CHLORPHENIRAMINE AND CODEINE
-
codeine phosphate and
chlorpheniramine maleate liquid
Breckenridge Pharmaceutical, Inc.
----------
Chlorpheniramine andCodeine Liquid
Rx Only
Antihistamine / Antitussive
Sugar free / Alcohol free / Dye free
DESCRIPTION
Chlorpheniramine and Codeine Liquid is a grape-flavored antihistamine/antitussive liquid for oral administration.
|
|
| Each 5 mL (1 teaspoonful) contains: | |
| Codeine Phosphate* | 10 mg |
| Chlorpheniramine Maleate | 2 mg |
Codeine Phosphate is an alkaloid, obtained from opium or prepared from morphine by methylation. Codeine Phosphate occurs as fine, white, needle-shaped crystals, or white, crystalline powder. It is affected by light. Its chemical name is: Morphinan-6-ol, 7,8-didehydroxy-4,5-epoxy-3-methoxy-17-methyl-, (5α,6α)-, phosphate (1:1) (salt), hemihydrate, and the following chemical structure:
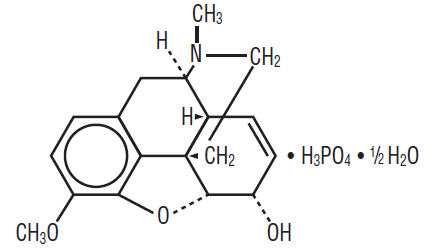
C18H21NO3 • H3PO4 • ½ H2O M.W. 406.37
Chlorpheniramine Maleate is an antihistamine having the chemical name: 2-Pyridinepropanamine, γ-(4-chlorophenyl)-N,N-dimethyl-, (Z)-2-butenedioate (1:1). Its chemical structure is as follows:
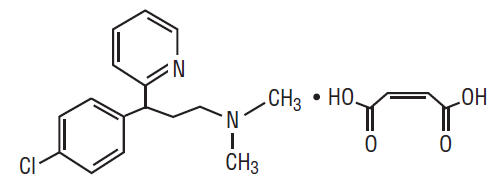
C16H19ClN2 • C4H4O4 M.W. 390.86
Inactive Ingredients: Methylparaben, Propylparaben, Benzoic Acid, Citric Acid, Sodium Saccharin, Grape Flavor, Natural Flavor, Propylene Glycol, Glycerin, Sorbitol, Purified Water.
CLINICAL PHARMACOLOGY
Codeine Phosphate
Codeine is a centrally acting analgesic and antitussive which is well absorbed orally. Following absorption, codeine is metabolized by the liver and metabolic products are excreted in the urine.
Chlorpheniramine Maleate
Chlorpheniramine is an alkylamine type antihistamine. This group of antihistamines is among the most active histamine antagonists and is generally effective in relatively low doses. The drugs are not so prone to produce drowsiness and are therefore among the most suitable agents for daytime use, but a significant proportion of patients do experience this effect.
INDICATIONS AND USAGE
Temporarily relieves runny nose, sneezing, itching of the nose or throat, and itchy, watery eyes due to hay fever or other upper respiratory allergies. Temporarily relieves cough due to minor bronchial irritations, the common cold or inhaled irritants.
CONTRAINDICATIONS
Contraindicated in patients with a known hypersensitivity or idiosyncrasy to any of its ingredients. Do not use in newborn infants, premature infant, in nursing mothers, in patients with severe hypertension, severe coronary artery disease, ischemic heart disease. This product should not be used during pregnancy or in nursing mothers.
Do not use this product if you are now taking a prescription monoamine oxidase inhibitor (MAOI), certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease, or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Antihistamines are contraindicated in patients with narrow-angle glaucoma, urinary retention, peptic ulcer, and during an asthma attack. Antihistamines should not be used to treat lower respiratory tract conditions, including asthma.
Do not take this product if you are taking sedatives or tranquilizers, without first consulting your doctor.
WARNINGS
Do not exceed recommended dosage.
May cause excitability especially in children.
Do not take this product, unless directed by a doctor, if you have a breathing problem such as emphysema or chronic bronchitis, or if you have glaucoma or difficulty in urination due to enlargement of the prostate gland. May cause drowsiness; alcohol, sedatives, and tranquilizers may increase the drowsiness effect. Avoid alcoholic beverages while taking this product. Do not take this product if you are taking sedatives or tranquilizers, without first consulting your doctor. Use caution when driving a motor vehicle or operating machinery. Do not give this product to children under 12 years of age, or children who are taking sedatives, or tranquilizers, without first consulting the child's doctor.
A persistent cough may be a sign of a serious condition. If cough persists for more than 1 week, tends to recur, or is accompanied by fever, rash, or persistent headache, consult a doctor. Do not take this product for persistent or chronic cough such as occurs with smoking, asthma, or emphysema, or if cough is accompanied by excessive phlegm (mucus) unless directed by a doctor. Adults and children who have a chronic pulmonary disease or shortness of breath, or children (over 12 years of age) who are taking other drugs, should not take this product unless directed by a doctor. May cause or aggravate constipation.
