FEIBA NF
-
anti-inhibitor coagulant complex
Baxter Healthcare Corporation
----------
FEIBA NF (Anti-Inhibitor Coagulant Complex)Nanofiltered and Vapor Heated
Lyophilized powder for solution
Intravenous
DESCRIPTION
FEIBA NF (Anti-Inhibitor Coagulant Complex), nanofiltered and vapor heated, is a freeze-dried sterile human plasma fraction with Factor VIII inhibitor bypassing activity. In vitro, FEIBA NF shortens the activated partial thromboplastin time (APTT) of plasma containing Factor VIII inhibitor. Factor VIII inhibitor bypassing activity is expressed in arbitrary units. One unit of activity is defined as that amount of FEIBA NF that shortens the APTT of a high titer Factor VIII inhibitor reference plasma to 50% of the blank value.
FEIBA NF contains Factors II, IX, and X, mainly non-activated, and Factor VII mainly in the activated form. The product contains approximately equal unitages of Factor VIII inhibitor bypassing activity and Prothrombin Complex Factors. In addition, 1–6 units of Factor VIII coagulant antigen (FVIII C:Ag) per mL are present. The preparation contains only traces of factors of the kinin generating system. It contains no heparin.
Reconstituted FEIBA NF contains 4 mg of trisodium citrate and 8 mg of sodium chloride per mL.
FEIBA NF is manufactured from large plasma pools of human plasma. Screening against potentially infectious agents begins with the donor selection process and continues throughout plasma collection and plasma preparation. Each individual plasma donation used in the manufacture of FEIBA NF is collected only at FDA approved blood establishments and is tested by FDA licensed serological tests for Hepatitis B Surface Antigen (HBsAg), and for antibodies to Human Immunodeficiency Virus (HIV-1/HIV-2) and Hepatitis C Virus (HCV) in accordance with U.S. Regulatory Requirements. As an additional safety measure, mini-pools of the plasma are tested for the presence of HIV-1 and HCV by FDA licensed Nucleic Acid Testing (NAT) and found negative. In addition, two dedicated and independent virus removal/inactivation steps have been integrated into the manufacturing process, namely 35 nm nanofiltration and a vapor heat treatment process. In addition, the DEAE-Sephadex adsorption contributes to the virus safety profile of FEIBA NF. Despite these measures, such products can still potentially transmit disease (see WARNINGS).
In vitro spiking studies have been used to validate the capability of the manufacturing process to remove and inactivate viruses. To establish the minimum applicable virus clearance capacity of the manufacturing process, these virus clearance studies were performed under extreme conditions (e.g. at minimum incubation times and temperatures below specifications for vapor-heat treatment). Virus clearance studies for FEIBA NF performed in accordance with good laboratory practices have demonstrated, that the manufacturing process of FEIBA NF ensures a high margin of safety with respect to adventitious viruses (Table 1).
|
|||||||
| Virus Type | Enveloped RNA | Enveloped DNA | Non-enveloped RNA | Non-enveloped DNA | |||
|
Virus Family |
Retroviridae |
Flaviviridae |
Herpesviridae |
Picornaviridae |
Parvoviridae |
||
| Virus* | HIV-1 | BVDV | WNV | PRV | HAV | B19V† | MMV |
| DEAE Sephadex Adsorption | 3.2 | 1.8 | n.d. | 2.5 | 1.5 | 1.7 | 1.2 |
| 35 nm Nanofiltration | > 5.3 | 2.1 | 4.7 | > 5.7 | 2.6 | 0.2‡ | 1.0 |
| Vapor-Heat Treatment | > 5.9 | > 5.6 | > 8.1 | > 6.7 | > 5.2 | 3.5 | 0.9‡ |
| Overall log reduction factor (ORF) | > 14.4 | > 9.5 | > 12.8 | > 14.9 | > 9.3 | 5.2 | 2.2 |
CLINICAL STUDIES
FEIBA NF is identical in formulation to FEIBA VH. Biochemical and preclinical studies have confirmed the comparability of FEIBA NF and FEIBA VH.
