascites may be at risk of accumulating nevirapine in the systemic circulation. Because nevirapine induces its own metabolism with multiple dosing, this single-dose trial may not reflect the impact of hepatic impairment on multiple-dose pharmacokinetics.
Do not administer nevirapine to patients with moderate or severe (Child-Pugh Class B or C, respectively) hepatic impairment [see Contraindications (4), Warnings and Precautions (5.1), and Use in Specific Populations (8.7)]. VIRAMUNE XR has not been evaluated in patients with hepatic impairment.
Gender
In the multinational 2NN trial of immediate-release VIRAMUNE, a population pharmacokinetic substudy of 1077 subjects was performed that included 391 females. Female subjects showed a 13.8% lower clearance of nevirapine than did men. Since neither body weight nor Body Mass Index (BMI) had an influence on the clearance of nevirapine, the effect of gender cannot solely be explained by body size.
The effects of gender on the pharmacokinetics of VIRAMUNE XR have been investigated in Trial 1100.1486. Female subjects tend to have higher (approximately 20 - 30%) trough concentrations in both VIRAMUNE XR and immediate-release VIRAMUNE treatment groups.
Race
An evaluation of nevirapine plasma concentrations (pooled data from several clinical trials) from HIV-1-infected subjects (27 Black, 24 Hispanic, 189 Caucasian) revealed no marked difference in nevirapine steady-state trough concentrations (median Cminss = 4.7 mcg/mL Black, 3.8 mcg/mL Hispanic, 4.3 mcg/mL Caucasian) with long-term treatment with immediate-release VIRAMUNE at 400 mg/day. However, the pharmacokinetics of nevirapine have not been evaluated specifically for the effects of ethnicity.
Black subjects (n=80/group) in Trial 1100.1486 showed approximately 30% higher trough concentrations than Caucasian subjects (250-325 subjects/group) in both immediate-release VIRAMUNE and VIRAMUNE XR treatment groups over 48 weeks of treatment at 400 mg/day.
Geriatric Patients
Nevirapine pharmacokinetics in HIV-1-infected adults do not appear to change with age (range 18–68 years); however, nevirapine has not been extensively evaluated in patients beyond the age of 65 years [see Use in Specific Populations (8.5)].
Drug Interactions [see Drug Interactions (7)]
Nevirapine induces hepatic cytochrome P450 metabolic isoenzymes 3A and 2B6. Co-administration of VIRAMUNE XR and drugs primarily metabolized by CYP3A or CYP2B6 may result in decreased plasma concentrations of these drugs and attenuate their therapeutic effects.
While primarily an inducer of cytochrome P450 3A and 2B6 enzymes, nevirapine may also inhibit this system. Among human hepatic cytochrome P450s, nevirapine was capable in vitro of inhibiting the 10-hydroxylation of (R)-warfarin (CYP3A). The estimated Ki for the inhibition of CYP3A was 270 micromolar, a concentration that is unlikely to be achieved in patients as the therapeutic range is less than 25 micromolar. Therefore, nevirapine may have minimal inhibitory effect on other substrates of CYP3A.
Nevirapine does not appear to affect the plasma concentrations of drugs that are substrates of other CYP450 enzyme systems, such as 1A2, 2D6, 2A6, 2E1, 2C9, or 2C19.
Table 4 (see below) contains the results of drug interaction trials performed with immediate-release VIRAMUNE and other drugs likely to be co-administered. The effects of nevirapine on the AUC, Cmax, and Cmin of co-administered drugs are summarized. Results of drug interaction studies with immediate-release VIRAMUNE are expected to also apply to VIRAMUNE XR.
| Co-administered Drug | Dose of Co-administered Drug | Dose Regimen of immediate-release VIRAMUNE | n | % Change of Co-administered Drug Pharmacokinetic Parameters (90% CI) | ||
|---|---|---|---|---|---|---|
| § = Cmin below detectable level of the assay ↑ = Increase, ↓ = Decrease, ⇔ = No Effect a For information regarding clinical recommendations, see Drug Interactions (7). b Pediatric subjects ranging in age from 6 months to 12 years c Parallel group design; n for VIRAMUNE+lopinavir/ritonavir, n for lopinavir/ritonavir alone. d Parallel group design; n=23 for atazanavir/ritonavir + nevirapine, n=22 for atazanavir/ritonavir without nevirapine. Changes in atazanavir PK are relative to atazanavir/ritonavir 300/100 mg alone. e Based on between-trial comparison. f Based on historical controls. |
