methotrexate (Methotrexate) injection
[Bedford Laboratories]
Rx ONLY
WARNINGS
METHOTREXATE SHOULD BE USED ONLY BY PHYSICIANS WHOSE KNOWLEDGE AND EXPERIENCE INCLUDES THE USE OF ANTIMETABOLITE THERAPY.
BECAUSE OF THE POSSIBILITY OF SERIOUS TOXIC REACTIONS (WHICH CAN BE FATAL):
- METHOTREXATE SHOULD BE USED ONLY IN LIFE THREATENING NEOPLASTIC DISEASES, OR IN PATIENTS WITH PSORIASIS WITH SEVERE, RECALCITRANT, DISABLING DISEASE WHICH IS NOT ADEQUATELY RESPONSIVE TO OTHER FORMS OF THERAPY.
- DEATHS HAVE BEEN REPORTED WITH THE USE OF METHOTREXATE IN THE TREATMENT OF MALIGNANCY AND PSORIASIS.
- PATIENTS SHOULD BE CLOSELY MONITORED FOR BONE MARROW, LIVER, LUNG AND KIDNEY TOXICITIES. (See PRECAUTIONS.)
- PATIENTS SHOULD BE INFORMED BY THEIR PHYSICIAN OF THE RISKS INVOLVED AND BE UNDER A PHYSICIAN’S CARE THROUGHOUT THERAPY.
THE USE OF METHOTREXATE HIGH DOSE REGIMENS RECOMMENDED FOR OSTEOSARCOMA REQUIRES METICULOUS CARE. (See DOSAGE AND ADMINISTRATION.) HIGH DOSE REGIMENS FOR OTHER NEOPLASTIC DISEASES ARE INVESTIGATIONAL AND A THERAPEUTIC ADVANTAGE HAS NOT BEEN ESTABLISHED.
METHOTREXATE FORMULATIONS AND DILUENTS CONTAINING PRESERVATIVES MUST NOT BE USED FOR INTRATHECAL OR HIGH DOSE METHOTREXATE THERAPY.
- Methotrexate has been reported to cause fetal death and/or congenital anomalies. Therefore, it is not recommended for women of childbearing potential unless there is clear medical evidence that the benefits can be expected to outweigh the considered risks. Pregnant women with psoriasis should not receive methotrexate (See CONTRAINDICATIONS).
- Methotrexate elimination is reduced in patients with impaired renal function, ascites, or pleural effusions. Such patients require especially careful monitoring for toxicity, and require dose reduction or, in some cases, discontinuation of methotrexate administration.
- Unexpectedly severe (sometimes fatal) bone marrow suppression and gastrointestinal toxicity have been reported with concomitant administration of methotrexate (usually in high dosage) along with some nonsteroidal anti-inflammatory drugs (NSAIDs). (See PRECAUTIONS, Drug Interactions.)
- Methotrexate causes hepatotoxicity, fibrosis and cirrhosis, but generally only after prolonged use. Acutely, liver enzyme elevations are frequently seen. These are usually transient and asymptomatic, and also do not appear predictive of subsequent hepatic disease. Liver biopsy after sustained use often shows histologic changes, and fibrosis and cirrhosis have been reported; these latter lesions may not be preceded by symptoms or abnormal liver function tests in the psoriasis population. For this reason, periodic liver biopsies are usually recommended for psoriatic patients who are under long-term treatment. (See PRECAUTIONS, Organ System Toxicity, Hepatic.)
- Methotrexate-induced lung disease is a potentially dangerous lesion, which may occur acutely at any time during therapy and which has been reported at doses as low as 7.5 mg/week. It is not always fully reversible. Pulmonary symptoms (especially a dry, nonproductive cough) may require interruption of treatment and careful investigation.
- Diarrhea and ulcerative stomatitis require interruption of therapy; otherwise, hemorrhagic enteritis and death from intestinal perforation may occur.
- Malignant lymphomas, which may regress following withdrawal ofmethotrexate, may occur in patients receiving low-dose methotrexate and, thus, may not require cytotoxic treatment. Discontinue methotrexate first and, if the lymphoma does not regress, appropriate treatment should be instituted.
- Like other cytotoxic drugs, methotrexate may induce “tumor lysis syndrome” in patients with rapidly growing tumors. Appropriate supportive and pharmacologic measures may prevent or alleviate this complication.
- Severe, occasionally fatal, skin reactions have been reported following single or multiple doses of methotrexate. Reactions have occurred within days of oral, intramuscular, intravenous, or intrathecal methotrexate administration. Recovery has been reported with discontinuation of therapy. (See PRECAUTIONS, Organ System Toxicity, Skin.)
- Potentially fatal opportunistic infections, especially Pneumocystis carinii pneumonia, may occur with methotrexate therapy.
- Methotrexate given concomitantly with radiotherapy may increase the risk of soft tissue necrosis and osteonecrosis.
DESCRIPTION
Methotrexate is an antimetabolite used in the treatment of certain neoplastic diseases and severe psoriasis. Chemically methotrexate is N-[4-[[(2,4-diamino-6-pteridinyl)methyl]methylamino]benzoyl]-L-glutamic acid. The structural formula is:
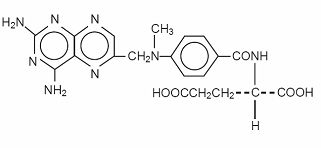
C20H22N8O5 M.W.=454.45
Methotrexate Injection, USP is available in 50, 100, 200, and 250 mg single-dose vials of sterile, isotonic liquid containing no preservatives for parenteral administration.
Each 25 mg/mL, 2 mL (50 mg) vial contains methotrexate sodium equivalent to 50 mg methotrexate. Inactive ingredients are sodium chloride for isotonicity and sodium hydroxide and, if necessary, hydrochloric acid to adjust pH to approximately 8.5.
Each 25 mg/mL, 4 mL (100 mg) vial contains methotrexate sodium equivalent to 100 mg methotrexate. Inactive ingredients are sodium chloride for isotonicity and sodium hydroxide and, if necessary, hydrochloric acid to adjust pH to approximately 8.5.
Each 25 mg/mL, 8 mL (200 mg) vial contains methotrexate sodium equivalent to 200 mg methotrexate. Inactive ingredients are sodium chloride for isotonicity and sodium hydroxide and, if necessary, hydrochloric acid to adjust pH to approximately 8.5.