The safety and efficacy of FEIBA has been demonstrated by two prospective clinical trials.1,2 The first, conducted by Sixma and collaborators, was a randomized double-blind study comparing the effect of FEIBA and PROTHROMPLEX IMMUNO (a non-activated prothrombin complex concentrate) in 15 patients with hemophilia A and inhibitors to Factor VIII. A total of 150 bleeding episodes (primarily joint and musculoskeletal plus a few mucocutaneous) were treated. A single dose of 88 Units per kg of body weight was used uniformly for treatments with FEIBA . The study showed that, based on subjective patient evaluation, FEIBA was fully effective in 41.0% and partly effective in 24.6% of episodes (i.e. combined effectiveness of 65.6%), while PROTHROMPLEX was rated fully effective in 25.0% and partly effective in 21.4% of episodes (i.e. combined effectiveness of 46.4%).
The second study with FEIBA was a multicenter study conducted by Hilgartner et al. It was designed to evaluate the efficacy of FEIBA in the treatment of joint, mucous membrane, musculocutaneous and emergency bleeding episodes such as central nervous system hemorrhages and surgical bleedings. In 49 patients with inhibitor titers of greater than 5 Bethesda Units (from nine co-operating hemophilia centers), 489 single doses were given for the treatment of 165 bleeding episodes. The usual dosage was 50 Units per kg of body weight, repeated at 12-hour intervals (6-hour intervals in mucous membrane bleedings), if necessary. Bleeding was controlled in 153 episodes (93%). In 130 (78%) of the episodes, hemostasis was achieved with one or more infusions within 36 hours. Of these, 36% were controlled with one infusion within 12 hours. An additional 14% of episodes responded after more than 36 hours.
Of the 489 single doses, only 18 (3.7%) caused minor transient reactions in recipients. Out of 49 patients, 10 (20%) showed a rise in their inhibitor titers. In 5 of these patients (10%), the rise was tenfold or more. However, of these 10 patients, 3 had received Factor VIII or Factor IX concentrates within 2 weeks prior to treatment with FEIBA. These anamnestic rises have not been observed to interfere with the efficacy of FEIBA.
INDICATIONS AND USAGE
FEIBA NF (Anti-Inhibitor Coagulant Complex) is indicated for the control of spontaneous bleeding episodes or to cover surgical interventions in hemophilia A and hemophilia B patients with inhibitors.
In addition, the use of FEIBA has been described in a few non-hemophiliacs with acquired inhibitors to multiple coagulation factors3,4. One case has been reported where FEIBA was effective in a patient with von Willebrand’s disease with an inhibitor.
Clinical experience suggests that patients with a Factor VIII inhibitor titer of less than 5 B.U. may be successfully treated with Antihemophilic Factor. Patients with titers ranging between 5 and 10 B.U. may either be treated with Antihemophilic Factor or FEIBA NF. Cases with Factor VIII inhibitor titers greater than 10 B.U. have generally been refractory to treatment with Antihemophilic Factor.
|
|||
| Patient’s Inhibitor | Clinical Situation | ||
| Titer | Minor Bleeding | Major Bleeding | Surgery (Emergency) |
| less than 5 B.U. | AHF* | AHF | AHF |
| 5 to 10 B.U. | AHF | AHF | AHF |
| FEIBA NF | FEIBA NF | FEIBA NF | |
| more than 10 B.U. | FEIBA NF | FEIBA NF | FEIBA NF |
Inadequate response to treatment may result from an abnormal platelet count or impaired platelet function5-7 that were present before treatment with FEIBA nanofiltered and vapor-heated.
CONTRAINDICATIONS
The use of FEIBA NF is contraindicated in patients who are known to have a normal coagulation mechanism.
