SPORANOX
-
itraconazole solution
Ortho-McNeil-Janssen Pharmaceuticals Inc.
----------
SPORANOX®(itraconazole)
Oral Solution
Boxed Warnings
Congestive Heart Failure: When itraconazole was administered intravenously to dogs and healthy human volunteers, negative inotropic effects were seen. If signs or symptoms of congestive heart failure occur during administration of SPORANOX® (itraconazole) Oral Solution, continued SPORANOX® use should be reassessed. (See CLINICAL PHARMACOLOGY: Special Populations, CONTRAINDICATIONS, WARNINGS, PRECAUTIONS: Drug Interactions and ADVERSE REACTIONS: Post-marketing Experience for more information.)
Drug Interactions: Coadministration of cisapride, pimozide, quinidine, dofetilide, or levacetylmethadol (levomethadyl) with SPORANOX® (itraconazole) Capsules, Injection or Oral Solution is contraindicated. SPORANOX®, a potent cytochrome P450 3A4 isoenzyme system (CYP3A4) inhibitor, may increase plasma concentrations of drugs metabolized by this pathway. Serious cardiovascular events, including QT prolongation, torsades de pointes, ventricular tachycardia, cardiac arrest, and/or sudden death have occurred in patients using cisapride, pimozide, levacetylmethadol (levomethadyl), or quinidine concomitantly with SPORANOX® and/or other CYP3A4 inhibitors. (See CONTRAINDICATIONS, WARNINGS, and PRECAUTIONS: Drug Interactions for more information.)
DESCRIPTION
SPORANOX® is the brand name for itraconazole, a synthetic triazole antifungal agent. Itraconazole is a 1:1:1:1 racemic mixture of four diastereomers (two enantiomeric pairs), each possessing three chiral centers. It may be represented by the following structural formula and nomenclature:
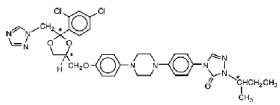
(±)-1-[(R*)-sec-butyl]-4-[p-[4-[p-[[(2R*,4S*)-2-(2,4-dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]-1-piperazinyl]phenyl]-∆2-1,2,4-triazolin-5-one mixture with (±)-1-[(R*)-sec-butyl]-4-[p-[4-[p-[[(2S*,4R*)-2-(2,4-dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]-1-piperazinyl]phenyl]-∆2-1,2,4-triazolin-5-one
(±)-1-[(RS)-sec-butyl]-4-[p-[4-[p-[[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]-1-piperazinyl]phenyl]-∆2-1,2,4-triazolin-5-one.
Itraconazole has a molecular formula of C35H38Cl2N8O4 and a molecular weight of 705.64. It is a white to slightly yellowish powder. It is insoluble in water, very slightly soluble in alcohols, and freely soluble in dichloromethane. It has a pKa of 3.70 (based on extrapolation of values obtained from methanolic solutions) and a log (n-octanol/water) partition coefficient of 5.66 at pH 8.1.
SPORANOX® (itraconazole) Oral Solution contains 10 mg of itraconazole per mL, solubilized by hydroxypropyl-β-cyclodextrin (400 mg/mL) as a molecular inclusion complex. SPORANOX® Oral Solution is clear and yellowish in color with a target pH of 2. Other ingredients are hydrochloric acid, propylene glycol, purified water, sodium hydroxide, sodium saccharin, sorbitol, cherry flavor 1, cherry flavor 2 and caramel flavor.
CLINICAL PHARMACOLOGY
Pharmacokinetics and Metabolism:
NOTE: The plasma concentrations reported below were measured by high-performance liquid chromatography (HPLC) specific for itraconazole. When itraconazole in plasma is measured by a bioassay, values reported may be higher than those obtained by HPLC due to the presence of the bioactive metabolite, hydroxyitraconazole. (See MICROBIOLOGY.)