||||||
| Antiretrovirals | AUC | Cmax | Cmin | |||
| Atazanavir/Ritonavira, d | 300/100 mg QD day 4–13, then 400/100 mg QD, day 14–23 | 200 mg BID day 1-23. Subjects were treated with nevirapine prior to trial entry. | 23 | Atazanavir 300/100 mg ↓42 (↓52 to ↓29) | Atazanavir 300/100 mg ↓28 (↓40 to ↓14) | Atazanavir 300/100 mg ↓72 (↓80 to ↓60) |
| Atazanavir 400/100 mg ↓19 (↓35 to ↑2) | Atazanavir 400/100 mg ↑2 (↓15 to ↑24) | Atazanavir 400/100 mg ↓59 (↓73 to ↓40) |
||||
| Darunavir/Ritonavir e | 400/100 mg BID | 200 mg BID | 8 | ↑24 (↓3 to ↑57) | ↑40 (↑14 to ↑73) | ↑2 (↓21 to ↑32) |
| Didanosine | 100-150 mg BID | 200 mg QD x 14 days; 200 mg BID x 14 days | 18 | ⇔ | ⇔ | § |
| Efavirenza | 600 mg QD | 200 mg QD x 14 days; 400 mg QD x 14 days | 17 | ↓28 (↓34 to ↓14) | ↓12 (↓23 to ↑1) | ↓32 (↓35 to ↓19) |
| Fosamprenavir | 1400 mg BID | 200 mg BID. Subjects were treated with nevirapine prior to trial entry. | 17 | ↓33 (↓45 to ↓20) | ↓25 (↓37 to ↓10) | ↓35 (↓50 to ↓15) |
| Fosamprenavir/Ritonavir | 700/100 mg BID | 200 mg BID. Subjects were treated with nevirapine prior to trial entry | 17 | ↓11 (↓23 to ↑3) | ⇔ | ↓19 (↓32 to ↓4) |
| Indinavira | 800 mg q8H | 200 mg QD x 14 days; 200 mg BID x 14 days | 19 | ↓31 (↓39 to ↓22) | ↓15 (↓24 to ↓4) | ↓44 (↓53 to ↓33) |
| Lopinavira, b | 300/75 mg/m2 (lopinavir/ ritonavir) b | 7 mg/kg or 4 mg/kg QD x 2 weeks; BID x 1 week | 12, 15 c | ↓22 (↓44 to ↑9) | ↓14 (↓36 to ↑16) | ↓55 (↓75 to ↓19) |
| Lopinavira | 400/100 mg BID (lopinavir/ritonavir) | 200 mg QD x 14 days; 200 mg BID >1 year | 22, 19 c | ↓27 (↓47 to ↓2) | ↓19 (↓38 to ↑5) | ↓51 (↓72 to ↓26) |
| Maraviroc f | 300 mg SD | 200 mg BID | 8 | ↑1 (↓35 to ↑55) | ↑54 (↓6 to ↑151) | ⇔ |
| Nelfinavira | 750 mg TID | 200 mg QD x 14 days; 200 mg BID x 14 days | 23 | ⇔ | ⇔ | ↓32 (↓50 to ↑5) |
| Nelfinavir-M8 metabolite | ↓62 (↓70 to ↓53) | ↓59 (↓68 to ↓48) | ↓66 (↓74 to ↓55) |
|||
| Ritonavir | 600 mg BID | 200 mg QD x 14 days; 200 mg BID x 14 days | 18 | ⇔ | ⇔ | ⇔ |
| Stavudine | 30-40 mg BID | 200 mg QD x 14 days; 200 mg BID x 14 days | 22 | ⇔ | ⇔ | § |
| Zalcitabine | 0.125-0.25 mg TID | 200 mg QD x 14 days; 200 mg BID x 14 days | 6 | ⇔ | ⇔ | § |
| Zidovudine | 100-200 mg TID | 200 mg QD x 14 days; 200 mg BID x 14 days | 11 | ↓28 (↓40 to ↓4) | ↓30 (↓51 to ↑14) | § |
| Other Medications | AUC | Cmax | Cmin | |||
| Clarithromycina | 500 mg BID | 200 mg QD x 14 days; 200 mg BID x 14 days | 15 | ↓31 (↓38 to ↓24) | ↓23 (↓31 to ↓14) | ↓56 (↓70 to ↓36) |
| Metabolite 14-OH-clarithromycin | ↑42 (↑16 to ↑73) | ↑47 (↑21 to ↑80) | ⇔ | |||
| Ethinyl estradiola
and Norethindronea | 0.035 mg (as Ortho-Novum® 1/35) | 200 mg QD x 14 days; 200 mg BID x 14 days | 10 | ↓20 (↓33 to ↓3) | ⇔ | § |
| 1 mg (as Ortho-Novum® 1/35) | ↓19 (↓30 to ↓7) | ↓16 (↓27 to ↓3) | § | |||
| Depomedroxy-progesterone acetate | 150 mg every 3 months | 200 mg QD x 14 days; 200 mg BID x 14 days | 32 | ⇔ | ⇔ | ⇔ |
| Fluconazole | 200 mg QD | 200 mg QD x 14 days; 200 mg BID x 14 days | 19 | ⇔ | ⇔ | ⇔ |
| Ketoconazolea | 400 mg QD | 200 mg QD x 14 days; 200 mg BID x 14 days | 21 | ↓72 (↓80 to ↓60) | ↓44 (↓58 to ↓27) | § |
| Methadonea | Individual Subject Dosing | 200 mg QD x 14 days; 200 mg BID ≥7 days | 9 | In a controlled pharmacokinetic trial with 9 subjects receiving chronic methadone to whom steady-state nevirapine therapy was added, the clearance of methadone was increased by 3-fold, resulting in symptoms of withdrawal, requiring dose adjustments in 10 mg segments, in 7 of the 9 subjects. Methadone did not have any effect on nevirapine clearance. | ||
| Rifabutina | 150 or 300 mg QD | 200 mg QD x 14 days; 200 mg BID x 14 days | 19 | ↑17 (↓2 to ↑40) | ↑28 (↑9 to ↑51) | ⇔ |
| Metabolite 25-O-desacetyl-rifabutin | ↑24 (↓16 to ↑84) | ↑29 (↓2 to ↑68) | ↑22 (↓14 to ↑74) |
|||
| Rifampina | 600 mg QD | 200 mg QD x 14 days; 200 mg BID x 14 days | 14 | ↑11 (↓4 to ↑28) | ⇔ | § |
Because of the design of the drug interaction trials (addition of 28 days of VIRAMUNE therapy to existing HIV-1 therapy), the effect of the concomitant drug on plasma nevirapine steady-state concentrations was estimated by comparison to historical controls.