WARNINGS
FEIBA NF (Anti-Inhibitor Coagulant Complex), nanofiltered and vapor heated, is made from human plasma. Products made from plasma may contain infectious agents, such as viruses, that can cause disease. The risk that such products will transmit an infectious agent has been reduced by effective donor screening, testing for the presence of certain current virus infections, by inactivating and/or removing certain viruses. Despite these measures, such products can still potentially transmit disease. Because this product is made from human blood, it may carry a risk of transmitting infectious agents, e.g. viruses, and theoretically the Creutzfeldt-Jakob disease (CJD) agent. Individuals who receive infusions of blood or plasma products may develop signs and/or symptoms of some viral infections, particularly non-A, non- B hepatitis. ALL infections thought by a physician possibly to have been transmitted by this product should be reported by the physician or other health care provider to Baxter Healthcare Corporation, at 1-800-423-2862 (in the U.S.). The physician should discuss the risks and benefits of this product with the patient.
As with all human plasma products, allergic reactions, ranging from mild, short-term urticarial rashes to severe anaphylactoid reactions, have been seen. Administration of FEIBA NF should be discontinued immediately if such signs appear. Allergic reactions should be treated with antihistamines and glucocorticoids. Shock should be treated in the usual way.
Thromboembolic events may occur in the course of treatment with preparations containing the prothrombin complex. Thromboembolic events are a rare (4.05 to 8.24 events per 100,000 infusions8,9) but well recognized potential complication of FEIBA infusion, particularly following the administration of high doses and/or in patients with thrombotic risk factors.
In individual instances, myocardial infarction was found to occur after high doses and/or prolonged administration and/or in the presence of risk factors predisposing to myocardial infarction.
FEIBA NF must be used only in patients with circulating inhibitors to one or more coagulation factors and should not be used for the treatment of bleeding episodes resulting from coagulation factor deficiencies in the absence of inhibitors. It should not be given to patients with significant signs of disseminated intravascular coagulation (DIC) or fibrinolysis.
Infusion of FEIBA NF should not exceed single dosage of 100 units per kg of body weight and daily doses of 200 units per kg of body weight. Patients receiving more than 100 units per kg of body weight of FEIBA NF must be monitored for the development of DIC and/or symptoms of acute coronary ischemia.
High doses of FEIBA NF should be given only as long as absolutely necessary to stop bleeding.
Anamnestic responses with rise in Factor VIII inhibitor titer have been observed in 20% of the cases (see CLINICAL STUDIES).
PRECAUTIONS
General
If clinical signs of intravascular coagulation occur, which include changes in blood pressure, changes in pulse rate, respiratory distress, chest pain and/or cough, the infusion should be stopped promptly and appropriate diagnostic and therapeutic measures are to be initiated.
Laboratory indications of DIC are decreased fibrinogen, decreased platelet count, and/or presence of fibrin-fibrinogen degradation products (FDP). Other indications of DIC include significantly prolonged thrombin time, prothrombin time, or partial thromboplastin time.
It has been reported that FEIBA NF and antifibrinolytics have been given simultaneously without complications. It is recommended not to use antifibrinolytics until 12 hours after the administration of FEIBA NF.
Information for Patients
Some viruses, such as parvovirus B19 or hepatitis A, are particularly difficult to remove or inactivate at this time. Parvovirus B19 most seriously affects pregnant women or immune-compromised individuals. Symptoms of parvovirus B19 infection include fever, drowsiness, chills, and runny nose followed about two weeks later by a rash, and joint pain. Evidence of hepatitis A may include several days to weeks of poor appetite, tiredness, and low-grade fever followed by nausea, vomiting, and pain in the belly. Dark urine and a yellowed complexion are also common symptoms. Patients should be encouraged to consult their physician if such symptoms appear.
Non-Hemophilic Patients
Non-hemophilic patients with acquired inhibitors against Factors VIII, IX or XII may have both a bleeding tendency and an increased risk of thrombosis at the same time.
Laboratory Tests and Clinical Efficacy
Tests used to control efficacy such as APTT, WBCT, and TEG do not correlate with clinical improvement. For this reason, attempts at normalizing these values by increasing the dose of FEIBA NF may not be successful and are strongly discouraged because of the potential hazard of producing DIC by overdose.