The absolute bioavailability of itraconazole administered as a non-marketed solution formulation under fed conditions was 55% in 6 healthy male volunteers. However, the bioavailability of SPORANOX® (itraconazole) Oral Solution is increased under fasted conditions reaching higher maximum plasma concentrations (Cmax) in a shorter period of time. In 27 healthy male volunteers, the steady-state area under the plasma concentration versus time curve (AUC0-24h) of itraconazole (SPORANOX® Oral Solution, 200 mg daily for 15 days) under fasted conditions was 131 ± 30% of that obtained under fed conditions. Therefore, unlike SPORANOX® Capsules, it is recommended that SPORANOX® Oral Solution be administered without food. Presented in the table below are the steady-state (Day 15) pharmacokinetic parameters for itraconazole and hydroxyitraconazole (SPORANOX® Oral Solution) under fasted and fed conditions:
| Itraconazole | Hydroxyitraconazole | |||
|---|---|---|---|---|
| Fasted | Fed | Fasted | Fed | |
| *mean ± standard deviation | ||||
| Cmax (ng/mL) | 1963 ± 601* | 1435 ± 477 | 2055 ± 487 | 1781 ± 397 |
| Tmax (hours) | 2.5 ± 0.8 | 4.4 ± 0.7 | 5.3 ± 4.3 | 4.3 ± 1.2 |
| AUC0-24h
(ng·h/mL) | 29271 ± 10285 | 22815 ± 7098 | 45184 ± 10981 | 38823 ± 8907 |
| t1/2 (hours) | 39.7 ± 13 | 37.4 ± 13 | 27.3 ± 13 | 26.1 ± 10 |
The bioavailability of SPORANOX® Oral Solution relative to SPORANOX® Capsules was studied in 30 healthy male volunteers who received 200 mg of itraconazole as the oral solution and capsules under fed conditions. The AUC0-∞ from SPORANOX® Oral Solution was 149 ± 68% of that obtained from SPORANOX® Capsules; a similar increase was observed for hydroxyitraconazole. In addition, a cross study comparison of itraconazole and hydroxyitraconazole pharmacokinetics following the administration of single 200 mg doses of SPORANOX® Oral Solution (under fasted conditions) or SPORANOX® Capsules (under fed conditions) indicates that when these two formulations are administered under conditions which optimize their systemic absorption, the bioavailability of the solution relative to capsules is expected to be increased further. Therefore, it is recommended that SPORANOX® Oral Solution and SPORANOX® Capsules not be used interchangeably. The following table contains pharmacokinetic parameters for itraconazole and hydroxyitraconazole following single 200 mg doses of SPORANOX® Oral Solution (n=27) or SPORANOX® Capsules (n=30) administered to healthy male volunteers under fasted and fed conditions, respectively:
| Itraconazole | Hydroxyitraconazole | |||
|---|---|---|---|---|
| Oral Solution fasted | Capsules fed | Oral Solution fasted | Capsules fed | |
| *mean standard deviation | ||||
| Cmax (ng/mL) | 544 ± 213* | 302 ± 119 | 622 ± 116 | 504 ± 132 |
| Tmax (hours) | 2.2 ± 0.8 | 5 ± 0.8 | 3.5 ± 1.2 | 5 ± 1 |
| AUC0-24h
(ng·h/mL) | 4505 ± 1670 | 2682 ± 1084 | 9552 ± 1835 | 7293 ± 2144 |
The plasma protein binding of itraconazole is 99.8% and that of hydroxyitraconazole is 99.5%. Following intravenous administration, the volume of distribution of itraconazole averaged 796 ± 185 L.
Itraconazole is metabolized predominately by the cytochrome P450 3A4 isoenzyme system (CYP3A4), resulting in the formation of several metabolites, including hydroxyitraconazole, the major metabolite. Results of a pharmacokinetics study suggest that itraconazole may undergo saturable metabolism with multiple dosing. Fecal excretion of the parent drug varies between 3-18% of the dose. Renal excretion of the parent drug is less than 0.03% of the dose. About 40% of the dose is excreted as inactive metabolites in the urine. No single excreted metabolite represents more than 5% of a dose. Itraconazole total plasma clearance averaged 381 ± 95 mL/minute following intravenous administration. (See CONTRAINDICATIONS and PRECAUTIONS: Drug Interactions for more information.)