Each 25 mg/mL, 10 mL (250 mg) vial contains methotrexate sodium equivalent to 250 mg methotrexate. Inactive ingredients are sodium chloride for isotonicity and sodium hydroxide and, if necessary, hydrochloric acid to adjust pH to approximately 8.5.
CLINICAL PHARMACOLOGY
Methotrexate inhibits dihydrofolic acid reductase. Dihydrofolates must be reduced to tetrahydrofolates by this enzyme before they can be utilized as carriers of one-carbon groups in the synthesis of purine nucleotides and thymidylate. Therefore, methotrexate interferes with DNA synthesis, repair, and cellular replication. Actively proliferating tissues such as malignant cells, bone marrow, fetal cells, buccal and intestinal mucosa and cells of the urinary bladder are in general more sensitive to this effect of methotrexate. When cellular proliferation in malignant tissues is greater than in most normal tissues, methotrexate may impair malignant growth without irreversible damage to normal tissues.
In psoriasis, the rate of production of epithelial cells in the skin is greatly increased over normal skin. This differential in proliferation rates is the basis for the use of methotrexate to control the psoriatic process.
Methotrexate in high doses, followed by leucovorin rescue, is used as a part of the treatment of patients with non-metastatic osteosarcoma. The original rationale for high dose methotrexate therapy was based on the concept of selective rescue of normal tissues by leucovorin. More recent evidence suggests that high dose methotrexate may also overcome methotrexate resistance caused by impaired active transport, decreased affinity of dihydrofolic acid reductase for methotrexate, increased levels of dihydrofolic acid reductase resulting from gene amplification, or decreased polyglutamation of methotrexate. The actual mechanism of action is unknown.
Two Pediatric Oncology Group studies (one randomized and one non-randomized) demonstrated a significant improvement in relapse-free survival in patients with non-metastatic osteosarcoma, when high dose methotrexate with leucovorin rescue was used in combination with other chemotherapeutic agents following surgical resection of the primary tumor. These studies were not designed to demonstrate the specific contribution of high dose methotrexate/leucovorin rescue therapy to the efficacy of the combination. However, a contribution can be inferred from the reports of objective responses to this therapy in patients with metastatic osteosarcoma, and from reports of extensive tumor necrosis following preoperative administration of this therapy to patients with non-metastatic osteosarcoma.
Pharmacokinetics
Absorption
Methotrexate is generally completely absorbed from parenteral routes of injection. After intramuscular injection, peak serum concentrations occur in 30 to 60 minutes.
Distribution
After intravenous administration, the initial volume of distribution is approximately 0.18 L/kg (18% of body weight) and steady-state volume of distribution is approximately 0.4 to 0.8 L/kg (40 to 80% of body weight). Methotrexate competes with reduced folates for active transport across cell membranes by means of a single carrier-mediated active transport process. At serum concentrations greater than 100 micromolar, passive diffusion becomes a major pathway by which effective intracellular concentrations can be achieved. Methotrexate in serum is approximately 50% protein bound. Laboratory studies demonstrate that it may be displaced from plasma albumin by various compounds including sulfonamides, salicylates, tetracyclines, chloramphenicol, and phenytoin.
Methotrexate does not penetrate the blood-cerebrospinal fluid barrier in therapeutic amounts when given orally or parenterally. High CSF concentrations of the drug may be attained by intrathecal administration.
Metabolism
After absorption, methotrexate undergoes hepatic and intracellular metabolism to polyglutamated forms which can be converted back to methotrexate by hydrolase enzymes. These polyglutamates act as inhibitors of dihydrofolate reductase and thymidylate synthetase. Small amounts of methotrexate polyglutamates may remain in tissues for extended periods. The retention and prolonged drug action of these active metabolites vary among different cells, tissues and tumors. A small amount of metabolism to 7-hydroxymethotrexate may occur at doses commonly prescribed. Accumulation of this metabolite may become significant at the high doses used in osteogenic sarcoma. The aqueous solubility of 7-hydroxymethotrexate is 3 to 5 fold lower than the parent compound. Methotrexate is partially metabolized by intestinal flora after oral administration.
Half-Life
The terminal half-life reported for methotrexate is approximately 3 to 10 hours for patients receiving treatment for psoriasis or low-dose antineoplastic therapy (less than 30 mg/m2). For patients receiving high doses of methotrexate, the terminal half-life is eight to 15 hours.
Excretion
Renal excretion is the primary route of elimination and is dependent upon dosage and route of administration. With IV administration, 80 to 90% of the administered dose is excreted unchanged in the urine within 24 hours. There is limited biliary excretion amounting to 10% or less of the administered dose. Enterohepatic recirculation of methotrexate has been proposed.
Renal excretion occurs by glomerular filtration and active tubular secretion. Nonlinear elimination due to saturation of renal tubular reabsorption has been observed in psoriatic patients at doses between 7.5 and 30 mg. Impaired renal function, as well as concurrent use of drugs such as weak organic acids that also undergo tubular secretion, can markedly increase methotrexate serum levels. Excellent correlation has been reported between methotrexate clearance and endogenous clearance.
Methotrexate clearance rates vary widely and are generally decreased at higher doses. Delayed drug clearance has been identified as one of the major factors responsible for methotrexate toxicity. It has been postulated that the toxicity of methotrexate for normal tissues is more dependent upon the duration of exposure to the drug rather than the peak level achieved. When a patient has delayed drug elimination due to compromised renal function, a third space effusion, or other causes, methotrexate serum concentrations may remain elevated for prolonged periods.
The potential for toxicity from high dose regimens or delayed excretion is reduced by the administration of leucovorin calcium during the final phase of methotrexate plasma elimination. Pharmacokinetic monitoring of methotrexate serum concentrations may help identify those patients at high risk for methotrexate toxicity and aid in proper adjustment of leucovorin dosing. Guidelines for monitoring serum methotrexate levels, and for adjustment of leucovorin dosing to reduce the risk of methotrexate toxicity, are provided below in DOSAGE AND ADMINISTRATION.
Methotrexate has been detected in human breast milk. The highest breast milk to plasma concentration ratio reached was 0.08:1.