Administration of rifampin had a clinically significant effect on nevirapine pharmacokinetics, decreasing AUC and Cmax by greater than 50%. Administration of fluconazole resulted in an approximate 100% increase in nevirapine exposure, based on a comparison to historic data [see Drug Interactions (7)]. The effect of other drugs listed in Table 4 on nevirapine pharmacokinetics was not significant. No significant interaction was observed when tipranavir was co-administered with low-dose ritonavir and nevirapine.
12.4 Microbiology
Mechanism of Action
Nevirapine is a non-nucleoside reverse transcriptase inhibitor (NNRTI) of HIV-1. Nevirapine binds directly to reverse transcriptase (RT) and blocks the RNA-dependent and DNA-dependent DNA polymerase activities by causing a disruption of the enzyme's catalytic site. The activity of nevirapine does not compete with template or nucleoside triphosphates. HIV-2 RT and eukaryotic DNA polymerases (such as human DNA polymerases α, β, γ, or δ) are not inhibited by nevirapine.
Antiviral Activity
The antiviral activity of nevirapine has been measured in a variety of cell lines including peripheral blood mononuclear cells, monocyte-derived macrophages, and lymphoblastoid cell lines. In an assay using human embryonic kidney 293 cells, the median EC50 value (50% inhibitory concentration) of nevirapine was 90 nM against a panel of 2923 wild-type isolates of HIV-1 that were primarily (93%) clade B clinical isolates from the United States. The 99th percentile EC50 value was 470 nM in this trial. The median EC50 value was 63 nM (range 14-302 nM, n=29) against clinical isolates of HIV-1 clades A, B, C, D, F, G, and H, and circulating recombinant forms CRF01_AE, CRF02_AG and CRF12_BF. Nevirapine had no antiviral activity in cell culture against group O HIV-1 isolates (n=3) or HIV-2 isolates (n=3) replicating in cord blood mononuclear cells. Nevirapine in combination with efavirenz exhibited strong antagonistic anti-HIV-1 activity in cell culture and was additive to antagonistic with the protease inhibitor ritonavir or the fusion inhibitor enfuvirtide. Nevirapine exhibited additive to synergistic anti-HIV-1 activity in combination with the protease inhibitors amprenavir, atazanavir, indinavir, lopinavir, nelfinavir, saquinavir and tipranavir, and the NRTIs abacavir, didanosine, emtricitabine, lamivudine, stavudine, tenofovir and zidovudine. The anti-HIV-1 activity of nevirapine was antagonized by the anti-HBV drug adefovir and by the anti-HCV drug ribavirin in cell culture.
Resistance
HIV-1 isolates with reduced susceptibility (100- to 250-fold) to nevirapine emerge in cell culture. Genotypic analysis showed mutations in the HIV-1 RT gene encoding Y181C and/or V106A substitutions depending upon the virus strain and cell line employed. Time to emergence of nevirapine resistance in cell culture was not altered when selection included nevirapine in combination with several other NNRTIs.
Phenotypic and genotypic changes in HIV-1 isolates from treatment-naïve subjects receiving either nevirapine (n=24) or nevirapine and ZDV (n=14) were monitored in Phase 1 and 2 trials over 1 to ≥12 weeks. After 1 week of nevirapine monotherapy, isolates from 3/3 subjects had decreased susceptibility to nevirapine in cell culture. One or more of the RT mutations resulting in amino acid substitutions K103N, V106A, V108I, Y181C, Y188C, and G190A were detected in HIV-1 isolates from some subjects as early as 2 weeks after therapy initiation. By week eight of nevirapine monotherapy, 100% of the subjects tested (n=24) had HIV-1 isolates with a greater than 100-fold decrease in susceptibility to nevirapine in cell culture compared to baseline, and had one or more of the nevirapine-associated RT resistance substitutions. Nineteen of these subjects (80%) had isolates with Y181C substitutions regardless of dose.
Genotypic analysis of isolates from antiretroviral-naïve subjects experiencing virologic failure (n=71) receiving nevirapine once daily (n=25) or twice daily (n=46) in combination with lamivudine and stavudine (trial 2NN) for 48 weeks showed that isolates from 8/25 and 23/46 subjects, respectively, contained one or more of the following NNRTI resistance-associated substitutions: Y181C, K101E, G190A/S, K103N, V106A/M, V108I, Y188C/L, A98G, F227L, and M230L.