Special Populations:
Pediatrics:
The pharmacokinetics of SPORANOX® Oral Solution were studied in 26 pediatric patients requiring systemic antifungal therapy. Patients were stratified by age: 6 months to 2 years (n=8), 2 to 5 years (n=7) and 5 to 12 years (n=11), and received itraconazole oral solution 5 mg/kg once daily for 14 days. Pharmacokinetic parameters at steady-state (Day 14) were not significantly different among the age strata and are summarized in the table below for all 26 patients:
| Itraconazole | Hydroxyitraconazole | |
|---|---|---|
| *mean ± standard deviation | ||
| Cmax (ng/mL) | 582.5 ± 382.4* | 692.4 ± 355.0 |
| Cmin (ng/mL) | 187.5 ± 161.4 | 403.8 ± 336.1 |
| AUC0-24h (ng·h/mL) | 7706.7 ± 5245.2 | 13356.4 ± 8942.4 |
| t1/2 (hours) | 35.8 ± 35.6 | 17.7 ± 13.0 |
Renal Insufficiency:
Limited data are available on the use of oral itraconazole in patients with renal impairment. A pharmacokinetic study using a single 200-mg dose of itraconazole (four 50-mg capsules) was conducted in three groups of patients with renal impairment (uremia: n=7; hemodialysis: n=7; and continuous ambulatory peritoneal dialysis: n=5). In uremic subjects with a mean creatinine clearance of 13 mL/min. × 1.73 m2, the exposure, based on AUC, was slightly reduced compared with normal population parameters. This study did not demonstrate any significant effect of hemodialysis or continuous ambulatory peritoneal dialysis on the pharmacokinetics of itraconazole (Tmax, Cmax, and AUC0-8). Plasma concentration-versus-time profiles showed wide intersubject variation in all three groups. Caution should be exercised when the drug is administered in this patient population. (See PRECAUTIONS and DOSAGE AND ADMINISTRATION.)
Hepatic Insufficiency:
Itraconazole is predominantly metabolized in the liver. Patients with impaired hepatic function should be carefully monitored when taking itraconazole. A pharmacokinetic study using a single oral 100 mg capsule dose of itraconazole was conducted in 6 healthy and 12 cirrhotic subjects. A statistically significant reduction in mean Cmax (47%) and a twofold increase in the elimination half-life (37 ± 17 hours vs. 16 ± 5 hours) of itraconazole were noted in cirrhotic subjects compared with healthy subjects. However, overall exposure to itraconazole, based on AUC, was similar in cirrhotic patients and in healthy subjects. The prolonged elimination half-life of itraconazole observed in the single oral dose clinical trial with itraconazole capsules in cirrhotic patients should be considered when deciding to initiate therapy with other medications metabolized by CYP3A4. Data are not available in cirrhotic patients during long-term use of itraconazole. (See BOX WARNING, CONTRAINDICATIONS, PRECAUTIONS: Drug Interactions and DOSAGE AND ADMINISTRATION.)
Decreased Cardiac Contractility:
When itraconazole was administered intravenously to anesthetized dogs, a dose-related negative inotropic effect was documented. In a healthy volunteer study of SPORANOX® Injection (intravenous infusion), transient, asymptomatic decreases in left ventricular ejection fraction were observed using gated SPECT imaging; these resolved before the next infusion, 12 hours later. If signs or symptoms of congestive heart failure appear during administration of SPORANOX® Oral Solution, monitor carefully and consider other treatment alternatives which may include discontinuation of SPORANOX® Oral Solution administration. (See WARNINGS, PRECAUTIONS: Drug Interactions and ADVERSE REACTIONS: Post-marketing Experience for more information.)
Cystic Fibrosis:
Seventeen cystic fibrosis patients, ages 7 to 28 years old, were administered itraconazole oral solution 2.5 mg/kg b.i.d. for 14 days in a pharmacokinetic study. Sixteen patients completed the study. Steady state trough concentrations >250 ng/mL were achieved in 6 out of 11 patients ≥16 years of age but in none of the 5 patients <16 years of age. Large variability was observed in the pharmacokinetic data (%CV for trough concentrations = 98% and 70% for ≥16 and <16 years, respectively; %CV for AUC = 75% and 58% for ≥16 and <16 years, respectively). If a patient with cystic fibrosis does not respond to SPORANOX® Oral Solution, consideration should be given to switching to alternative therapy.
MICROBIOLOGY
Mechanism of Action:
In vitro studies have demonstrated that itraconazole inhibits the cytochrome P450-dependent synthesis of ergosterol, which is a vital component of fungal cell membranes.
Activity In Vitro and In Vivo:
Itraconazole exhibits in vitro activity against Blastomyces dermatitidis, Histoplasma capsulatum, Histoplasma duboisii, Aspergillus flavus, Aspergillus fumigatus, Candida albicans, and Cryptococcus neoformans. Itraconazole also exhibits varying in vitro activity against Sporothrix schenckii, Trichophyton species, Candida krusei, and other Candida species.
Candida krusei, Candida glabrata and Candida tropicalis are generally the least susceptible Candida species, with some isolates showing unequivocal resistance to itraconazole in vitro. Itraconazole is not active against Zygomycetes (e.g., Rhizopus spp., Rhizomucor spp., Mucor spp. and Absidia spp.), Fusarium spp., Scedosporium spp. and Scopulariopsis spp.