Use caution when giving to children (see PRECAUTIONS, Pediatric Use), or patients with chronic pulmonary disease, shortness of breath, difficulty in breathing, asthma, emphysema, high blood pressure, heart disease, diabetes, thyroid disease, or difficulty in urination due to the enlargement of the prostate gland unless directed by a physician.
Antihistamines may cause hyperexcitability, especially in children. At doses higher than the recommended dose, nervousness, dizziness, or sleeplessness may occur. Especially in infants and small children, antihistamines in overdosage may cause hallucinations, convulsions, and death.
If a hypertensive crisis occurs, these drugs should be discontinued immediately and therapy to lower blood pressure should be instituted immediately. Fever should be managed by means of external cooling.
Do not take this product if you are pregnant or nursing.
PRECAUTIONS
General
Before prescribing medication to suppress or modify a cough, it is important to ascertain that the underlying cause of the cough is identified, that modification of the cough does not increase the risk of clinical or physiologic complications, and that appropriate therapy for the primary disease is provided. Because of its sympathomimetic component, this product should be used with caution in patients with diabetes, hypertension, heart disease, or thyroid disease.
Information for Patients
Patients should be warned about engaging in activities requiring mental alertness and motor coordination, such as driving a car or operating machinery. Patients should be cautioned to get up slowly from a lying or sitting position and to lie down if nausea occurs.
Drug Interactions
Patients receiving other narcotic analgesics, anti-psychotics, anti-anxiety agents, or other CNS depressants (including alcohol) concomitantly with this drug may exhibit and additive CNS depression. When such combined therapy is contemplated, the dose of one or both agents should be reduced. The concurrent use of anticholinergics with codeine may produce paralytic ileus. MAO inhibitors (or for 14 days after stopping MAOI therapy) prolong and intensify the anticholinergic effects of antihistamines. Antihistamines may have additive effects with alcohol and other CNS depressants, e.g., hypnotics, sedatives, tranquilizers, antianxiety agents.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal studies of this product to assess the carcinogenic and mutagenic potential on the effect on fertility have not been performed.
Usage in Pregnancy
Animal reproduction studies have not been conducted with this product. It is also not know whether this product can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. This product should not be given to a pregnant woman.
Nursing Mothers
This product is contraindicated in nursing mothers (see CONTRAINDICATIONS).
Codeine Warning
When physicians prescribe codeine-containing drugs to nursing women, they should inform their patients about the potential risks and the signs of morphine overdose. Nursing women taking codeine need to carefully watch their infants for signs of morphine overdose and seek medical attention immediately if the infant develops increased sleepiness (more than usual), difficulty breastfeeding or breathing, or decreased tone (limpness). Nursing mothers may also experience overdose symptoms such as extreme sleepiness, confusion, shallow breathing or severe constipation. When prescribing codeine to nursing mothers, physicians should choose the lowest effective dose the shortest period of time and should closely monitor mother-infant pairs.
Drug metabolism is a complex process involving multiple genetic, environmental and physiologic factors. Limited evidence suggests that individuals who are ultra-rapid metabolizers (those with a specific CYP2D6 genotype) may convert codeine to its active metabolite, morphine, more rapidly and completely than other people. In nursing mothers, this metabolism can result in higher than expected serum and breast milk morphine levels. One published case report of an infant death raises concern that nursing babies may be at increased risk of morphine overdose if their mothers are taking codeine and are ultra-rapid metabolizers of the drug.
Pediatric Use
Safety and effectiveness in the pediatric population, under 12, have not been established.
Geriatric Use
The elderly (60 years of age or older) are more likely to exhibit adverse reactions. Caution should be taken when prescribing this drug to elderly patients.
ADVERSE REACTIONS
The most frequent adverse reactions to this product include: sedation, dryness of mouth, nose and throat, thickening of bronchial secretions, and dizziness. Other adverse reactions may include:
Cardiovascular System: Hypotension, headache, palpitations, tachycardia, extra systoles.
Dermatologic: Urticaria, drug rash, photosensitivity, and pruritus.
Central Nervous System: Sedation, sleepiness, dizziness, disturbed coordination, fatigue, confusion, restlessness, excitation, nervousness, tremor, irritability, insomnia, euphoria, paresthesias, blurred vision, diplopia, vertigo, tinnitus, acute labyrinthitis, hysteria, neuritis, convulsions.
Gastrointestinal: Epigastric distress, anorexia, nausea, vomiting, diarrhea, constipation.
Genitourinary: Urinary frequency, difficult urination, urinary retention, early menses.
Respiratory: Thickening of bronchial secretions, tightness of chest and wheezing, nasal stuffiness.
Hematologic System: Hemolytic anemia, thrombocytopenia, agranulocytosis.