For trial 1100.1486, genotypic analysis was performed for baseline and on-therapy isolates from 23 and 34 virologic failure subjects in the VIRAMUNE XR and immediate-release VIRAMUNE treatment group, respectively. Nevirapine resistance-associated substitutions developed in the on-therapy isolates of 78% (18/23) of the virologic failures subjects in the VIRAMUNE XR treatment group and 88% (30/34) of the subjects in the immediate-release VIRAMUNE treatment group, respectively. The Y181C nevirapine resistance-associated substitution was found alone or in combination with other nevirapine resistance-associated substitutions (K101E, K103N, V106A, V108I, Y188 C/F/H/L/N, G190A, P225H, F227L, M230L) in isolates from 14 subjects failing VIRAMUNE XR treatment and 25 subjects failing immediate-release VIRAMUNE treatment. On-therapy isolates from 1 subject in VIRAMUNE XR treatment group developed a novel amino acid substitution Y181I and isolates from another subject in the immediate-release VIRAMUNE treatment group developed a novel amino acid substitution Y188N. Phenotypic analysis showed that Y188N and Y181I substitutions conferred 103- and 22-fold reductions in susceptibility to nevirapine, respectively.
Cross-resistance
Rapid emergence of HIV-1 strains which are cross-resistant to NNRTIs has been observed in cell culture. Nevirapine-resistant HIV-1 isolates were cross-resistant to the NNRTIs delavirdine and efavirenz. The Y188N conferred 22- and 7-fold reductions in susceptibility to delavirdine and efavirenz, respectively, but no decrease in susceptibility to etravirine. Similarly, the Y181I substitution reduced susceptibility to delavirdine and etravirine 3- and 8-fold, respectively, but did not reduce susceptibility to efavirenz. However, nevirapine-resistant isolates were susceptible to the NRTIs ddI and ZDV. Similarly, ZDV-resistant isolates were susceptible to nevirapine in cell culture.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity studies in mice and rats were carried out with nevirapine. Mice were dosed with 0, 50, 375 or 750 mg/kg/day for two years. Hepatocellular adenomas and carcinomas were increased at all doses in males and at the two high doses in females. In studies in which rats were administered nevirapine at doses of 0, 3.5, 17.5 or 35 mg/kg/day for two years, an increase in hepatocellular adenomas was seen in males at all doses and in females at the high dose. The systemic exposure (based on AUCs) at all doses in the two animal studies was lower than that measured in humans at the 200 mg twice daily dose of immediate-release VIRAMUNE. The mechanism of the carcinogenic potential is unknown. However, in genetic toxicology assays, nevirapine showed no evidence of mutagenic or clastogenic activity in a battery of in vitro and in vivo studies. These included microbial assays for gene mutation (Ames: Salmonella strains and E. coli), mammalian cell gene mutation assay (CHO/HGPRT), cytogenetic assays using a Chinese hamster ovary cell line and a mouse bone marrow micronucleus assay following oral administration. Given the lack of genotoxic activity of nevirapine, the relevance to humans of hepatocellular neoplasms in nevirapine-treated mice and rats is not known. In reproductive toxicology studies, evidence of impaired fertility was seen in female rats at doses providing systemic exposure, based on AUC, approximately equivalent to that provided with the recommended clinical dose.
14 CLINICAL STUDIES
14.1 Clinical Trials in Adults
The clinical efficacy of VIRAMUNE XR is based on 48-week data from an ongoing, randomized, double-blind, double-dummy Phase 3 trial (Trial 1100.1486, VERxVE) in treatment-naïve subjects and on 24-week data in an ongoing, randomized, open-label trial in subjects who switched from immediate-release VIRAMUNE tablets administered twice daily to VIRAMUNE XR tablets administered once daily (Trial 1100.1526, TRANxITION).
Treatment-naïve Subjects
Trial 1100.1486 (VERxVE) is a Phase 3 trial in which treatment-naïve subjects received immediate-release VIRAMUNE 200 mg once daily for 14 days and then were randomized to receive either immediate-release VIRAMUNE 200 mg twice daily or VIRAMUNE XR 400 mg once daily. All subjects received tenofovir + emtricitabine as background therapy. Randomization was stratified by screening HIV-1 RNA level (less than or equal to 100,000 copies/mL and greater than 100,000 copies/mL). Subject demographic and baseline disease characteristics were balanced between the two treatment groups. With respect to demographics: 85% of the subjects were male, 75% were white, 20% were black, and approximately 29% were from North America. With respect to baseline disease characteristics: mean viral load was 4.7 log10 copies/mL, mean CD4+ cell count was 228 cells/mm3 and 73% of subjects had clade B HIV-1 subtype. Approximately two-thirds of the subjects had a baseline HIV-RNA level of less than or equal to 100,000 copies/mL.
Table 5 describes week 48 outcomes in the Trial 1100.1486 (VERxVE). These outcomes include all subjects who were randomized after the 14 day lead-in with immediate-release VIRAMUNE and received at least one dose of blinded study medication.
| VIRAMUNE Immediate-Release N=506 | VIRAMUNE XR N=505 |
|
| #Includes subjects who changed optimized background therapy (OBT) to new class or changed OBT not permitted per protocol or due to lack of efficacy prior to Week 48, subjects who discontinued prior to Week 48 for lack or loss of efficacy and subjects with HIV RNA greater than or equal to 50 copies/mL in the Week 48 window. *Includes subjects who discontinued due to adverse events or death at any time point from Day 1 through the Week 48 window if this resulted in no virologic data on treatment during the specified window. **Other includes: withdrew consent, lost to follow-up, moved away, etc. |
||
| Virologic Success - HIV RNA < 50 copies/mL | 75% | 80% |
| Virologic Failure # | 13% | 11% |
| No Virologic Data at Week 48 Window Reasons Discontinued trial/study drug due to adverse event or death* Discontinued trial/study drug for other reasons** Missing data during window but on trial | 9% 3% <1% | 7% 2% |
At 48 weeks, mean change from baseline in CD4+ cell count adjusting for baseline HIV-1 viral load stratum was 191 cells/mm3 and 206 cells/mm3 for the groups receiving immediate-release VIRAMUNE and VIRAMUNE XR, respectively.