INDICATIONS AND USAGE
Neoplastic Diseases
Methotrexate is indicated in the treatment of gestational choriocarcinoma, chorioadenoma destruens and hydatidiform mole.
In acute lymphocytic leukemia, methotrexate is indicated in the prophylaxis of meningeal leukemia and is used in maintenance therapy in combination with other chemotherapeutic agents. Methotrexate is also indicated in the treatment of meningeal leukemia.
Methotrexate is used alone or in combination with other anticancer agents in the treatment of breast cancer, epidermoid cancers of the head and neck, advanced mycosis fungoides, and lung cancer, particularly squamous cell and small cell types. Methotrexate is also used in combination with other chemotherapeutic agents in the treatment of advanced stage non-Hodgkin’s lymphomas.
Methotrexate in high doses followed by leucovorin rescue in combination with other chemotherapeutic agents is effective in prolonging relapse-free survival in patients with non-metastatic osteosarcoma who have undergone surgical resection or amputation for the primary tumor.
Psoriasis
Methotrexate is indicated in the symptomatic control of severe, recalcitrant, disabling psoriasis which is not adequately responsive to other forms of therapy, but only when the diagnosis has been established, as by a biopsy and/or after dermatologic consultation. It is important to ensure that a psoriasis “flare” is not due to an undiagnosed concomitant disease affecting immune responses.
CONTRAINDICATIONS
Methotrexate can cause fetal death or teratogenic effects when administered to a pregnant woman. Methotrexate is contraindicated in pregnant women with psoriasis and should be used in the treatment of neoplastic diseases only when the potential benefit outweighs the risk to the fetus. Women of childbearing potential should not be started on methotrexate until pregnancy is excluded and should be fully counseled on the serious risk to the fetus (See PRECAUTIONS) should they become pregnant while undergoing treatment. Pregnancy should be avoided if either partner is receiving methotrexate; during and for a minimum of 3 months after therapy for male patients, and during and for at least one ovulatory cycle after therapy for female patients (See Boxed WARNINGS).
Because of the potential for serious adverse reactions from methotrexate in breast fed infants, it is contraindicated in nursing mothers.
Patients with psoriasis with alcoholism, alcoholic liver disease or other chronic liver disease should not receive methotrexate.
Patients with psoriasis who have overt or laboratory evidence of immunodeficiency syndromes should not receive methotrexate.
Patients with psoriasis who have preexisting blood dyscrasias, such as bone marrow hypoplasia, leukopenia, thrombocytopenia, or significant anemia, should not receive methotrexate.
Patients with known hypersensitivity to methotrexate should not receive the drug.
WARNINGS
See Boxed WARNINGS.
PRECAUTIONS
General
Methotrexate has the potential for serious toxicity (See Boxed WARNINGS). Toxic effects may be related in frequency and severity to dose or frequency of administration but have been seen at all doses. Because they can occur at any time during therapy, it is necessary to follow patients on methotrexate closely. Most adverse reactions are reversible if detected early. When such reactions do occur, the drug should be reduced in dosage or discontinued and appropriate corrective measures should be taken. If necessary, this could include the use of leucovorin calcium and/or acute, intermittent hemodialysis with a high-flux dialyzer. (See OVERDOSAGE). If methotrexate therapy is reinstituted, it should be carried out with caution, with adequate consideration of further need for the drug and increased alertness as to possible recurrence of toxicity.
The clinical pharmacology of methotrexate has not been well studied in older individuals. Due to diminished hepatic and renal function as well as decreased folate stores in this population, relatively low doses should be considered, and these patients should be closely monitored for early signs of toxicity.
Information for Patients
Patients should be informed of the early signs and symptoms of toxicity, of the need to see their physician promptly if they occur, and the need for close follow-up, including periodic laboratory tests to monitor toxicity.
Patients should be informed of the potential benefit and risk in the use of methotrexate. The risk of effects on reproduction should be discussed with both male and female patients taking methotrexate.
Laboratory Tests
Patients undergoing methotrexate therapy should be closely monitored so that toxic effects are detected promptly. Baseline assessment should include complete blood count with differential and platelet counts, hepatic enzymes, renal function tests, and a chest X-ray. During therapy of psoriasis, monitoring of these parameters is recommended; hematology at least monthly, and liver and renal function every 1 to 2 months. More frequent monitoring is usually indicated during antineoplastic therapy. During initial or changing doses, or during periods of increased risk of elevated methotrexate blood levels (e.g., dehydration), more frequent monitoring may also be indicated.
Transient liver function test abnormalities are observed frequently after methotrexate administration and are usually not cause for modification of methotrexate therapy. Persistent liver function test abnormalities, and/or depression of serum albumin may be indicators of serious liver toxicity and require evaluation. (See PRECAUTIONS, Organ System Toxicity, Hepatic.)
A relationship between abnormal liver function tests and fibrosis or cirrhosis of the liver has not been established for patients with psoriasis.
Pulmonary function tests may be useful if methotrexate-induced lung disease is suspected, especially if baseline measurements are available.
Drug Interactions
Nonsteroidal anti-inflammatory drugs should not be administered prior to or concomitantly with the high doses of methotrexate used in the treatment of osteosarcoma. Concomitant administration of some NSAIDs with high dose methotrexate therapy has been reported to elevate and prolong serum methotrexate levels, resulting in deaths from severe hematologic and gastrointestinal toxicity.
Caution should be used when NSAIDs or salicylates are administered concomitantly with lower doses of methotrexate. These drugs have been reported to reduce the tubular secretion of methotrexate in an animal model and may enhance its toxicity.
Methotrexate is partially bound to serum albumin, and toxicity may be increased because of displacement by certain drugs, such as salicylates, phenylbutazone, phenytoin, and sulfonamides. Renal tubular transport is also diminished by probenecid; use of methotrexate with this drug should be carefully monitored.
In the treatment of patients with osteosarcoma, caution must be exercised if high-dose methotrexate is administered in combination with a potentially nephrotoxic chemotherapeutic agent (e.g., cisplatin).
Oral antibiotics such as tetracycline, chloramphenicol, and nonabsorbable broad spectrum antibiotics, may decrease intestinal absorption of methotrexate or interfere with the enterohepatic circulation by inhibiting bowel flora and suppressing metabolism of the drug by bacteria.
