anzemet (dolasetron mesylate) injection
[sanofi-aventis U.S. LLC]
DESCRIPTION
ANZEMET (dolasetron mesylate) is an antinauseant and antiemetic agent. Chemically, dolasetron mesylate is (2α,6α,8α,9aβ)-octahydro-3-oxo-2,6-methano-2H-quinolizin-8-yl-1H-indole-3-carboxylate monomethanesulfonate, monohydrate. It is a highly specific and selective serotonin subtype 3 (5-HT3) receptor antagonist both in vitro and in vivo. Dolasetron mesylate has the following structural formula:
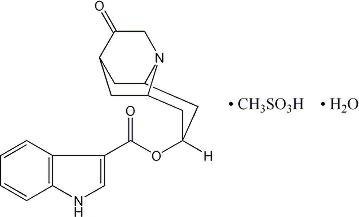
The empirical formula is C19H20N2O3• CH3SO3H • H2O, with a molecular weight of 438.50. Approximately 74% of dolasetron mesylate monohydrate is dolasetron base.
Dolasetron mesylate monohydrate is a white to off-white powder that is freely soluble in water and propylene glycol, slightly soluble in ethanol, and slightly soluble in normal saline.
ANZEMET Injection is a clear, colorless, nonpyrogenic, sterile solution for intravenous administration. Each milliliter of ANZEMET Injection contains 20 mg of dolasetron mesylate and 38.2 mg mannitol, USP, with an acetate buffer in water for injection. The pH of the resulting solution is 3.2 to 3.8.
ANZEMET Injection multidose vials contain a clear, colorless, nonpyrogenic, sterile solution for intravenous administration. Each ANZEMET multidose vial contains 25 mL (500 mg) dolasetron mesylate. Each milliliter contains 20 mg dolasetron mesylate, 29 mg mannitol, USP, and 5 mg phenol, USP, with an acetate buffer in water for injection. The pH of the resulting solution is 3.2 to 3.7.
CLINICAL PHARMACOLOGY
Dolasetron mesylate and its active metabolite, hydrodolasetron (MDL 74,156), are selective serotonin 5-HT3receptor antagonists not shown to have activity at other known serotonin receptors and with low affinity for dopamine receptors. The serotonin 5-HT3receptors are located on the nerve terminals of the vagus in the periphery and centrally in the chemoreceptor trigger zone of the area postrema. It is thought that chemotherapeutic agents produce nausea and vomiting by releasing serotonin from the enterochromaffin cells of the small intestine, and that the released serotonin then activates 5-HT3receptors located on vagal efferents to initiate the vomiting reflex.
Acute, usually reversible, ECG changes (PR and QTcprolongation; QRS widening), caused by dolasetron mesylate, have been observed in healthy volunteers and in controlled clinical trials. The active metabolites of dolasetron may block sodium channels, a property unrelated to its ability to block 5-HT3receptors. QTcprolongation is primarily due to QRS widening. Dolasetron appears to prolong both depolarization and, to a lesser extent, repolarization time. The magnitude and frequency of the ECG changes increased with dose (related to peak plasma concentrations of hydrodolasetron but not the parent compound). These ECG interval prolongations usually returned to baseline within 6 to 8 hours, but in some patients were present at 24 hour follow up. Dolasetron mesylate administration has little or no effect on blood pressure.
In healthy volunteers (N=64), dolasetron mesylate in single intravenous doses up to 5 mg/kg produced no effect on pupil size or meaningful changes in EEG tracings. Results from neuropsychiatric tests revealed that dolasetron mesylate did not alter mood or concentration. Multiple daily doses of dolasetron have had no effect on colonic transit in humans. Dolasetron mesylate has no effect on plasma prolactin concentrations.
Pharmacokinetics in Humans
Intravenous dolasetron mesylate is rapidly eliminated (t1/2<10 min) and completely metabolized to the most clinically relevant species, hydrodolasetron.
The reduction of dolasetron to hydrodolasetron is mediated by a ubiquitous enzyme, carbonyl reductase. Cytochrome P-450 (CYP)IID6 is primarily responsible for the subsequent hydroxylation of hydrodolasetron and both CYPIIIA and flavin monooxygenase are responsible for the N-oxidation of hydrodolasetron.