Subjects Switching from Immediate-release VIRAMUNE to VIRAMUNE XR
Trial 1100.1526 (TRANxITION) is a Phase 3 trial to evaluate safety and antiviral activity of switching from immediate-release VIRAMUNE to VIRAMUNE XR. In this open-label trial, 443 subjects already on an antiviral regimen containing immediate-release VIRAMUNE 200 mg twice daily with HIV-1 RNA less than 50 copies/mL were randomized in a 2:1 ratio to VIRAMUNE XR 400 mg once daily or immediate-release VIRAMUNE 200 mg twice daily. Approximately half of the subjects had tenofovir+emtricitabine as their background therapy, with the remaining subjects receiving abacavir sulfate+lamivudine or zidovudine+lamivudine. Approximately half of the subjects had at least 3 years of exposure to immediate-release VIRAMUNE prior to entering the trial.
At 24 weeks after randomization in Trial 1100.1526, 94% of subjects receiving immediate-release VIRAMUNE 200 mg twice daily and 95% of subjects receiving VIRAMUNE XR 400 mg once daily continued to have HIV-1 RNA less than 50 copies/mL.
16 HOW SUPPLIED/STORAGE AND HANDLING
VIRAMUNE XR tablets, 400 mg, are yellow, oval, biconvex tablets, debossed with "V04" on one side and the Boehringer Ingelheim logo on the other side.
VIRAMUNE XR tablets are supplied in bottles of 30 (NDC 0597-0123-30).
17 PATIENT COUNSELING INFORMATION
- "See FDA-approved patient labeling (Medication Guide)".
17.1 Hepatotoxicity and Skin Reactions
Inform patients of the possibility of severe liver disease or skin reactions associated with nevirapine that may result in death. Instruct patients developing signs or symptoms of liver disease or severe skin reactions to discontinue nevirapine and seek medical attention immediately, including performance of laboratory monitoring. Symptoms of liver disease include fatigue, malaise, anorexia, nausea, jaundice, acholic stools, liver tenderness or hepatomegaly. Symptoms of severe skin or hypersensitivity reactions include rash accompanied by fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial edema, and/or hepatitis.
Intensive clinical and laboratory monitoring, including liver enzymes, is essential during the first 18 weeks of therapy with nevirapine to detect potentially life-threatening hepatotoxicity and skin reactions. However, liver disease can occur after this period; therefore, monitoring should continue at frequent intervals throughout nevirapine treatment. Extra vigilance is warranted during the first 6 weeks of therapy, which is the period of greatest risk of hepatic events and skin reactions. Advise patients with signs and symptoms of hepatitis to discontinue nevirapine and seek medical evaluation immediately. If nevirapine is discontinued due to hepatotoxicity, do not restart it. Patients, particularly women, with increased CD4+ cell count at initiation of nevirapine therapy (greater than 250 cells/mm3 in women and greater than 400 cells/mm3 in men) are at substantially higher risk for development of symptomatic hepatic events, often associated with rash. Advise patients that co-infection with hepatitis B or C and/or increased transaminases at the start of therapy with nevirapine are associated with a greater risk of later symptomatic events (6 weeks or more after starting nevirapine) and asymptomatic increases in AST or ALT [see Boxed Warning and Warnings and Precautions (5.1)].
The majority of rashes associated with nevirapine occur within the first 6 weeks of initiation of therapy. Instruct patients that if any rash occurs during the two-week lead-in period with immediate-release VIRAMUNE, do not initiate VIRAMUNE XR until the rash resolves. The total duration of the lead-in dosing period with immediate-release VIRAMUNE should not exceed 28 days, at which point an alternative regimen may need to be started. Any patient experiencing a rash should have their liver enzymes (AST, ALT) evaluated immediately. Patients with severe rash or hypersensitivity reactions should discontinue nevirapine immediately and consult a physician. Nevirapine should not be restarted following severe skin rash or hypersensitivity reaction. Women tend to be at higher risk for development of nevirapine-associated rash. For patients who interrupt VIRAMUNE XR dosing for more than 7 days and for whom restarting nevirapine therapy is not contraindicated, restart the recommended lead-in dosing with immediate-release VIRAMUNE using one 200 mg tablet daily for the first 14 days [see Dose Adjustment (2.3)]. [see Boxed Warning and Warnings and Precautions (5.2)].
17.2 Administration
Inform patients to take VIRAMUNE XR every day as prescribed. Patients should not alter the dose without consulting their doctor. If a dose is missed, patients should take the next dose as soon as possible. However, if a dose is skipped, the patient should not double the next dose. Advise patients to report to their doctor the use of any other medications.
Instruct patients to swallow VIRAMUNE XR tablets whole. They must not be chewed, crushed, or divided.