Pregnancy Category C
Animal reproduction studies have not been conducted with FEIBA NF. It is also not known whether FEIBA NF can cause fetal harm when administered to a pregnant woman or can affect reproductive capacity. FEIBA NF should be given to a pregnant woman only if clearly needed.
Pediatric Use
No data are available regarding the use of FEIBA NF in newborns.
ADVERSE REACTIONS
The following adverse reactions have been identified during post approval use of FEIBA: Myocardial infarction, disseminated intravascular coagulopathy, injection site pain, anaphylactic reaction, hypersensitivity, urticaria, blood pressure decreased, hypoaesthesia, hypoaesthesia facial, embolism.
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
OVERDOSAGE
Overdosage of FEIBA NF may increase the risk of thromboembolism, DIC or myocardial infarction (See WARNINGS).
DOSAGE AND ADMINISTRATION
(See under For Intravenous Injection/Infusion).
Clinical trials1,2 demonstrated that the response to treatment with FEIBA may differ from patient to patient with no correlation to the patient’s inhibitor titer. Response may also vary between different types of hemorrhage (e.g. joint hemorrhage vs. CNS hemorrhage). As a general guideline, a dosage range of 50 to 100 Units of FEIBA NF, per kg of body weight is recommended. However, care should be taken to distinguish between the following four indications, all of which have undergone careful clinical evaluation:
Joint Hemorrhage
In joint hemorrhage, a dose of 50 units per kg of body weight is recommended at 12-hour intervals, which may be increased to doses of 100 units per kg of body weight at 12-hour intervals.
Treatment should be continued until clear signs of clinical improvement appear, such as relief of pain, reduction of swelling or mobilization of the joint.
Mucous Membrane Bleeding
A dose of 50 units per kg of body weight is recommended to be given at 6-hour intervals under careful monitoring (visible bleeding site, repeated measurements of the patient’s hematocrit). If hemorrhage does not stop, the dose may be increased to 100 units per kg of body weight at 6-hour intervals. Two such administrations or 200 units per kg of body weight a day should not be exceeded.
Soft Tissue Hemorrhage
For serious soft tissue bleeding, such as retroperitoneal bleeding, doses of 100 units per kg of body weight at 12-hour intervals are recommended. A daily dosage of 200 units per kg of body weight should not be exceeded.
Other Severe Hemorrhages
Severe hemorrhages, such as CNS bleedings have been effectively treated with doses of 100 units per kg of body weight at 12-hour intervals. Sometimes, FEIBA NF may be indicated at 6-hour intervals until clear clinical improvement is achieved.
Reconstitution
- Warm the unopened vial containing Sterile Water for Injection (diluent) to room temperature (not above 37°C, 98°F).
- Remove caps from the concentrate and diluent vials to expose central portions of the rubber stoppers.
- Cleanse exposed surface of the rubber stoppers with germicidal solution and allow to dry.
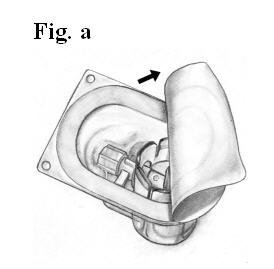
- Open the package of BAXJECT device by peeling away the lid without touching the inside (Fig. a).
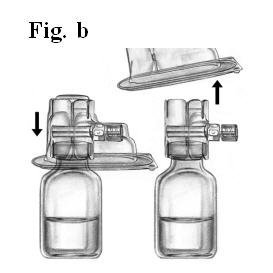
- Do not remove the device from the package. Turn the package over and insert the plastic spike through diluent stopper (Fig. b).
- Grip the package at its edge and pull the package off the device (Fig. b).
- Turn the system over, so that the vial is on top. Quickly insert the other plastic spike into the FEIBA NF stopper (Fig. c). The vacuum will draw the diluent into the FEIBA NF vial. Please make sure that the connection of the two vials should be done expeditiously to close the open fluid pathway created by the first insertion of the spike to the diluent vial!