Hydrodolasetron is excreted in the urine unchanged (53.0% of administered intravenous dose). Other urinary metabolites include hydroxylated glucuronides and N-oxide.
Hydrodolasetron appeared rapidly in plasma, with a maximum concentration occurring approximately 0.6 hour after the end of intravenous treatment, and was eliminated with a mean half-life of 7.3 hours (%CV=24) and an apparent clearance of 9.4 mL/min/kg (%CV=28) in 24 adults. Hydrodolasetron is eliminated by multiple routes, including renal excretion and, after metabolism, mainly glucuronidation, and hydroxylation. Hydrodolasetron exhibits linear pharmacokinetics over the intravenous dose range of 50 to 200 mg and they are independent of infusion rate. Doses lower than 50 mg have not been studied. Two thirds of the administered dose is recovered in the urine and one third in the feces. Hydrodolasetron is widely distributed in the body with a mean apparent volume of distribution of 5.8 L/kg (%CV=25, N=24) in adults.
Sixty-nine to 77% of hydrodolasetron is bound to plasma protein. In a study with14C labeled dolasetron, the distribution of radioactivity to blood cells was not extensive. The binding of hydrodolasetron to α1-acid glycoprotein is approximately 50%. The pharmacokinetics of hydrodolasetron are linear and similar in men and women.
The pharmacokinetics of hydrodolasetron, in special and targeted patient populations following intravenous administration of ANZEMET Injection, are summarized in Table 1. The pharmacokinetics of hydrodolasetron are similar in adult (young and elderly) healthy volunteers and in adult cancer patients receiving chemotherapeutic agents. The apparent clearance of hydrodolasetron in pediatric and adolescent patients is 1.4 times to twofold higher than in adults. The apparent clearance of hydrodolasetron is not affected by age in adult cancer patients. Following intravenous administration, the apparent clearance of hydrodolasetron remains unchanged with severe hepatic impairment and decreases 47% with severe renal impairment. No dose adjustment is necessary for elderly patients (see PRECAUTIONS, Geriatric Use) or for patients with hepatic or renal impairment.
In a pharmacokinetic study in pediatric cancer patients (ages 3 to 11, N=25; ages 12 to 17, N=21) given a single 0.6, 1.2, 1.8, or 2.4 mg/kg dose of ANZEMET Injection intravenously, apparent clearance values were highest and half-lives were lowest in the youngest age group. For the 3 to 11 and the 12 to 17 year age groups, all receiving doses between 0.6 to 2.4 mg/kg, mean apparent clearances are 2 and 1.3 times greater, respectively, than for healthy adults receiving the same range of doses.
Thirty-two pediatric cancer patients ages 3 to 11 years (N=19) and 12 to 17 years (N=13), received 0.6, 1.2, or 1.8 mg/kg ANZEMET Injection diluted with either apple or apple-grape juice and administered orally. In this study, the mean apparent clearances were 3 times greater in the younger pediatric group and 1.8 times greater in the older pediatric group than those observed in healthy adult volunteers. Across this spectrum of pediatric patients, maximum plasma concentrations were 0.6 to 0.7 times those observed in healthy adults receiving similar doses.
In a pharmacokinetic study in 18 pediatric patients (2 to 11 years of age) undergoing surgery with general anesthesia and administered a single 1.2 mg/kg intravenous dose of ANZEMET Injection, mean apparent clearance was greater (40%) and terminal half-life shorter (36%) for hydrodolasetron than in healthy adults receiving the same dose.