Inform patients that it is not known whether VIRAMUNE XR therapy reduces the risk of transmission of HIV-1 to others through sexual contact. Effective treatment combined with safer sex practices may reduce the chance of passing HIV to others through sexual contact. Patients should be advised to continue to practice safer sex and to use latex or polyurethane condoms to lower the chance of sexual contact with any body fluids such as semen, vaginal secretions or blood. Patients should be advised never to re-use or share needles.
Inform patients that they may occasionally see soft remnants of VIRAMUNE XR in their stool. These occurrences have not been shown to affect drug levels or response.
VIRAMUNE XR is not a cure for HIV-1 infection; patients may continue to experience illnesses associated with advanced HIV-1 infection, including opportunistic infections. Advise patients to remain under the care of a physician when using VIRAMUNE XR.
17.3 Drug Interactions
VIRAMUNE XR may interact with some drugs; therefore, patients should be advised to report to their doctor the use of any other prescription, non-prescription medication or herbal products, particularly St. John's wort [see Warnings and Precautions (5.4) and Drug Interactions (7)].
17.4 Contraceptives
Hormonal methods of birth control, other than depomedroxy-progesterone acetate (DMPA), should not be used as the sole method of contraception in women taking VIRAMUNE XR, since VIRAMUNE XR may lower the plasma levels of these medications. Additionally, when oral contraceptives are used for hormonal regulation during VIRAMUNE XR therapy, the therapeutic effect of the hormonal therapy should be monitored [see Drug Interactions (7)].
17.5 Methadone
VIRAMUNE XR may decrease plasma concentrations of methadone by increasing its hepatic metabolism. Narcotic withdrawal syndrome has been reported in patients treated with nevirapine and methadone concomitantly. Monitor methadone-maintained patients beginning nevirapine therapy for evidence of withdrawal and adjust methadone dose accordingly [see Drug Interactions (7)].
17.6 Fat Redistribution
Inform patients that redistribution or accumulation of body fat may occur in patients receiving antiretroviral therapy and that the cause and long-term health effects of these conditions are not known at this time [see Warnings and Precautions (5.6)].
MEDICATION GUIDE
VIRAMUNE® (VIH-rah-mune)
(nevirapine)
Tablets
VIRAMUNE® (VIH-rah-mune)
(nevirapine)
Oral Suspension
VIRAMUNE® XR™ (VIH-rah-mune)
(nevirapine)
Extended-Release Tablets
Read this Medication Guide before you start taking VIRAMUNE and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or treatment.
What is the most important information I should know about VIRAMUNE?
-
VIRAMUNE can cause serious side effects. These include severe liver and skin problems that can cause death. These problems can happen at any time during treatment, but your risk is highest during the first 18 weeks of treatment.
- 1.
-
Severe liver problems: Anyone who takes VIRAMUNE may get severe liver problems. In some cases these liver problems can lead to liver failure and the need for a liver transplant, or death.
- People who have a higher CD4+ cell count when they begin VIRAMUNE treatment have a higher risk of liver problems, especially:
- Women with CD4+ counts higher than 250 cells/mm3. This group has the highest risk.
- Men with CD4+ counts higher than 400 cells/mm3.
- If you are a woman with CD4+ counts higher than 250 cells/mm3 or a man with CD4+ counts higher than 400 cells/mm3, you and your doctor will decide whether starting VIRAMUNE is right for you.
- In general, women have a higher risk of liver problems compared to men.
- People who have abnormal liver test results before starting VIRAMUNE treatment and people with hepatitis B or C also have a greater chance of getting liver problems.
- You may get a rash if you have liver problems.
-
Stop taking VIRAMUNE and call your doctor right away if you have any of the following symptoms of liver problems:
- dark (tea colored) urine
- yellowing of your skin or whites of your eyes
- light-colored bowel movements (stools)
- fever
- nausea (feeling sick to your stomach)
- feel unwell or like you have the flu
- pain or tenderness on your right side below your ribs
- tiredness
- loss of appetite
- Your doctor should see you and do blood tests often to check your liver function during the first 18 weeks of treatment with VIRAMUNE. You should continue to have your liver checked regularly during your treatment with VIRAMUNE. It is important for you to keep all of your doctor appointments.
- 2.
-
Severe rash and skin reactions: Skin rash is the most common side effect of VIRAMUNE. Most rashes happen in the first 6 weeks of taking VIRAMUNE. Rashes and skin reactions may be severe, life-threatening, and in some people, may lead to death. Stop using VIRAMUNE and call your doctor right away if you get a rash with any of the following symptoms:
- blisters
- mouth sores
- red or inflamed eyes, like "pink eye" (conjunctivitis)
- liver problems (see symptoms of liver problems above)
- swelling of your face
- fever
- feel unwell or like you have the flu
- tiredness
- muscle or joint aches
-
If your doctor tells you to stop treatment with VIRAMUNE because you have had any of the serious liver or skin problems described above, you should never take VIRAMUNE again.
- See the section "What are the possible side effects of VIRAMUNE?" for more information.
What is VIRAMUNE?
- VIRAMUNE is a prescription medicine used to treat Human Immunodeficiency Virus (HIV), the virus that causes AIDS (Acquired Immune Deficiency Syndrome).
- VIRAMUNE is a type of anti-HIV medicine called a "non-nucleoside reverse transcriptase inhibitor" (NNRTI). VIRAMUNE works by lowering the amount of HIV in your blood ("viral load"). You must take VIRAMUNE with other anti-HIV medicines. When you take VIRAMUNE with other anti-HIV medicines, VIRAMUNE can lower your viral load and increase the number of CD4+ cells ("T cells"). CD4+ cells are a type of immune helper cell in the blood. VIRAMUNE may not have these effects in every person.