OVERDOSAGE
Codeine
Signs and Symptoms
Overdosage with codeine is characterized by respiratory depression (a decrease in respiratory rate and / or tidal volume, Cheyne-Stokes respiration, cyanosis), pinpoint pupils, extreme somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, and sometimes bradycardia and hypotension. In severe overdosage, particularly by the intravenous route, apnea, circulatory collapse, cardiac arrest and death may occur.
Treatment
Induce emesis if patient is alert and is seen prior to 6 hours following ingestion. Precautions against aspiration must be taken, especially in infants and small children. Gastric lavage may be carried out, although in some instances tracheotomy may be necessary prior to lavage. CNS stimulants may counter CNS depression. Should CNS hyperactivity or convulsive seizures occur, intravenous short-acting barbiturates may be indicated. Hypertensive responses and /or tachycardia should be treated appropriately. Oxygen, intravenous fluids, and other supportive measures should be employed as indicated.
Chlorpheniramine Maleate
Signs and Symptoms
Antihistamine overdosage may vary from central nervous system depression (sedation, apnea, and cardiovascular collapse) to stimulation (insomnia, hallucinations, tremors or convulsions). Other signs and symptoms may be dizziness, tinnitus, ataxia, blurred vision, and hypotension. Stimulation and atropine-like signs and symptoms (dry mouth; fixed, dilated pupils; flushing; hyperthermia, and gastrointestinal symptoms) are particularly likely in children.
Treatment
Emergency treatment should be started immediately. Vomiting should be induced, even if it has occurred spontaneously. Vomiting by the administration of ipecac syrup is preferred. Vomiting should not be induced in patients with impaired consciousness. The action of ipecac is facilitated by physical activity and by the administration of eight to twelve fluid ounces of water. If emesis does not occur within fifteen minutes, the dose of ipecac should be repeated. Precautions against aspiration must be taken, especially in infants and children. Following emesis, any drug remaining in the stomach may be absorbed by activated charcoal administered as a slurry with water. If vomiting is unsuccessful or contraindicated, gastric lavage should be performed. Isotonic and one-half isotonic saline are the lavage solutions of choice. Saline cathartics, such as milk of magnesia, draw water into the bowel by osmosis and, therefore, may be valuable for their action in rapid dilution of bowel content. After emergency treatment the patient should continue to be medically monitored. Treatment of the signs and symptoms of overdosage is symptomatic and supportive.
Stimulants (analeptic agents) should not be used. Vasopressors may be used to treat hypotension. Short-acting barbiturates, diazepam, or paraldehyde may be administered to control seizures. Hyperpyrexia, especially in children, may require treatment with tepid water sponge baths or a hypothermic blanket. Apnea is treated with ventilatory support.
DOSAGE AND ADMINISTRATION
Adults and Children 12 years of age and older
1 to 2 teaspoonfuls (5 to 10 mL) every 4 to 6 hours, not to exceed 8 teaspoonfuls in a 24 hour period.
This product is not indicated for use in children under 12 years of age. (see PRECAUTIONS, Pediatric Use.)
HOW SUPPLIED
Chlorpheniramine and Codeine Liquid is supplied as a sugar free, alcohol free, dye free grape-flavored liquid for oral administration. Available in 16 oz. (473 mL) bottles, NDC 51991-657-16.
Dispense in a tight, light-resistant container with a child-resistant closure as defined in the USP/NF.
Store at 25° C (77° F ); excursions permitted to 15° – 30°C (59° – 86° F). See USP Controlled Room Temperature. Protect from freezing.
Warning: Keep this and all medications out of the reach of children. In case of accidental overdose, seek professional assistance or contact a poison control center immediately.
All prescription substitutions using this product shall be pursuant to state statutes as applicable. This is not an Orange Book product.
Manufactured by:
Provident Pharmaceuticals
Colorado Springs, CO 80919
Distributed by:
Breckenridge Pharmaceutical, Inc.
Boca Raton, FL 33487
PL372
Iss 7/09
PRINCIPAL DISPLAY PANEL - 473 mL Bottle Label
Breckenridge
Pharmaceutical, Inc.
NDC 51991-657-16
Chlorpheniramine and
Codeine Liquid
Antihistamine • Antitussive
Sugar free • Alcohol free • Dye free
CV
| Each 5 mL (1 teaspoonful) contains: | |
| Codeine Phosphate* | 10 mg |
| *(WARNING: MAY BE HABIT-FORMING) | |
| Chlorpheniramine Maleate | 2 mg |
| Do not use if safety seal under cap is broken or missing. |
Rx Only
ONE PINT (473 mL)

| CHLORPHENIRAMINE AND CODEINE
codeine phosphate and chlorpheniramine maleate liquid |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| UNAPPROVED DRUG OTHER | 10/01/2009 | 02/28/2011 | |
| Labeler - Breckenridge Pharmaceutical, Inc. (150554335) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Provident Pharms Inc | 171901445 | MANUFACTURE | |