- Swirl gently until FEIBA NF is completely dissolved.
Do not refrigerate after reconstitution!
After complete reconstitution of FEIBA NF its injection or infusion should be commenced as promptly as practicable, but must be completed within three hours following reconstitution. The solution must be given by intravenous injection or intravenous drip infusion.
Rate of Administration:
The maximum injection or infusion rate must not exceed 2 units per kg of body weight per minute. For a patient with a body weight of 75 kg, this corresponds to an infusion rate of 2.5 - 7.5 mL per minute depending on the number of units per vial (see label on vial).
For Intravenous Injection or Infusion:
- After reconstituting the concentrate as described under Reconstitution , parenteral drug products should be inspected for particulate matter and discoloration prior to administration, whenever solution and container permit. Plastic Luer lock syringes are recommended for use with this product since protein such as FEIBA NF tends to stick to the surface of all-glass syringes.
- Turn the BAXJECT device handle down towards the FEIBA NF concentrate vial and remove the cap attached to the syringe connection of the BAXJECT device (Fig. d).
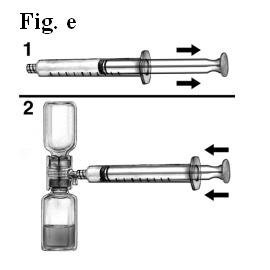
- Draw air into the syringe, connect the syringe to the BAXJECT device, inject air into the concentrate vial (Fig. e).
- While keeping the syringe plunger in place, turn the system upside down (concentrate vial now on top). Draw the concentrate into the syringe by pulling the plunger back slowly (Fig. f).
- Turn the BAXJECT handle to its original position (facing side way).
- Disconnect the syringe, attach a suitable needle and inject or infuse intravenously as instructed under Rate of Administration.



HOW SUPPLIED
FEIBA NF is available in single-dose vials in the following nominal dosage strengths:
Blue - 500 Units per vial (NDC 64193-223-02)
Green - 1000 Units per vial (NDC 64193-224-02)
Purple - 2500 Units per vial (NDC 64193-225-02)
The number of Units of Factor VIII inhibitor bypassing activity is stated on the label of each vial.
FEIBA NF is packaged with a suitable volume (20 mL or 50 mL) of Sterile Water for Injection, U.S.P., one BAXJECT Needleless Transfer Device, and one Package Insert.
Certain components of the packaging material contain Dry Natural Rubber Latex.
STORAGE
Store at refrigerated temperature (2° to 8°C, 35° to 46°F). Within the indicated shelf life, the product may be stored at room temperature (not exceeding 25°C, 77°F) for up to 6 months.
After storage at room temperature, the product must not be returned to the refrigerator.
Please note: If you transfer the product from the refrigerator to room temperature, it expires at the end of the 6 months period or at the end of shelf life, whatever comes earlier.
Record the date on the package prior to shifting the product at room temperature.
Avoid freezing, which may damage the diluent vial.
REFERENCES
- Sjamsoedin L. J. M., Heijnen L., Mauser-Bunschoten E. P., van Geijlswijk J. L., van Houwelingen H., van Asten P., Sixma J. J.: The Effect of Activated Prothrombin-Complex Concentrate (FEIBA) on Joint and Muscle Bleeding in Patients with Hemophilia A and Antibodies to Factor VIII. The New Engl. J. of Med. 305: 717, 1981.
- Hilgartner M. W., Knatterud G. AND THE FEIBA STUDY GROUP: The Use of Factor-Eight-Inhibitor-By-Passing-Activity (FEIBA IMMUNO) Product for Treatment of Bleeding Episodes in Hemophiliacs with Inhibitors. Blood 61: 36, 1983.
- Thomas T., William. H., Williams Y., Hunt J.: FEIBA in Haemophiliacs with Factor VIII Inhibitor. Brit. Med. J. 1: 52, 1977.