Penicillins may reduce the renal clearance of methotrexate; increased serum concentrations of methotrexate with concomitant hematologic and gastrointestinal toxicity have been observed with high and low dose methotrexate. Use of methotrexate with penicillins should be carefully monitored.
The potential for increased hepatotoxicity when methotrexate is administered with other hepatotoxic agents has not been evaluated. However, hepatotoxicity has been reported in such cases. Therefore, patients receiving concomitant therapy with methotrexate and other potential hepatotoxins (e.g., azathioprine, retinoids, sulfasalazine) should be closely monitored for possible risk of hepatotoxicity.
Methotrexate may decrease the clearance of theophylline; theophylline levels should be monitored when used concurrently with methotrexate.
Vitamin preparations containing folic acid or its derivatives may decrease responses to systemically administered methotrexate. Preliminary animal and human studies have shown that small quantities of intravenously administered leucovorin enter the CSF primarily as 5-methyltetrahydrofolate and, in humans, remain 1 to 3 orders of magnitude lower than the usual methotrexate concentrations following intrathecal administration. However, high doses of leucovorin may reduce the efficacy of intrathecally administered methotrexate.
Folate deficiency states may increase methotrexate toxicity. Trimethoprim/sulfamethoxazole has been reported rarely to increase bone marrow suppression in patients receiving methotrexate, probably by an additive antifolate effect.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No controlled human data exist regarding the risk of neoplasia with methotrexate. Methotrexate has been evaluated in a number of animal studies for carcinogenic potential with inconclusive results. Although there is evidence that methotrexate causes chromosomal damage to animal somatic cells and human bone marrow cells, the clinical significance remains uncertain. Non-Hodgkin's lymphoma and other tumors have been reported in patients receiving low-dose oral methotrexate. However, there have been instances of malignant lymphoma arising during treatment with low-dose oral methotrexate, which have regressed completely following withdrawal of methotrexate, without requiring active anti-lymphoma treatment. Benefits should be weighed against this potential risk before using methotrexate alone or in combination with other drugs, especially in pediatric patients or young adults. Methotrexate causes embryotoxicity, abortion, and fetal defects in humans. It has also been reported to cause impairment of fertility, oligospermia and menstrual dysfunction in humans, during and for a short period after cessation of therapy.
Pregnancy
Teratogenic Effects, Pregnancy Category X
Psoriasis: Methotrexate is in Pregnancy Category X (See CONTRAINDICATIONS).
Nursing Mothers
See CONTRAINDICATIONS.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established, other than in cancer chemotherapy.
Geriatric Use
Clinical studies of methotrexate did not include sufficient numbers of subjects age 65 and over to determine whether they respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious reflecting the greater frequency of decreased hepatic and renal function, decreased folate stores, concomitant disease or other drug therapy (ie, that interfere with renal function, methotrexate or folate metabolism) in this population (see PRECAUTIONS, Drug Interactions). Since decline in renal function may be associated with increases in adverse events and serum creatinine measurements may over estimate renal function in the elderly, more accurate methods (ie, creatinine clearance) should be considered. Serum methotrexate levels may also be helpful. Elderly patients should be closely monitored for early signs of hepatic, bone marrow and renal toxicity. In chronic use situations, certain toxicities may be reduced by folate supplementation. Post-marketing experience suggests that the occurrence of bone marrow suppression, thrombocytopenia, and pneumonitis may increase with age. See Boxed WARNINGS and ADVERSE REACTIONS.
Organ System Toxicity
Gastrointestinal
If vomiting, diarrhea, or stomatitis occur, which may result in dehydration, methotrexate should be discontinued until recovery occurs. Methotrexate should be used with extreme caution in the presence of peptic ulcer disease or ulcerative colitis.
Hematologic
Methotrexate can suppress hematopoiesis and cause anemia, leukopenia, and/or thrombocytopenia. In patients with malignancy and preexisting hematopoietic impairment, the drug should be used with caution, if at all.
In psoriasis, methotrexate should be stopped immediately if there is a significant drop in blood counts. In the treatment of neoplastic diseases, methotrexate should be continued only if the potential benefit warrants the risk of severe myelosuppression. Patients with profound granulocytopenia and fever should be evaluated immediately and usually require parenteral broad-spectrum antibiotic therapy.
Hepatic
Methotrexate has the potential for acute (elevated transaminases) and chronic (fibrosis and cirrhosis) hepatotoxicity. Chronic toxicity is potentially fatal, it generally has occurred after prolonged use (generally two years or more) and after a total dose of at least 1.5 grams. In studies in psoriatic patients, hepatotoxicity appeared to be a function of total cumulative dose and appeared to be enhanced by alcoholism, obesity, diabetes, and advanced age. An accurate incidence rate has not been determined; the rate of progression and reversibility of lesions is not known. Special caution is indicated in the presence of preexisting liver damage or impaired hepatic function.
In psoriasis, liver function tests, including serum albumin, should be performed periodically prior to dosing but are often normal in the face of developing fibrosis or cirrhosis. These lesions may be detectable only by biopsy. The usual recommendation is to obtain a liver biopsy at 1) pretherapy or shortly after initiation of therapy (2 to 4 months), 2) a total cumulative dose of 1.5 grams, and 3) after each additional 1.0 to 1.5 grams. Moderate fibrosis or any cirrhosis normally leads to discontinuation of the drug; mild fibrosis normally suggests a repeat biopsy in 6 months. Milder histologic findings such as fatty change and low grade portal inflammation are relatively common pretherapy. Although these mild changes are usually not a reason to avoid or discontinue methotrexate therapy, the drug should be used with caution.
Infection or Immunologic States
Methotrexate should be used with extreme caution in the presence of active infection, and is usually contraindicated in patients with overt or laboratory evidence of immunodeficiency syndromes. Immunization may be ineffective when given during methotrexate therapy. Immunization with live virus vaccines is generally not recommended. There have been reports of disseminated vaccinia infections after smallpox immunizations in patients receiving methotrexate therapy Hypogammaglobulinemia has been reported rarely.
Potentially fatal opportunistic infections, especially Pneumocystis carinii pneumonia may occur with methotrexate therapy. When a patient presents with pulmonary symptoms, the possibility of Pneumocystis carinii pneumonia should be considered.