For 12 pediatric patients, ages 2 to 12 years receiving 1.2 mg/kg ANZEMET Injection diluted in apple or apple-grape juice and administered orally, the mean apparent clearance was 34% greater and half-life was 21% shorter than in healthy adults receiving the same dose.
| Age (years) |
Dose | CLapp
(mL/min/kg) | t1/2
(h) | Cmax
(ng/mL) |
|
|---|---|---|---|---|---|
| CLapp: apparent clearance t1/2: terminal elimination half-life ( ): coefficient of variation in % | |||||
| Young Healthy Volunteers (N=24) | 19–40 | 100 mg | 9.4 (28%) | 7.3 (24%) | 320 (25%) |
| Elderly Healthy Volunteers (N=15) | 65–75 | 2.4 mg/kg | 8.3 (30%) | 6.9 (22%) | 620 (31%) |
| Cancer Patients | |||||
| Adults (N=273) | 19–87 | 0.6–3.0 mg/kg | 10.2 (34%)† | 7.5 (43%)† | 505 (26%)‡ |
| Adolescents (N=21) | 12–17 | 0.6–3.0 mg/kg | 12.5 (37%) | 5.5 (31%) | 562 (45%)§ |
| Children (N=25) | 3–11 | 0.6–2.4 mg/kg | 19.2 (30%) | 4.4 (24%) | 505 (100%)¶ |
| Pediatric Surgery Patients (N=18) | 2–11 | 1.2 mg/kg | 13.1 (47%) | 4.8 (23%) | 255 (22%) |
| Patients with Severe Renal Impairment (N=12) (Creatinine clearance ≤10 mL/min) | 28–74 | 200 mg | 5.0 (33%) | 10.9 (30%) | 867 (31%) |
| Patients with Severe Hepatic Impairment (N=3) | 42–52 | 150 mg | 9.6 (19%) | 11.7 (22%) | 396 (45%) |
Clinical Studies
Prevention of Cancer Chemotherapy-Induced Nausea and Vomiting
ANZEMET Injection administered intravenously at a dose of 1.8 mg/kg gave similar results in preventing nausea and vomiting as the other selective serotonin 5-HT3receptor antagonists studied as active comparators. It was more effective than metoclopramide. Efficacy was based on complete response rates (0 emetic episodes and no rescue medication).
Cisplatin Based Chemotherapy
A randomized, double-blind trial compared single intravenous doses of ANZEMET Injection with metoclopramide in 226 (160 men and 66 women) adult cancer patients receiving ≥80 mg/m2cisplatin. ANZEMET Injection at a dose of 1.8 mg/kg was significantly more effective than metoclopramide in the prevention of chemotherapy-induced nausea and vomiting in this study (Table 2).
| Number of Patients | ANZEMET Injection 1.8 mg/kg† |
Metoclopramide‡ |
p-value |
|---|---|---|---|
| 72 | 69 | ||
|
|||
| Response Over 24 Hours | |||
| Complete Response§ | 41 (57%) | 24 (35%) | 0.0009 |
| Nausea Score¶ | 4 | 30 | 0.0400 |
A second randomized, double-blind trial compared single intravenous doses of ANZEMET Injection with intravenous ondansetron in 609 (377 men and 232 women) adult cancer patients receiving ≥70 mg/m2cisplatin. A single intravenous 1.8 mg/kg dose of ANZEMET Injection was shown to be equivalent to a single intravenous 32 mg dose of ondansetron (Table 3).
| Number of Patients | ANZEMET Injection 1.8 mg/kg† | Ondansetron 32 mg‡ |
p-value |
|---|---|---|---|
| 198 | 206 | ||
|
|||
| Response Over 24 Hours | |||
| Complete Response§ | 88 (44%) | 88 (43%) | NS |
| Nausea Scorel¶ | 10 | 16 | NS |
Another randomized, double-blind trial compared single IV doses of ANZEMET with a single 3-mg IV dose of granisetron in 474 (315 men and 159 women) patients receiving ≥80 mg/m2cisplatin chemotherapy.
A single intravenous 1.8-mg/kg dose of ANZEMET gave similar results as those from granisetron.
Cyclophosphamide Based Chemotherapy
In a study of ANZEMET Injection in 309 patients (96 men and 213 women) receiving moderately emetogenic chemotherapy such as cyclophosphamide based regimens, a single intravenous 1.8 mg/kg dose of ANZEMET Injection was equivalent to metoclopramide administered as a 2 mg/kg intravenous bolus followed by 3 mg/kg intravenously over 8 hours. Complete response rates were 63% and 52%, respectively, p=0.12.