- Rolovic Z., Elezovic I., Obrenovic B.: Life-Threatening Bleeding Due to an Acquired Inhibitor to Factor XII–XI Successfully Treated with Feiba: Proceedings of Joint Meeting of the 18th Congress of the International Society of Hematology and 16th Congress of the International Society of Blood Transfusion, Montreal. Abstract. P-703, 1980.
- Vermylen J., Schetz J., Semeraro N., Mertens F., Verstraete M.: Evidence that ‘Activated’ Prothrombin Concentrates Enhance Platelet Coagulant Activity. Brit. J. Haematol. 38: 235, 1978.
- Semeraro N., Vermylen J.: Evidence that Washed Human Platelets Possess Factor-X Activator Activity. Brit. J. Haematol. 36: 107, 1977.
- Wensley R. T.: General Summary of the Use of FEIBA in Haemophiliacs with Inhibitors to FVIII. Presentation at the Second Workshop on Factor VIII Inhibitor Patients, Vienna, 1979.
- Aledort LM: Comparative Thrombotic Event Incidence After Infusion of Recombinant Factor VIIa Versus Factor VIII Inhibitor Bypass Activity. J Thromb Haemost 2004; 2 : 1700–08.
- Ehrlich HJ, Henzl MJ and Gomperts ED: Safety of Factor VIII Inhibitor Bypass Activity (FEIBA): 10-year Compilation of Thrombotic Adverse Events. Haemophilia (2002), 8, 83-90.
To enroll in the confidential, Industry-wide Patient Notification System,
Call 1-888-UPDATE U(1-888-873-2838).
Baxter, FEIBA and PROTHROMPLEX are trademarks of Baxter AG, Vienna, Austria. Baxter, FEIBA and BAXJECT are trademarks of Baxter International Inc., registered in the U.S. Patent and Trademark Office.
Baxter Healthcare Corporation
Westlake Village, CA 91362 USA
U.S. License No. 140
Revised May 2009
U.S. Pat. Nos. 4, 640, 834
Principle Display Panel

FEIBA NF 500U vial label
20 mL size, dried NDC 64193-323-01
Anti-Inhibitor Coagulant Complex
FEIBA NF
Nanofiltered & Vapor Heated
Baxter (Logo)
For Intravenous Use Only Contains no preservative
Dosage & Administration: See accompanying package insert
Storage: Store between 2° - 8°C (35-46°F). May be stored at room temperature not to exceed 25°C (77°F) for up to 6 months
Reconstitution: Use 20 mL sWFI, U.S.P.; Keep at room temperature not to exceed 25°C (77°F); Use within 3 hours of reconstitution
Rx only

FEIBA NF 500U unit carton
20 mL size, dried NDC 64193-223-02
Anti-Inhibitor Coagulant Complex
FEIBA NF
Nanofiltered & Vapor Heated
Baxter (Logo)
For Intravenous Use Only Contains no preservative
Dosage & Administration: See accompanying package insert
Storage: Store between 2° - 8°C (35-46°F). Within indicated shelf life, product may be stored at room temperature not to exceed 25°C (77°F) for up to 6 months. Do not return to refrigerator. Record date on the package prior to storing at room temperature
Please Note: Product transferred from the refrigerator to room temperature expires at the end of 6 months or end of shelf life, whichever comes firsg
Reconstitution: Use 20 mL sWFI, U.S.P.; Keep at room temperature not to exceed 25°C (77°F); Use within 3 hours of reconstitution
Do Not Freeze Rx only
Includes BAXJECT Needleless Transfer Device
| FEIBA
NF
anti-inhibitor coagulant complex kit |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| BLA | BLA101447 | 03/31/2010 | |
| FEIBA
NF
anti-inhibitor coagulant complex kit |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| BLA | BLA101447 | 03/31/2010 | |
| FEIBA
NF
anti-inhibitor coagulant complex kit |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| BLA | BLA101447 | 03/31/2010 | |
| Labeler - Baxter Healthcare Corporation (085206634) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Baxter Innovations GmbH | 300178716 | MANUFACTURE | |