B CLEAN HAND SANITIZER- ethyl alcohol gel
AGE DO BRASIL INDUSTRIA E COMERCIO LTDA.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
B Clean - Hand Sanitizer 440g
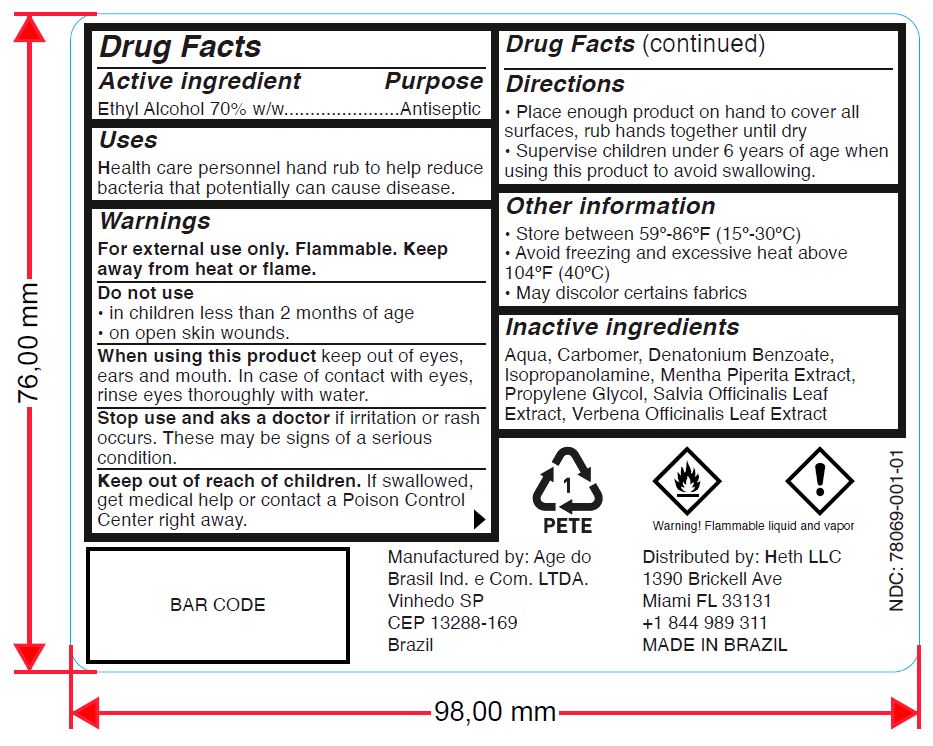
Warnings
For external use only. Flammable. Keep away from heat or flame.
Directions
- Place enough product on hands to cover all surfaces. Rub hands together until dry.
- Supervise children under 6 years of age when using this product to avoid swallowing.
Other information
- Store between 59º-86ºF (15º-30ºC)
- Avoid freezing and excessive heat above 104ºF (40ºC)
- May discolor certains fabrics
Inactive Ingredients
Aqua, Carbomer, Denatonium Benzoate, Isopropanolamine, Mentha Piperita Extract, Propylene Glycol, Salvia Officinalis Leaf Extract, Verbena Officinalis Leaf Extract
Package Label - Principal Display Panel
HAND SANITIZER
ALCOHOL-BASED GEL
moisturizing
Net wt. 15.52 oz (440g)

Manufactured by: Age do
Brasil Ind. e Com. LTDA.
Vinhedo SP
CEP 13288-169
Brazil
Distributed by: Heth LLC
1390 Brickell Ave
Miami FL 33131
+1 844 989 311
MADE IN BRAZIL
Warning! Flammable liquid and vapor

| B CLEAN HAND SANITIZER
ethyl alcohol gel |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - AGE DO BRASIL INDUSTRIA E COMERCIO LTDA. (920307841) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| AGE DO BRASIL INDUSTRIA E COMERCIO LTDA. | 920307841 | manufacture(78069-001) | |