Prevention of Postoperative Nausea and Vomiting
ANZEMET Injection administered intravenously at a dose of 12.5 mg approximately 15 minutes before the cessation of general balanced anesthesia (short-acting barbiturate, nitrous oxide, narcotic and analgesic, and skeletal muscle relaxant) was significantly more effective than placebo in preventing postoperative nausea and vomiting. No increased efficacy was seen with higher doses.
One trial compared single intravenous ANZEMET Injection doses of 12.5, 25, 50, and 100 mg with placebo in 635 women surgical patients undergoing laparoscopic procedures. ANZEMET Injection at a dose of 12.5 mg was statistically superior to placebo for complete response (no vomiting, no rescue medication) (p=.0003). Complete response rates were 50% and 31%, respectively.
Another trial compared single intravenous ANZEMET Injection doses of 12.5, 25, 50, and 100 mg with placebo in 1030 (722 women and 308 men) surgical patients. In women, the 12.5 mg dose was statistically superior to placebo for complete response. The complete response rates were 50% and 40%, respectively. However, in men, there was no statistically significant difference in complete response between any ANZEMET dose and placebo.
Treatment of Postoperative Nausea and/or Vomiting
Two randomized, double-blinded trials compared single intravenous ANZEMET Injection doses of 12.5, 25, 50, and 100 mg with placebo in 124 male and 833 female patients who had undergone surgery with general balanced anesthesia and presented with early postoperative nausea or vomiting requiring antiemetic treatment.
In both studies, the 12.5 mg intravenous dose of ANZEMET was statistically superior to placebo for complete response (no vomiting, no escape medication). No significant increased efficacy was seen with higher doses.
INDICATIONS AND USAGE
ANZEMET Injection is indicated for the following:
- (1)
- the prevention of nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including high dose cisplatin;
- (2)
- the prevention of postoperative nausea and vomiting.As with other antiemetics, routine prophylaxis is not recommended for patients in whom there is little expectation that nausea and/or vomiting will occur postoperatively. In patients where nausea and/or vomiting must be avoided postoperatively, ANZEMET Injection is recommended even where the incidence of postoperative nausea and/or vomiting is low;
- (3)
- the treatment of postoperative nausea and/or vomiting.
CONTRAINDICATIONS
ANZEMET Injection is contraindicated in patients known to have hypersensitivity to the drug.
WARNINGS
ANZEMET can cause ECG interval changes (PR, QTc, JT prolongation and QRS widening). These changes are related in magnitude and frequency to blood levels of the active metabolite. These changes are self-limiting with declining blood levels. Some patients have interval prolongations for 24 hours or longer. Interval prolongation could lead to cardiovascular consequences, including heart block or cardiac arrhythmias. These have rarely been reported.
A cardiac conduction abnormality observed on an intra-operative cardiac rhythm monitor (interpreted as complete heart block) was reported in a 61-year-old woman who received 200 mg ANZEMET for the prevention of postoperative nausea and vomiting. This patient was also taking verapamil. A similar event also interpreted as complete heart block was reported in one patient receiving placebo.
A 66-year-old man with Stage IV non-Hodgkins lymphoma died suddenly 6 hours after receiving 1.8 mg/kg (119 mg) intravenous ANZEMET Injection. This patient had other potential risk factors including substantial exposure to doxorubicin and concomitant cyclophosphamide.
Pediatric Use
Dolasetron should be administered with caution in pediatric patients who have or may develop prolongation of cardiac conduction intervals, particularly QTc. Rare cases of sustained supraventricular and ventricular arrhythmias, cardiac arrest leading to death, and myocardial infarction have been reported in children and adolescents(See Precautions, General, and Adverse Reactions – Postmarketing Experience).
PRECAUTIONS
General
Dolasetron should be administered with caution in patients who have or may develop prolongation of cardiac conduction intervals, particularly QTc. These include patients with hypokalemia or hypomagnesemia, patients taking diuretics with potential for inducing electrolyte abnormalities, patients with congenital QT syndrome, patients taking anti-arrhythmic drugs or other drugs which lead to QT prolongation, and cumulative high dose anthracycline therapy.