- VIRAMUNE comes in 3 different forms. This Medication Guide provides information about all 3 forms of VIRAMUNE.
- VIRAMUNE tablets
- VIRAMUNE oral suspension
- VIRAMUNE XR extended release tablets
- VIRAMUNE XR extended-release tablets are not for use in children.
- VIRAMUNE does not cure HIV or AIDS, and it is not known if it will help you live longer with HIV. People taking VIRAMUNE may still get infections common in people with HIV (opportunistic infections). It is very important that you stay under the care of your doctor.
- It is not known if VIRAMUNE lowers the chance of passing HIV to other people. Effective treatment combined with safer sex practices, may reduce the chance of passing HIV to others through sexual contact. Always practice safer sex by using a latex or polyurethane condom or other barrier method to lower the chance of sexual contact with any body fluids such as semen, vaginal secretions, or blood. Never re-use or share needles. Take your HIV medicines as prescribed.
Who should not take VIRAMUNE?
- Tell your doctor if you have or have had liver problems. Your doctor may tell you not to take VIRAMUNE if you have certain liver problems.
- VIRAMUNE is only for people diagnosed with HIV. If you have not been diagnosed as HIV positive, then do not take VIRAMUNE.
What should I tell my doctor before taking VIRAMUNE?
-
Before you take VIRAMUNE, tell your doctor if you:
- have or have had hepatitis (inflammation of your liver) or problems with your liver. See "What is the most important information I should know about VIRAMUNE?" and "Who should not take VIRAMUNE?"
- receive dialysis
- have skin problems, such as a rash
- are pregnant or plan to become pregnant. It is not known if VIRAMUNE will harm your unborn baby.
Pregnancy Registry: There is a pregnancy registry for women who take antiviral medicines during pregnancy. The purpose of the registry is to collect information about the health of you and your baby. Talk to your doctor about how you can take part in this registry. - are breast-feeding or plan to breast-feed. VIRAMUNE can pass into your breast milk and may harm your baby. It is also recommended that HIV-positive women should not breast-feed their babies. Do not breast-feed during treatment with VIRAMUNE. Talk to your doctor about the best way to feed your baby.
-
Tell your doctor and pharmacist about all the medicines you take, including prescription and non-prescription medicines, vitamins and herbal supplements. VIRAMUNE may affect the way other medicines work, and other medicines may affect how VIRAMUNE works.
- You should not take VIRAMUNE if you also take:
- St. John's Wort. St. John's Wort can lower the amount of VIRAMUNE in your body.
- efavirenz (Sustiva®, Atripla®). Efavirenz may cause you to have an increased chance of side effects.
- atazanavir (Reyataz®)
- lopinavir and ritonavir (Kaletra®)
- fosamprenavir calcium (Lexiva®)
- itraconazole (Sporanox®)
- ketoconazole (Nizoral®)
- rifampin (Rifadin®, Rifamate®, Rifater®)
- Birth control pills. Birth control pills taken by mouth (oral contraceptives) and other hormone types of birth control may not work to prevent pregnancy. Talk with your doctor about other types of birth control that you can use to prevent pregnancy during treatment with VIRAMUNE.
- Also tell your doctor if you take:
- clarithromycin (Biaxin®)
- fluconazole (Diflucan®)
- indinavir sulfate (Crixivan®)
- methadone
- nelfinavir mesylate (Viracept®)
- rifabutin (Mycobutin®)
- warfarin (Coumadin®, Jantoven®)
- saquinavir mesylate (Invirase®)
-
If you are not sure if you take a medicine above, ask your doctor or pharmacist.
- Know the medicines you take. Keep a list of them to show your doctor or pharmacist when you get a new medicine.
How should I take VIRAMUNE?
- VIRAMUNE is always taken in combination with other anti-HIV medications.
- Take VIRAMUNE exactly as your doctor tells you to take it. Do not change your dose unless your doctor tells you to.
- You should never take more than one form of VIRAMUNE at the same time. Talk to your doctor if you have any questions.
- Do not crush or chew VIRAMUNE XR extended-release tablets.
- You may take VIRAMUNE with or without food.
- Do not miss a dose of VIRAMUNE, because this could make HIV harder to treat. If you miss a dose of VIRAMUNE, take the missed dose as soon as you remember. If it is almost time for your next dose, do not take the missed dose, just take the next dose at your regular time. Do not take two doses at the same time.
- If you stop taking VIRAMUNE for more than 7 days, ask your doctor how much to take before you start taking it again. You may need to begin taking the VIRAMUNE starting dose again, which is taken 1 time each day for 14 days.
Starting VIRAMUNE tablets:- Your doctor should start you with 1 dose each day to lower your chance of getting a serious rash. It is important that you only take 1 dose of VIRAMUNE each day for the first 14 days.
- Call your doctor right away if you get a skin rash during the first 14 days of VIRAMUNE treatment and do not increase your dose to 2 times a day.
- You should never take your starting dose for longer than 28 days. If after 28 days you are still receiving this starting dose because you have a rash, you and your doctor should talk about prescribing another HIV medicine for you instead of VIRAMUNE.
-
Do not increase your dose to 2 times a day if you have a rash.