Neurologic
There have been reports of leukoencephalopathy following intravenous administration of methotrexate to patients who have had craniospinal irradiation. Serious neurotoxicity, frequently manifested as generalized or focal seizures, has been reported with unexpectedly increased frequency among pediatric patients with acute lymphoblastic leukemia who were treated with intermediate-dose intravenous methotrexate (1 gm/m2). Symptomatic patients were commonly noted to have leukoencephalopathy and/or microangiopathic calcifications on diagnostic imaging studies. Chronic leukoencephalopathy has also been reported in patients who received repeated doses of high-dose methotrexate with leucovorin rescue even without cranial irradiation. Discontinuation of methotrexate does not always result in complete recovery.
A transient acute neurologic syndrome has been observed in patients treated with high dosage regimens. Manifestations of this stroke-like encephalopathy may include confusion, hemiparesis, seizures and coma. The exact cause is unknown.
After the intrathecal use of methotrexate, the central nervous system toxicity which may occur can be classified as follows: acute chemical arachnoiditis manifested by such symptoms as headache, back pain, nuchal rigidity, and fever; sub-acute myelopathy characterized by paraparesis/paraplegia associated with involvement with one or more spinal nerve roots; chronic leukoencephalopathy manifested by confusion, irritability, somnolence, ataxia, dementia, seizures and coma. This condition can be progressive and even fatal.
Pulmonary
Pulmonary symptoms (especially a dry nonproductive cough) or a nonspecific pneumonitis occurring during methotrexate therapy may be indicative of a potentially dangerous lesion and require interruption of treatment and careful investigation. Although clinically variable, the typical patient with methotrexate induced lung disease presents with fever, cough, dyspnea, hypoxemia, and an infiltrate on chest X-ray; infection needs to be excluded. This lesion can occur at all dosages.
Renal
High doses of methotrexate used in the treatment of osteosarcoma may cause renal damage leading to acute renal failure. Nephrotoxicity is due primarily to the precipitation of methotrexate and 7-hydroxymethotrexate in the renal tubules. Close attention to renal function including adequate hydration, urine alkalinization and measurement of serum methotrexate and creatinine levels are essential for safe administration.
Skin
Severe, occasionally fatal, dermatologic reactions, including toxic epidermal necrolysis, Stevens-Johnson syndrome, exfoliative dermatitis, skin necrosis, and erythema multiforme, have been reported in children and adults, within days of oral, intramuscular, intravenous, or intrathecal methotrexate administration. Reactions were noted after single or multiple, low, intermediate or high doses of methotrexate in patients with neoplastic and non-neoplastic diseases.
Other Precautions
Methotrexate should be used with extreme caution in the presence of debility.
Methotrexate exits slowly from third space compartments (e.g., pleural effusions or ascites). This results in a prolonged terminal plasma half-life and unexpected toxicity. In patients with significant third space accumulations, it is advisable to evacuate the fluid before treatment and to monitor plasma methotrexate levels.
Lesions of psoriasis may be aggravated by concomitant exposure to ultraviolet radiation. Radiation dermatitis and sunburn may be “recalled” by the use of methotrexate.
ADVERSE REACTIONS
IN GENERAL, THE INCIDENCE AND SEVERITY OF ACUTE SIDE EFFECTS ARE RELATED TO DOSE AND FREQUENCY OF ADMINISTRATION. THE MOST SERIOUS REACTIONS ARE DISCUSSED ABOVE UNDER ORGAN SYSTEM TOXICITY IN THE PRECAUTIONS SECTION. THAT SECTION SHOULD ALSO BE CONSULTED WHEN LOOKING FOR INFORMATION ABOUT ADVERSE REACTIONS WITH METHOTREXATE.
The most frequently reported adverse reactions include ulcerative stomatitis, leukopenia, nausea, and abdominal distress. Other frequently reported adverse effects are malaise, undue fatigue, chills and fever, dizziness and decreased resistance to infection.
Other adverse reactions that have been reported with methotrexate are listed below by organ system. In the oncology setting, concomitant treatment and the underlying disease make specific attribution of a reaction to methotrexate difficult.
Alimentary System - gingivitis, pharyngitis, stomatitis, anorexia, nausea, vomiting, diarrhea, hematemesis, melena, gastrointestinal ulceration and bleeding, enteritis, pancreatitis.
Blood and Lymphatic System Disorders - suppressed hematopoiesis causing anemia, aplastic anemia, pancytopenia, leukopenia, neutropenia and/or thrombocytopenia, lymphadenopathy and lymphoproliferative disorders (including reversible). Hypogammaglobulinemia has been reported rarely.
Cardiovascular - pericarditis, pericardial effusion, hypotension, and thromboembolic events (including arterial thrombosis, cerebral thrombosis, deep vein thrombosis, retinal vein thrombosis, thrombophlebitis, and pulmonary embolus).
Central Nervous System - headaches, drowsiness, blurred vision, transient blindness, speech impairment including dysarthria and aphasia, hemiparesis, paresis and convulsions have also occurred following administration of methotrexate. Following low doses, there have been occasional reports of transient subtle cognitive dysfunction, mood alteration, unusual cranial sensations, leukoencephalopathy, or encephalopathy.
Hepatobiliary Disorders - hepatotoxicity, acute hepatitis, chronic fibrosis and cirrhosis, decrease in serum albumin, liver enzyme elevations.
Infection - infections, pneumonia, sepsis, nocardiosis, histoplasmosis, cryptococcosis, Herpes zoster, H. simplex hepatitis, and disseminated H. simplex. There have been case reports of sometimes fatal opportunistic infections in patients receiving methotrexate therapy for neoplastic and non-neoplastic diseases. Pneumocystis carinii pneumonia was the most common infection.
Musculoskeletal System - stress fracture.
Ophthalmic - conjunctivitis, serious visual changes of unknown etiology.
Pulmonary System– respiratory fibrosis, respiratory failure, interstitial pneumonitis; deaths have been reported, and chronic interstitial obstructive pulmonary disease has occasionally occurred.
Skin - erythematous rashes, pruritus, urticaria, photosensitivity, pigmentary changes, alopecia, ecchymosis, telangiectasia, acne, furunculosis, erythema multiforme, toxic epidermal necrolysis, Stevens-Johnson syndrome, skin necrosis, and exfoliative dermatitis.