The bioactive metabolite, hydroxyitraconazole, has not been evaluated against Histoplasma capsulatum, Blastomyces dermatitidis, Zygomycete, Fusarium spp., Scedosporium spp. and Scopulariopsis spp. Correlation between minimum inhibitory concentration (MIC) results in vitro and clinical outcome has yet to be established for azole antifungal agents.
Itraconazole administered orally was active in a variety of animal models of fungal infection using standard laboratory strains of fungi. Fungistatic activity has been demonstrated against disseminated fungal infections caused by Blastomyces dermatitidis, Histoplasma duboisii, Aspergillus fumigatus, Coccidioides immitis, Cryptococcus neoformans, Paracoccidioides brasiliensis, Sporothrix schenckii, Trichophyton rubrum, and Trichophyton mentagrophytes.
Itraconazole administered at 2.5 mg/kg and 5 mg/kg via the oral and parenteral routes increased survival rates and sterilized organ systems in normal and immunosuppressed guinea pigs with disseminated Aspergillus fumigatus infections. Oral itraconazole administered daily at 40 mg/kg and 80 mg/kg increased survival rates in normal rabbits with disseminated disease and in immunosuppressed rats with pulmonary Aspergillus fumigatus infection, respectively. Itraconazole has demonstrated antifungal activity in a variety of animal models infected with Candida albicans and other Candida species.
Resistance:
Isolates from several fungal species with decreased susceptibility to itraconazole have been isolated in vitro and from patients receiving prolonged therapy.
Several in vitro studies have reported that some fungal clinical isolates, including Candida species, with reduced susceptibility to one azole antifungal agent may also be less susceptible to other azole derivatives. The finding of cross-resistance is dependent on a number of factors, including the species evaluated, its clinical history, the particular azole compounds compared, and the type of susceptibility test that is performed. The relevance of these in vitro susceptibility data to clinical outcome remains to be elucidated.
Candida krusei, Candida glabrata and Candida tropicalis are generally the least susceptible Candida species, with some isolates showing unequivocal resistance to itraconazole in vitro.
Itraconazole is not active against Zygomycetes (e.g., Rhizopus spp., Rhizomucor spp., Mucor spp. and Absidia spp.), Fusarium spp., Scedosporium spp. and Scopulariopsis spp.
Studies (both in vitro and in vivo) suggest that the activity of amphotericin B may be suppressed by prior azole antifungal therapy. As with other azoles, itraconazole inhibits the 14C-demethylation step in the synthesis of ergosterol, a cell wall component of fungi. Ergosterol is the active site for amphotericin B. In one study the antifungal activity of amphotericin B against Aspergillus fumigatus infections in mice was inhibited by ketoconazole therapy. The clinical significance of test results obtained in this study is unknown.
CLINICAL STUDIES
Empiric Therapy in Febrile Neutropenic Patients:
An open randomized trial compared the efficacy and safety of itraconazole (intravenous followed by oral solution) with amphotericin B for empiric therapy in 384 febrile, neutropenic patients with hematologic malignancies who had suspected fungal infections. Patients received either itraconazole (injection, 200 mg b.i.d. for 2 days followed by 200 mg once daily for up to 14 days, followed by oral solution, 200 mg b.i.d.) or amphotericin B (total daily dose of 0.7-1.0 mg/kg body weight). The longest treatment duration was 28 days. An outcome assignment of "success" required (a) patient survival with resolution of fever and neutropenia within 28 days of treatment, (b) absence of emergent fungal infections, (c) no discontinuation of therapy due to toxicity or lack of efficacy, and (d) treatment for three or more days. The success rate using an intent-to-treat analysis was 47% for the itraconazole group and 38% for the amphotericin B arm.