Cross hypersensitivity reactions have been reported in patients who received other selective 5-HT3receptor antagonists. These reactions have not been seen with dolasetron mesylate.
Drug Interactions
The potential for clinically significant drug-drug interactions posed by dolasetron and hydrodolasetron appears to be low for drugs commonly used in chemotherapy or surgery, because hydrodolasetron is eliminated by multiple routes. See PRECAUTIONS, Generalfor information about potential interaction with other drugs that prolong the QTcinterval. Blood levels of hydrodolasetron increased 24% when dolasetron was coadministered with cimetidine (nonselective inhibitor of cytochrome P-450) for 7 days, and decreased 28% with coadministration of rifampin (potent inducer of cytochrome P-450) for 7 days. ANZEMET Injection has been safely coadministered with drugs used in chemotherapy and surgery. As with other agents which prolong ECG intervals, caution should be exercised in patients taking drugs which prolong ECG intervals, particularly QTc.
In patients taking furosemide, nifedipine, diltiazem, ACE inhibitors, verapamil, glyburide, propranolol, and various chemotherapy agents, no effect was shown on the clearance of hydrodolasetron. Clearance of hydrodolasetron decreased by about 27% when dolasetron mesylate was administered intravenously concomitantly with atenolol. ANZEMET did not influence anesthesia recovery time in patients. Dolasetron mesylate did not inhibit the antitumor activity of four chemotherapeutic agents (cisplatin, 5-fluorouracil, doxorubicin, cyclophosphamide) in four murine models.
Carcinogenesis, Mutagenesis, Impairment Of Fertility
In a 24-month carcinogenicity study, there was a statistically significant (P<0.001) increase in the incidence of combined hepatocellular adenomas and carcinomas in male mice treated with 150 mg/kg/day and above. In this study, mice (CD-1) were treated orally with dolasetron mesylate 75, 150 or 300 mg/kg/day (225, 450 or 900 mg/m2/day). For a 50 kg person of average height (1.46 m2body surface area), these doses represent 3.4, 6.8 and 13.5 times the recommended clinical dose (66.6 mg/m2, intravenous) on a body surface area basis. No increase in liver tumors was observed at a dose of 75 mg/kg/day in male mice and at doses up to 300 mg/kg/day in female mice.
In a 24-month rat (Sprague-Dawley) carcinogenicity study, oral dolasetron mesylate was not tumorigenic at doses up to 150 mg/kg/day (900 mg/m2/day, 13.5 times the recommended human dose based on body surface area) in male rats and 300 mg/kg/day (1800 mg/m2/day, 27 times the recommended human dose based on body surface area) in female rats.
Dolasetron mesylate was not genotoxic in the Ames test, the rat lymphocyte chromosomal aberration test, the Chinese hamster ovary (CHO) cell (HGPRT) forward mutation test, the rat hepatocyte unscheduled DNA synthesis (UDS) test or the mouse micronucleus test.
Dolasetron mesylate was found to have no effect on fertility and reproductive performance at oral doses up to 100 mg/kg/day (600 mg/m2/day, 9 times the recommended human dose based on body surface area) in female rats and up to 400 mg/kg/day (2400 mg/m2/day, 36 times the recommended human dose based on body surface area) in male rats.
Pregnancy
Teratogenic Effects
Pregnancy Category B
Teratology studies have not revealed evidence of impaired fertility or harm to the fetus due to dolasetron mesylate. These studies have been performed in pregnant rats at intravenous doses up to 60 mg/kg/day (5.4 times the recommended human dose based on body surface area) and pregnant rabbits at intravenous doses up to 20 mg/kg/day (3.2 times the recommended human dose based on body surface area). There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nursing Mothers
It is not known whether dolasetron mesylate is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when ANZEMET Injection is administered to a nursing woman.
Pediatric Use
Dolasetron should be administered with caution in pediatric patients who have or may develop prolongation of cardiac conduction intervals, particularly QTc. Rare cases of sustained supraventricular and ventricular arrhythmias, cardiac arrest leading to death, and myocardial infarction have been reported in children and adolescents(See Warningsand Adverse Reactions – Postmarketing Experience).