- Day 15, you will take 1 VIRAMUNE tablet two times a day.
Starting VIRAMUNE XR extended-release tablets and this is the first time you are taking any form of VIRAMUNE:- Your doctor should start you with 1 dose of VIRAMUNE tablets each day to lower your chance of getting a serious rash. It is important that you only take 1 dose of VIRAMUNE each day for the first 14 days.
- Call your doctor right away if you get a skin rash during the first 14 days of VIRAMUNE treatment and do not increase your dose to 2 times a day.
- You should never take your starting dose for longer than 28 days. If after 28 days you are still receiving this starting dose because you have a rash, you and your doctor should talk about prescribing another HIV medicine for you instead of VIRAMUNE.
- Do not start VIRAMUNE XR extended-release tablets if you have a rash.
- Day 15, you will take 1 VIRAMUNE XR extended-release tablet each day.
Switching from VIRAMUNE tablets to VIRAMUNE XR extended-release tablets:- Take VIRAMUNE XR extended-release tablet 1 time a day.
If you take VIRAMUNE Oral Suspension: - Your doctor should start you with 1 dose each day to lower your chance of getting a serious rash. It is important that you only take 1 dose of VIRAMUNE each day for the first 14 days.
- If you or your child takes VIRAMUNE suspension (liquid), shake it gently before each use. Use an oral dosing syringe or dosing cup to measure the right dose. The oral dosing syringe and dosing cup are not provided with VIRAMUNE Suspension. Ask your pharmacist for a syringe or cup if you do not have one.
- After drinking the medicine, fill the dosing cup with water and drink it to make sure you get all the medicine.
- If the dose is less than 1 teaspoon (5 mL), use the syringe instead of the dosing cup.
What are the possible side effects of VIRAMUNE?
- VIRAMUNE may cause serious side effects, including:
- See "What is the most important information I should know about VIRAMUNE?"
- Changes in your immune system (Immune Reconstitution Syndrome) can happen when you start taking HIV medicines. Your immune system may get stronger and begin to fight infections that have been hidden in your body for a long time. Tell your doctor if you start having new symptoms after starting your HIV medicine.
- Changes in body fat can happen in some people who take antiretroviral therapy. These changes may include increased amount of fat in the upper back and neck ("buffalo hump"), breast, and around the middle of your body (trunk). Loss of fat from your legs, arms, and face can also happen. The cause and long-term health effects of these problems are not known at this time.
- See "What is the most important information I should know about VIRAMUNE?"
- The most common side effect of VIRAMUNE is rash.
- Tell your doctor if you have any side effect that bothers you or that does not go away.
- These are not all the possible side effects of VIRAMUNE. For more information, ask your doctor or pharmacist.
- Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store VIRAMUNE?
- Store VIRAMUNE at 59°F to 86°F (15°C to 30°C).
- Throw away VIRAMUNE that is no longer needed or out-of-date.
- Keep VIRAMUNE and all medicines out of the reach of children.
General information about VIRAMUNE.
- Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use VIRAMUNE for a condition for which it was not prescribed. Do not give VIRAMUNE to other people, even if they have the same condition you have. It may harm them.
- This Medication Guide summarizes the most important information about VIRAMUNE. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about VIRAMUNE that is written for health professionals.
For more information, go to www.viramune.com or www.viramunexr.com or call Boehringer Ingelheim Pharmaceuticals, Inc., at 1-800-542-6257, or (TTY) 1-800-459-9906.
What are the ingredients in VIRAMUNE?
- Active ingredient: nevirapine
Inactive ingredients:
VIRAMUNE Tablets: microcrystalline cellulose, lactose monohydrate, povidone, sodium starch glycolate, colloidal silicon dioxide, and magnesium stearate
VIRAMUNE Oral Suspension: carbomer 934P, methylparaben, propylparaben, sorbitol, sucrose, polysorbate 80, sodium hydroxide, and purified water
VIRAMUNE XR Tablets: lactose monohydrate, hypromellose, iron oxide, and magnesium stearate
This Medication Guide has been approved by the U.S. Food and Drug Administration
Distributed by:
Boehringer Ingelheim Pharmaceuticals, Inc.
Ridgefield, CT 06877 USA
Revised: November 2011
The brands listed are trademarks of their respective owners and are not trademarks of Boehringer Ingelheim Pharmaceuticals, Inc. The makers of these brands are not affiliated with and do not endorse Boehringer Ingelheim Pharmaceuticals, Inc., or its products.
Copyright 2011 Boehringer Ingelheim Pharmaceuticals, Inc.
ALL RIGHTS RESERVED
IT5243EK152011
10006580/03
| VIRAMUNE
nevirapine tablet, extended release |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA201152 | 04/11/2011 | |
| Labeler - Boehringer Ingelheim Pharmaceuticals Inc. (603175944) |
| Registrant - Boehringer Ingelheim Pharmaceuticals, Inc. (603175944) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Boehringer Ingelheim Roxane, Inc. | 058839929 | MANUFACTURE, ANALYSIS, LABEL, PACK | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Boehringer Ingelheim Roxane, Inc. | 002655556 | ANALYSIS | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Boehringer Ingelheim Chemicals, Inc. | 093561652 | API MANUFACTURE, ANALYSIS | |
Revised: 11/2011 Boehringer Ingelheim Pharmaceuticals Inc.