| Efficacy Parameters | SPORANOX®
N=179 (%) | Amphotericin B
N=181 (%) |
|---|---|---|
| *Treatment duration ≤ 3 days (including patients who died within 3 days, withdrew because of adverse events or were deemed ineligible due to a confirmed pre-treatment infection). | ||
| Success | 84 (47%) | 68 (38%) |
| Unevaluable* | 24 (13%) | 44 (24%) |
| Failure | 71 (40%) | 69 (38%) |
| Reason for Failure | ||
| Intolerance after > 3 days of antifungal medication | 12 | 37 |
| Persistent fever | 20 | 7 |
| Change in antifungal medication due to fever | 13 | 1 |
| Emergent fungal infection | 10 | 9 |
| Documented bacterial or viral infection | 7 | 8 |
| Insufficient response | 6 | 5 |
| Deterioration of signs and symptoms | 2 | 0 |
| Death after > 3 days antifungal medication | 1 | 2 |
| Resolution of fever | 131 (73%) | 127 (70%) |
| Survival | 161 (90%) | 156 (86%) |
Oropharyngeal Candidiasis:
Two randomized, controlled studies for the treatment of oropharyngeal candidiasis have been conducted (total n=344). In one trial, clinical response to either 7 or 14 days of itraconazole oral solution, 200 mg/day, was similar to fluconazole tablets and averaged 84% across all arms. Clinical response in this study was defined as cured or improved (only minimal signs and symptoms with no visible lesions). Approximately 5% of subjects were lost to follow-up before any evaluations could be performed. Response to 14 days therapy of itraconazole oral solution was associated with a lower relapse rate than 7 days of itraconazole therapy. In another trial, the clinical response rate (defined as cured or improved) for itraconazole oral solution was similar to clotrimazole troches and averaged approximately 71% across both arms, with approximately 3% of subjects lost to follow-up before any evaluations could be performed. Ninety-two percent of the patients in these studies were HIV seropositive.
In an uncontrolled, open-label study of selected patients clinically unresponsive to fluconazole tablets (n=74, all patients HIV seropositive), patients were treated with itraconazole oral solution 100 mg b.i.d. (Clinically unresponsive to fluconazole in this study was defined as having received a dose of fluconazole tablets at least 200 mg/day for a minimum of 14 days.) Treatment duration was 14-28 days based on response. Approximately 55% of patients had complete resolution of oral lesions. Of patients who responded and then entered a follow-up phase (n=22), all relapsed within 1 month (median 14 days) when treatment was discontinued. Although baseline endoscopies had not been performed, several patients in this study developed symptoms of esophageal candidiasis while receiving therapy with itraconazole oral solution. Itraconazole oral solution has not been directly compared to other agents in a controlled trial of similar patients.
Esophageal Candidiasis:
A double-blind randomized study (n=119, 111 of whom were HIV seropositive) compared itraconazole oral solution (100 mg/day) to fluconazole tablets (100 mg/day). The dose of each was increased to 200 mg/day for patients not responding initially. Treatment continued for 2 weeks following resolution of symptoms, for a total duration of treatment of 3-8 weeks. Clinical response (a global assessment of cured or improved) was not significantly different between the two study arms, and averaged approximately 86% with 8% lost to follow-up. Six of 53 (11%) itraconazole-treated patients and 12/57 (21%) fluconazole-treated patients were escalated to the 200 mg dose in this trial. Of the subgroup of patients who responded and entered a follow-up phase (n=88), approximately 23% relapsed across both arms within 4 weeks.
INDICATIONS AND USAGE
SPORANOX® (itraconazole) Injection/Oral Solution is indicated for empiric therapy of febrile neutropenic patients with suspected fungal infections. (NOTE: In a comparative trial, the overall response rate for itraconazole-treated subjects was higher than for amphotericin B-treated subjects. However, compared to amphotericin B-treated subjects, a larger number of itraconazole-treated subjects discontinued treatment due to persistent fever and a change in antifungal medication due to fever. Whereas, a larger number of amphotericin B-treated subjects discontinued due to drug intolerance. (See CLINICAL STUDIES section.)
SPORANOX® (itraconazole) Oral Solution is also indicated for the treatment of oropharyngeal and esophageal candidiasis.
(See CLINICAL PHARMACOLOGY: Special Populations, WARNINGS, and ADVERSE REACTIONS: Post-marketing Experience for more information.)
CONTRAINDICATIONS
Congestive Heart Failure:
SPORANOX® (itraconazole) Oral Solution should not be administered to patients with evidence of ventricular dysfunction such as congestive heart failure (CHF) or a history of CHF except for the treatment of life-threatening or other serious infections. (See CLINICAL PHARMACOLOGY: Special Populations, WARNINGS, PRECAUTIONS: Drug Interactions-Calcium Channel Blockers, and ADVERSE REACTIONS: Post-marketing Experience.)