Geriatric Use
Prevention of cancer chemotherapy-induced nausea and vomiting (CINV)
In controlled clinical trials in the prevention of chemotherapy-induced nausea and vomiting, 723 (32%) of 2264 patients were 65 years of age or older. Of the 723 geriatric patients in the trial, 563 received intravenous ANZEMET Injection. No overall differences in safety or effectiveness were observed between geriatric and younger patients, and other reported clinical experience has not identified differences in responses between geriatric and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Prevention and treatment of post-operative nausea and vomiting (PONV)
Controlled clinical studies in the prevention and treatment of post-operative nausea and vomiting did not include sufficient numbers of patients aged 65 years or older – only 57 (2%) geriatric patients (43 received intravenous ANZEMET Injection) out of 3289 total patients participated in the controlled PONV trials – to determine whether they respond differently from younger patients. Other reported clinical experiences have not identified differences in responses between geriatric and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
The pharmacokinetics, including clearance of intravenous ANZEMET Injection, in elderly and younger patients are similar (see CLINICAL PHARMACOLOGY, Pharmacokinetics in Humans). Dosage adjustment is not needed in patients over the age of 65.
ADVERSE REACTIONS
Chemotherapy Patients
In controlled clinical trials, 2265 adult patients received ANZEMET Injection. The overall adverse event rates were similar with 1.8 mg/kg ANZEMET Injection and ondansetron or granisetron. Patients were receiving concurrent chemotherapy, predominantly high-dose (≥50 mg/m2) cisplatin. Following is a combined listing of all adverse events reported in ≥2% of patients in these controlled trials (Table 4).
|
Event | ANZEMET Injection 1.8 mg/kg (n=695) | Ondansetron/ Granisetron* (n=356) |
||
|---|---|---|---|---|
|
||||
| Headache | 169 | (24.3%) | 73 | (20.5%) |
| Diarrhea | 89 | (12.4%) | 25 | (7.0%) |
| Fever | 30 | (4.3%) | 18 | (5.1%) |
| Fatigue | 25 | (3.6%) | 12 | (3.4%) |
| Hepatic Function Abnormal† | 25 | (3.6%) | 12 | (3.4%) |
| Abdominal Pain | 22 | (3.2%) | 7 | (2.0%) |
| Hypertension | 20 | (2.9%) | 9 | (2.5%) |
| Pain | 17 | (2.4%) | 7 | (2.0%) |
| Dizziness | 15 | (2.2%) | 7 | (2.0%) |
| Chills/Shivering | 14 | (2.0%) | 6 | (1.7%) |
Postoperative Patients
In controlled clinical trials with 2550 adult patients, headache and dizziness were reported more frequently with 12.5 mg ANZEMET Injection than with placebo. Rates of other adverse events were similar. Following is a listing of all adverse events reported in ≥2% of patients receiving either placebo or 12.5 mg ANZEMET Injection for the prevention or treatment of postoperative nausea and vomiting in controlled clinical trials (Table 5).
|
Event | ANZEMET Injection 12.5 mg (n=615) | Placebo (n=739) |
|---|---|---|
| Headache | 58 (9.4%) | 51 (6.9%) |
| Dizziness | 34 (5.5%) | 23 (3.1%) |
| Drowsiness | 15 (2.4%) | 18 (2.4%) |
| Pain | 15 (2.4%) | 21 (2.8%) |
| Urinary Retention | 12 (2.0%) | 16 (2.2%) |
In clinical trials, the following infrequently reported adverse events, assessed by investigators as treatment-related or causality unknown, occurred following oral or intravenous administration of ANZEMET to adult patients receiving concomitant cancer chemotherapy or surgery:
Cardiovascular:Hypotension; rarely–edema, peripheral edema. The following events also occurred rarely and with a similar frequency as placebo and/or active comparator: Mobitz I AV block, chest pain, orthostatic hypotension, myocardial ischemia, syncope, severe bradycardia, and palpitations. See PRECAUTIONSsection for information on potential effects on ECG.