Urogenital System - severe nephropathy or renal failure, azotemia, cystitis, hematuria; defective oogenesis or spermatogenesis, transient oligospermia, menstrual dysfunction, vaginal discharge, and gynecomastia; infertility, abortion, fetal defects.
Other rarer reactions related to or attributed to the use of methotrexate such as nodulosis, vasculitis, arthralgia/myalgia, loss of libido/impotence, diabetes, osteoporosis, sudden death, reversible lymphomas, tumor lysis syndrome, soft tissue necrosis, and osteonecrosis. Anaphylactoid reactions have been reported.
Adverse Reactions in Psoriasis
There are no recent placebo-controlled trials in patients with psoriasis. There are two literature reports (Roenigk, 1969, and Nyfors, 1978) describing large series (n=204, 248) of psoriasis patients treated with methotrexate. Dosages ranged up to 25 mg per week and treatment was administered for up to four years. With the exception of alopecia, photosensitivity, and “burning of skin lesions” (each 3 to 10%), the adverse reaction rates in these reports were very similar to those in the rheumatoid arthritis studies. Rarely, painful plaque erosions may appear (Pearce, HP and Wilson, BB: Am Acad Dermatol 35: 835-838, 1996).
OVERDOSAGE
Leucovorin is indicated to diminish the toxicity and counteract the effect of inadvertently administered overdosages of methotrexate. Leucovorin administration should begin as promptly as possible. As the time interval between methotrexate administration and leucovorin initiation increases, the effectiveness of leucovorin in counteracting toxicity decreases. Monitoring of the serum methotrexate concentration is essential in determining the optimal dose and duration of treatment with leucovorin.
In cases of massive overdosage, hydration and urinary alkalinization may be necessary to prevent the precipitation of methotrexate and/or its metabolites in the renal tubules. Generally speaking, neither hemodialysis nor peritoneal dialysis have been shown to improve methotrexate elimination. However, effective clearence of methotrexate has been reported with acute, intermittent hemodialysis using a high-flux dialyzer (Wall, SM et al: AM J Kidney Dis 28(6): 846-854, 1996).
Accidental intrathecal overdosage may require intensive systemic support, high-dose systemic leucovorin, alkaline diuresis and rapid CSF drainage and ventriculolumbar perfusion.
In postmarketing experience, overdose with methotrexate has generally occurred with oral and intrathecal administration, although intravenous and intramuscular overdose have also been reported.
Symptoms of intrathecal overdose are generally central nervous system (CNS) symptoms, including headache, nausea and vomiting, seizure or convulsion, and acute toxic encephalopathy. In some cases, no symptoms were reported. There have been reports of death following intrathecal overdose. In these cases, cerebellar herniation associated with increased intracranial pressure, and acute toxic encephalopathy have also been reported.
DOSAGE AND ADMINISTRATION
Neoplastic Diseases
Oral administration in tablet form is often preferred when low doses are being administered since absorption is rapid and effective serum levels are obtained. Methotrexate sodium injection may be given by the intramuscular, intravenous, intra-arterial, or intrathecal route. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Choriocarcinoma and Similar Trophoblastic Diseases
Methotrexate is administered orally or intramuscularly in doses of 15 to 30 mg daily for a 5-day course. Such courses are usually repeated for 3 to 5 times as required, with rest periods of one or more weeks interposed between courses, until any manifesting toxic symptoms subside. The effectiveness of therapy is ordinarily evaluated by 24-hour quantitative analysis of urinary chorionic gonadatropin (hCG), which should return to normal or less than 50 IU/24 hr usually after the third or fourth course and usually be followed by a complete resolution of measurable lesions in 4 to 6 weeks. One to two courses of methotrexate after normalization of hCG is usually recommended. Before each course of the drug careful clinical assessment is essential. Cyclic combination therapy of methotrexate with other antitumor drugs has been reported as being useful.
Since hydatidiform mole may precede choriocarcinoma, prophylactic chemotherapy with methotrexate has been recommended.
Chorioadenoma destruens is considered to be an invasive form of hydatidiform mole. Methotrexate is administered in these disease states in doses similar to those recommended for choriocarcinoma.
Leukemia
Acute lymphoblastic leukemia in pediatric patients and young adolescents is the most responsive to present day chemotherapy. In young adults and older patients, clinical remission is more difficult to obtain and early relapse is more common.
Methotrexate alone or in combination with steroids was used initially for induction of remission in lymphoblastic leukemias. More recently corticosteroid therapy, in combination with other antileukemic drugs or in cyclic combinations with methotrexate included, has appeared to produce rapid and effective remissions. When used for induction, methotrexate in doses of 3.3 mg/m2 in combination with 60 mg/m2 of prednisone, given daily, produced remissions in 50% of patients treated, usually within a period of 4 to 6 weeks. Methotrexate in combination with other agents appears to be the drug of choice for securing maintenance of drug-induced remissions. When remission is achieved and supportive care has produced general clinical improvement, maintenance therapy is initiated, as follows: methotrexate is administered 2 times weekly either by mouth or intramuscularly in total weekly doses of 30 mg/m2. It has also been given in doses of 2.5 mg/kg intravenously every 14 days. If and when relapse does occur, reinduction of remission can again usually be obtained by repeating the initial induction regimen.
A variety of combination chemotherapy regimens have been used for both induction and maintenance therapy in acute lymphoblastic leukemia. The physician should be familiar with the new advances in antileukemic therapy.
Meningeal Leukemia
In the treatment or prophylaxis of meningeal leukemia, methotrexate must be administered intrathecally. Preservative free methotrexate is diluted to a concentration of 1 mg/mL with an appropriate sterile, preservative free medium such as 0.9% Sodium Chloride Injection, USP.
The cerebrospinal fluid volume is dependent on age and not body surface area. The CSF is at 40% of the adult volume at birth and reaches the adult volume in several years.
Intrathecal methotrexate administration at a dose of 12 mg/m2 (maximum 15 mg) has been reported to result in low CSF methotrexate concentrations and reduced efficacy in pediatric patients and high concentrations and neurotoxicity in adults. The following dosage regimen is based on age instead of body surface area:
| Age (years) | Dose (mg) |
| <1 | 6 |
| 1 | 8 |
| 2 | 10 |
| 3 or older | 12 |
In one study in patients under the age of 40, this dosage regimen appeared to result in more consistent CSF methotrexate concentrations and less neurotoxicity. Another study in pediatric patients with acute lymphocytic leukemia compared the above regimen to a dose of 12 mg/m2 (maximum 15 mg). A significant reduction in the rate of CNS relapse was observed in the group whose dose was based on age.