In addition, the following asymptomatic treatment-emergent ECG changes were seen at rates less than or equal to those for active or placebo controls: bradycardia, tachycardia, T wave change, ST-T wave change, sinus arrhythmia, extrasystole (APCs or VPCs), poor R-wave progression, bundle branch block (left and right), nodal arrhythmia, U wave change, atrial flutter/fibrillation.
Furthermore, severe hypotension, bradycardia and syncope have been reported immediately or closely following IV administration.
Dermatologic:Rash, increased sweating.
Gastrointestinal System:Constipation, dyspepsia, abdominal pain, anorexia; rarely–pancreatitis.
Hearing, Taste and Vision:Taste perversion, abnormal vision; rarely–tinnitus, photophobia.
Hematologic:Rarely–hematuria, epistaxis, prothrombin time prolonged, PTT increased, anemia, purpura/hematoma, thrombocytopenia.
Hypersensitivity:Rarely–anaphylactic reaction, facial edema, urticaria.
Liver and Biliary System:Transient increases in AST (SGOT) and/or ALT (SGPT) values have been reported as adverse events in less than 1% of adult patients receiving ANZEMET in clinical trials. The increases did not appear to be related to dose or duration of therapy and were not associated with symptomatic hepatic disease. Similar increases were seen with patients receiving active comparator. Rarely–hyperbilirubinemia, increased GGT.
Metabolic and Nutritional:Rarely–alkaline phosphatase increased.
Musculoskeletal:Rarely–myalgia, arthralgia.
Nervous System:Flushing, vertigo, paraesthesia, tremor; rarely–ataxia, twitching.
Psychiatric:Agitation, sleep disorder, depersonalization; ; rarely–confusion, anxiety, abnormal dreaming.
Respiratory System:Rarely–dyspnea, bronchospasm.
Urinary System:Rarely–dyspnea, polyuria, acute renal failure.
Vascular (Extracardiac):Local pain or burning on IV administration; rarely–peripheral ischemia, thrombophlebitis/phlebitis.
Postmarketing Experience
Rare cases of sustatined supraventricular and ventricular arrhythmias, cardiac arrest leading to death, and myocardial infarction have been reported in children and adolescents.
OVERDOSAGE
A 59-year-old man with metastatic melanoma and no known pre-existing cardiac conditions developed severe hypotension and dizziness 40 minutes after receiving a 15 minute intravenous infusion of 1000 mg (13 mg/kg) of dolasetron mesylate. Treatment for the overdose consisted of infusion of 500 mL of a plasma expander, dopamine, and atropine. The patient had normal sinus rhythm and prolongation of PR, QRS and QTcintervals on an ECG recorded 2 hours after the infusion. The patient's blood pressure was normal 3 hours after the event and the ECG intervals returned to baseline on follow-up. The patient was released from the hospital 6 hours after the event.
Following a suspected overdose of ANZEMET Injection, a patient found to have second-degree or higher
AV conduction block with ECG should undergo cardiac telemetry monitoring.
There is no known specific antidote for dolasetron mesylate, and patients with suspected overdose should be managed with supportive therapy. Individual doses as large as 5 mg/kg intravenously or 400 mg orally have been safely given to healthy volunteers or cancer patients.
It is not known if dolasetron mesylate is removed by hemodialysis or peritoneal dialysis.
A 7-year-old boy received 6 mg/kg dolasetron mesylate orally before surgery. No symptoms occurred and no treatment was required.
Single intravenous doses of dolasetron mesylate at 160 mg/kg in male mice and 140 mg/kg in female mice and rats of both sexes (6.3 to 12.6 times the recommended human dose based on body surface area) were lethal. Symptoms of acute toxicity were tremors, depression and convulsions.
DOSAGE AND ADMINISTRATION
The recommended dose of ANZEMET Injection should not be exceeded.
Prevention of Cancer Chemotherapy-Induced Nausea and Vomiting
Adults
The recommended intravenous dosage of ANZEMET Injection from clinical trial results is 1.8 mg/kg given as a single dose approximately 30 minutes before chemotherapy (see Administration). Alternatively, for most patients, a fixed dose of 100 mg can be administered over 30 seconds.
Pediatric Patients
The recommended intravenous dosage in pediatric patients 2 to 16 years of age is 1.8 mg/kg given as a single dose approximately 30 minutes before chemotherapy, up to a maximum of 100 mg (see Administration). Safety and effectiveness in pediatric patients under 2 years of age have not been established.
ANZEMET Injection mixed in apple or apple-grape juice may be used for oral dosing of pediatric patients. When ANZEMET Injection is administered orally, the recommended dosage in pediatric patients 2 to 16 years of age is 1.8 mg/kg up to a maximum 100 mg dose given within 1 hour before chemotherapy.
The diluted product may be kept up to 2 hours at room temperature before use.
Use in the Elderly, in Renal Failure Patients, or in Hepatically Impaired Patients
No dosage adjustment is recommended.
Prevention or Treatment of Postoperative Nausea and/or Vomiting
Adults
The recommended intravenous dosage of ANZEMET Injection is 12.5 mg given as a single dose approximately 15 minutes before the cessation of anesthesia (prevention) or as soon as nausea or vomiting presents (treatment).
Pediatric Patients
The recommended intravenous dosage in pediatric patients 2 to 16 years of age is 0.35 mg/kg, with a maximum dose of 12.5 mg, given as a single dose approximately 15 minutes before the cessation of anesthesia or as soon as nausea or vomiting presents. Safety and effectiveness in pediatric patients under 2 years of age have not been established.
ANZEMET Injection mixed in apple or apple-grape juice may be used for oral dosing of pediatric patients. When ANZEMET Injection is administered orally, the recommended oral dosage in pediatric patients 2 to 16 years of age is 1.2 mg/kg up to a maximum 100-mg dose given within 2 hours before surgery. The diluted product may be kept up to 2 hours at room temperature before use.
Use in the Elderly, in Renal Failure Patients, or in Hepatically Impaired Patients
No dosage adjustment is recommended.
ADMINISTRATION
ANZEMET Injection can be safely infused intravenously as rapidly as 100 mg/30 seconds or diluted in a compatible intravenous solution (see below) to 50 mL and infused over a period of up to 15 minutes. ANZEMET Injection should not be mixed with other drugs. Flush the infusion line before and after administration of ANZEMET Injection.
STABILITY
After dilution, ANZEMET Injection is stable under normal lighting conditions at room temperature for 24 hours or under refrigeration for 48 hours with the following compatible intravenous fluids: 0.9% sodium chloride injection, 5% dextrose injection, 5% dextrose and 0.45% sodium chloride injection, 5% dextrose and Lactated Ringer's injection, Lactated Ringer's injection, and 10% mannitol injection. Although ANZEMET Injection is chemically and physically stable when diluted as recommended, sterile precautions should be observed because diluents generally do not contain preservative. After dilution, do not use beyond 24 hours, or 48 hours if refrigerated.
Parenteral drug products should be inspected visually for particulate matter and discoloration before administration whenever solution and container permit.
HOW SUPPLIED
ANZEMET Injection (dolasetron mesylate injection) is supplied as a clear, colorless solution in single and multidose vials, and Carpuject® sterile cartridges with Luer Lock.
| ANZEMET®Injection (dolasetron mesylate injection) 20 mg/mL |
||
|---|---|---|
| Strength | Description | NDC Number |
| 12.5 mg | 0.625mL single use vial*(Box of 6) | 0088-1208-06 |
| 12.5 mg | 0.625mL fill in single-use 2mL Carpuject with Luer Lock†(Box of 10) | 0088-1208-76 |
| 100 mg/5 mL | 5mL single-use vial* | 0088-1206-32 |
| 500 mg/25 mL | 25 mL multidose vial* | 0088-1209-26 |
Store at 20–25 C (68–77 F) with excursions permitted to 15–30 C (59–86 F) [See USP Controlled Room Temperature]. Protect from light.
Prescribing information as of October 2007
Mfd. for: sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
Carpuject is a registered trademark of Hospira Inc.
©2007 sanofi-aventis U.S. LLC
| Anzemet (dolasetron mesylate) | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Anzemet (dolasetron mesylate) | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Anzemet (dolasetron mesylate) | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
Revised: 01/2008sanofi-aventis U.S. LLC