Because the CSF volume and turnover may decrease with age, a dose reduction may be indicated in elderly patients.
For the treatment of meningeal leukemia, intrathecal methotrexate may be given at intervals of 2 to 5 days. However, administration at intervals of less than 1 week may result in increased subacute toxicity. Methotrexate is administered until the cell count of the cerebrospinal fluid returns to normal. At this point one additional dose is advisable. For prophylaxis against meningeal leukemia, the dosage is the same as for treatment except for the intervals of administration. On this subject, it is advisable for the physician to consult the medical literature.
Untoward side effects may occur with any given intrathecal injection and are commonly neurological in character. Large doses may cause convulsions. Methotrexate given by the intrathecal route appears significantly in the systemic circulation and may cause systemic methotrexate toxicity. Therefore, systemic antileukemic therapy with the drug should be appropriately adjusted, reduced or discontinued. Focal leukemic involvement of the central nervous system may not respond to intrathecal chemotherapy and is best treated with radiotherapy.
Lymphomas
In Burkitt’s Tumor, Stages I-II, methotrexate has produced prolonged remissions in some cases. Recommended dosage is 10 to 25 mg/day orally for 4 to 8 days. In Stage III, methotrexate is commonly given concomitantly with other antitumor agents. Treatment in all stages usually consists of several courses of the drug interposed with 7 to 10 day rest periods. Lymphosarcomas in Stage III may respond to combined drug therapy with methotrexate given in doses of 0.625 to 2.5 mg/kg/daily.
Mycosis Fungoides (cutaneous T cell lymphoma)
Therapy with methotrexate as a single agent appears to produce clinical responses in up to 50% of patients treated. Dosage in early stages is usually 5 to 50 mg once weekly. Dose reduction or cessation is guided by patient response and hematologic monitoring. Methotrexate has also been administered twice weekly in doses ranging from 15 to 37.5 mg in patients who have responded poorly to weekly therapy. Combination chemotherapy regimens that include intravenous methotrexate administered at higher doses with leucovorin rescue have been utilized in advanced stages of the disease.
Osteosarcoma
An effective adjuvant chemotherapy regimen requires the administration of several cytotoxic chemotherapeutic agents. In addition to high-dose methotrexate with leucovorin rescue, these agents may include doxorubicin, cisplatin, and the combination of bleomycin, cyclophosphamide and dactinomycin (BCD) in the doses and schedule shown in the table below. The starting dose for high dose methotrexate treatment is 12 grams/m2. If this dose is not sufficient to produce a peak serum methotrexate concentration of 1,000 micromolar (10-3 mol/L) at the end of the methotrexate infusion, the dose may be escalated to 15 grams/m2 in subsequent treatments. If the patient is vomiting or is unable to tolerate oral medication, leucovorin is given IV or IM at the same dose and schedule.
|
||
| Drug* | Dose* | Treatment Week After Surgery |
| Methotrexate | 12 g/m2 IV as 4 hour infusion (starting dose) | 4,5,6,7,11,12,15,16,29,30,44,45 |
| Leucovorin | 15 mg orally every six hours for 10 doses starting at 24 hours after start of methotrexate infusion. | |
| Doxorubicin† as a single drug | 30 mg/m2/day IV x 3 days | 8,17 |
| Doxorubicin†
Cisplatin† | 50 mg/m2 IV 100 mg/m2 IV | 20,23,33,36 20,23,33,36 |
| Bleomycin†
Cyclophosphamide† Dactinomycin† | 15 units/m2 IV x 2 days 600 mg/m2 IV x 2 days 0.6 mg/m2 IV x 2 days | 2,13,26,39,42 2,13,26,39,42 2,13,26,39,42 |
When these higher doses of methotrexate are to be administered, the following safety guidelines should be closely observed.
GUIDELINES FOR METHOTREXATE THERAPY WITH LEUCOVORIN RESCUE
- Administration of methotrexate should be delayed until recovery if:
- the WBC count is less than 1500/microliter
- the neutrophil count is less than 200/microliter
- the platelet count is less than 75,000/microliter
- the serum bilirubin level is greater than 1.2 mg/dL
- the SGPT level is greater than 450 U
- mucositis is present, until there is evidence of healing
- persistent pleural effusion is present; this should be drained dry prior to infusion.
- Adequate renal function must be documented.
- a. Serum creatinine must be normal, and creatinine clearance must be greater than 60 mL/min, before initiation of therapy.
- b. Serum creatinine must be measured prior to each subsequent course of therapy. If serum creatinine has increased by 50% or more compared to a prior value, the creatinine clearance must be measured and documented to be greater than 60 mL/min (even if the serum creatinine is still within the normal range).
- Patients must be well hydrated, and must be treated with sodium bicarbonate for urinary alkalinization.
- a. Administer 1,000 mL/m2 of intravenous fluid over 6 hours prior to initiation of the methotrexate infusion. Continue hydration at 125 mL/m2/hr (3 liters/m2/day) during the methotrexate infusion, and for 2 days after the infusion has been completed.
- b. Alkalinize urine to maintain pH above 7.0 during methotrexate infusion and leucovorin calcium therapy. This can be accomplished by the administration of sodium bicarbonate orally or by incorporation into a separate intravenous solution.
- Repeat serum creatinine and serum methotrexate 24 hours after starting methotrexate and at least once daily until the methotrexate level is below 5x10-8 mol/L (0.05 micromolar).
- The table below provides guidelines for leucovorin calcium dosage based upon serum methotrexate levels. (See table below.‡)
Patients who experience delayed early methotrexate elimination are likely to develop nonreversible oliguric renal failure. In addition to appropriate leucovorin therapy, these patients require continuing hydration and urinary alkalinization, and close monitoring of fluid and electrolyte status, until the serum methotrexate level has fallen to below 0.05 micromolar and the renal failure has resolved. If necessary, acute, intermittent hemodialysis with a high-flux dialyzer may also be beneficial in these patients. - Some patients will have abnormalities in methotrexate elimination, or abnormalities in renal function following methotrexate administration, which are significant but less severe than the abnormalities described in the table below. These abnormalities may or may not be associated with significant clinical toxicity. If significant clinical toxicity is observed, leucovorin rescue should be extended for an additional 24 hours (total 14 doses over 84 hours) in subsequent courses of therapy. The possibility that the patient is taking other medications which interact with methotrexate (e.g., medications which may interfere with methotrexate binding to serum albumin, or elimination) should always be reconsidered when laboratory abnormalities or clinical toxicities are observed.
CAUTION DO NOT ADMINISTER LEUCOVORIN INTRATHECALLY.
Psoriasis
The patient should be fully informed of the risks involved and should be under constant supervision of the physician (See Information for Patients under PRECAUTIONS). Assessment of hematologic, hepatic, renal, and pulmonary function should be made by history, physical examination, and laboratory tests before beginning, periodically during, and before reinstituting methotrexate therapy (See PRECAUTIONS). Appropriate steps should be taken to avoid conception during methotrexate therapy (See PRECAUTIONS and CONTRAINDICATIONS).
All schedules should be continually tailored to the individual patient. An initial test dose may be given prior to the regular dosing schedule to detect any extreme sensitivity to adverse effects (See ADVERSE REACTIONS). Maximal myelosuppression usually occurs in 7 to 10 days.
Psoriasis: Recommended Starting Dose Schedules
- Weekly single oral, IM or IV dosage schedule: 10 to 25 mg per week until adequate response is achieved.
- Divided oral dosage schedule: 2.5 mg at 12 hour intervals for three doses.
Dosages in each schedule may be gradually adjusted to achieve optimal clinical response; 30 mg/week should not ordinarily be exceeded.
Once optimal clinical response has been achieved, each dosage schedule should be reduced to the lowest possible amount of drug and to the longest possible rest period. The use of methotrexate may permit the return to conventional topical therapy, which should be encouraged.
Handling and Disposal
Procedures for proper handling and disposal of anticancer drugs should be considered. Several guidelines on this subject have been published.1-7 There is no general agreement that all of the procedures recommended in the guidelines are necessary or appropriate.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Dilution Instructions
If dilutions are required, they should be made immediately prior to use, depending on the route of administration, using an appropriate sterile, preservative-free medium such as 0.9% Sodium Chloride Injection, USP. Methotrexate Injection, USP without preservative does not contain an antimicrobial agent and care must be taken to ensure the sterility of prepared solutions. For intrathecal injection dilute to a concentration of 1 mg/mL with an appropriate sterile, preservative-free medium such as 0.9% Sodium Chloride Injection, USP. Although the sterile solution has been shown to be stable for 24 hours at room temperature; since the product does not contain a preservative, any unused portion should be discarded immediately after use.
HOW SUPPLIED
Methotrexate Injection, USP is supplied in a single-dose vial (10 vials to a carton) containing 25 mg/mL of methotrexate as the base in the following package strengths:
NDC 55390-031-10 50 mg in 2 mL
NDC 55390-032-10 100 mg in 4 mL
NDC 55390-033-10 200 mg in 8 mL
NDC 55390-034-10 250 mg in 10 mL
Store product at controlled room temperature 15° to 30°C (59° to 86°F). Protect from light.
REFERENCES
- Recommendations for the Safe Handling of Parenteral Antineoplastic Drugs. NIH Publication No. 83-2621. For sale by the Superintendent of Documents, U.S. Government Printing Office, Washington, D.C. 20402.
- AMA Council Report. Guidelines for Handling Parenteral Antineoplastics. JAMA, March 15, 1985.
- National Study Commission on Cytotoxic Exposure-Recommendations for Handling Cytotoxic Agents. Available from Louis P. Jeffrey, Sc. D., Chairman, National Study Commission on Cytotoxic Exposure, Massachusetts College of Pharmacy and Allied Health Sciences, 179 Longwood Ave., Boston, Massachusetts 02115.
- Clinical Oncological Society of Australia; Guidelines and Recommendations for Safe-Handling of Antineoplastic Agents. Med J Australia 1: 426-428, 1983.
- Jones RB et al: Safe Handling of Chemotherapeutic Agents: A Report from the Mount Sinai Medical Center. CA-A Cancer Journal for Clinicians: Sept/Oct, 258-263, 1983.
- American Society of Hospital Pharmacists Technical Assistance Bulletin on Handling Cytotoxic and Hazardous Drugs. Am J Hosp Pharm 47:1033-1049, 1990.
- Controlling occupational exposure to hazardous drugs (OSHA Work-Practice Guidelines). AM J Health Syst Pharm 1996: 53: 1669-1685.
MANUFACTURED BY: MANUFACTURED FOR:
Ben Venue Laboratories, Inc. Bedford Laboratories™
Bedford, OH 44146 Bedford, OH 44146
January 2004 Div-MTX-P06
| Clinical Situation | Laboratory Findings | Leucovorin Dosage and Duration |
|
Normal Methotrexate Elimination | Serum methotrexate level approximately 10 micromolar at 24 hours after administration, 1 micromolar at 48 hours, and less than 0.2 micromolar at 72 hours. |
15 mg PO, IM or IV q 6 hours for 60 hours (10 doses starting at 24 hours after start of methotrexate infusion). |
|
Delayed Late Methotrexate Elimination |
Serum methotrexate level remaining above 0.2 micromolar at 72 hours, and more than 0.05 micromolar at 96 hours after administration. |
Continue 15 mg PO, IM or IV q 6 hours, until methotrexate level is less than 0.05 micromolar. |
|
Delayed Early Methotrexate Elimination and/or Evidence of Acute Renal Injury |
Serum methotrexate level of 50 micromolar or more at 24 hours, or 5 micromolar or more at 48 hours after administration, OR; a 100% or greater increase in serum creatinine level at 24 hours after methotrexate administration (e.g. an increase from 0.5 mg/dL to a level of 1 mg/dL or more). |
150 mg IV q 3 hours, until methotrexate level is less than 1 micro-molar; then 15 mg IV q 3 hours, until methotrexate level is less than 0.05 micromolar. |
| Methotrexate (Methotrexate) | |||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
Revised: 11/2006