Drug Interactions:
Concomitant administration of SPORANOX® (itraconazole) Capsules, Injection, or Oral Solution and certain drugs metabolized by the cytochrome P450 3A4 isoenzyme system (CYP3A4) may result in increased plasma concentrations of those drugs, leading to potentially serious and/or life-threatening adverse events. Cisapride, oral midazolam, nisoldipine, pimozide, quinidine, dofetilide, triazolam and levacetylmethadol (levomethadyl) are contraindicated with SPORANOX®. HMG CoA-reductase inhibitors metabolized by CYP3A4, such as lovastatin and simvastatin, are also contraindicated with SPORANOX®. Ergot alkaloids metabolized by CYP3A4 such as dihydroergotamine, ergometrine (ergonovine), ergotamine and methylergometrine (methylergonovine) are contraindicated with SPORANOX®. (See BOX WARNING, and PRECAUTIONS: Drug Interactions.)
SPORANOX® is contraindicated for patients who have shown hypersensitivity to itraconazole or its excipients. There is no information regarding cross-hypersensitivity between itraconazole and other azole antifungal agents. Caution should be used when prescribing SPORANOX® to patients with hypersensitivity to other azoles.
WARNINGS
SPORANOX® (itraconazole) Oral Solution and SPORANOX® Capsules should not be used interchangeably. Only SPORANOX® Oral Solution has been demonstrated effective for oral and/or esophageal candidiasis. SPORANOX® Oral Solution contains the excipient hydroxypropyl-β-cyclodextrin which produced pancreatic adenocarcinomas in a rat carcinogenicity study. These findings were not observed in a similar mouse carcinogenicity study. The clinical relevance of these findings is unknown. (See Carcinogenesis, Mutagenesis, and Impairment of Fertility.)
Hepatic Effects:
SPORANOX®has been associated with rare cases of serious hepatotoxicity, including liver failure and death. Some of these cases had neither pre-existing liver disease nor a serious underlying medical condition and some of these cases developed within the first week of treatment. If clinical signs or symptoms develop that are consistent with liver disease, treatment should be discontinued and liver function testing performed. Continued SPORANOX®use or reinstitution of treatment with SPORANOX®is strongly discouraged unless there is a serious or life-threatening situation where the expected benefit exceeds the risk. (See PRECAUTIONS: Information for Patients and ADVERSE REACTIONS.)
Cardiac Dysrhythmias:
Life-threatening cardiac dysrhythmias and/or sudden death have occurred in patients using cisapride, pimozide, levacetylmethadol (levomethadyl), or quinidine concomitantly with SPORANOX® and/or other CYP3A4 inhibitors. Concomitant administration of these drugs with SPORANOX® is contraindicated. (See BOX WARNING, CONTRAINDICATIONS, and PRECAUTIONS: Drug Interactions.)
Cardiac Disease:
SPORANOX® Oral Solution should not be used in patients with evidence of ventricular dysfunction unless the benefit clearly outweighs the risk. For patients with risk factors for congestive heart failure, physicians should carefully review the risks and benefits of SPORANOX® therapy. These risk factors include cardiac disease such as ischemic and valvular disease; significant pulmonary disease such as chronic obstructive pulmonary disease; and renal failure and other edematous disorders. Such patients should be informed of the signs and symptoms of CHF, should be treated with caution, and should be monitored for signs and symptoms of CHF during treatment. If signs or symptoms of CHF appear during administration of SPORANOX® Oral Solution, monitor carefully and consider other treatment alternatives which may include discontinuation of SPORANOX® Oral Solution administration.
Itraconazole has been shown to have a negative inotropic effect. When itraconazole was administered intravenously to anesthetized dogs, a dose-related negative inotropic effect was documented. In a healthy volunteer study of SPORANOX® Injection (intravenous infusion), transient, asymptomatic decreases in left ventricular ejection fraction were observed using gated SPECT imaging; these resolved before the next infusion, 12 hours later.
SPORANOX® has been associated with reports of congestive heart failure. In post-marketing experience, heart failure was more frequently reported in patients receiving a total daily dose of 400 mg although there were also cases reported among those receiving lower total daily doses.
Calcium channel blockers can have negative inotropic effects which may be additive to those of itraconazole. In addition, itraconazole can inhibit the metabolism of calcium channel blockers. Therefore, caution should be used when co-administering itraconazole and calcium channel blockers due to an increased risk of CHF. Concomitant administration of SPORANOX® and nisoldipine is contraindicated.
Cases of CHF, peripheral edema, and pulmonary edema have been reported in the post-marketing period among patients being treated for onychomycosis and/or systemic fungal infections. (See CLINICAL PHARMACOLOGY: Special Populations, PRECAUTIONS: Drug Interactions, and ADVERSE REACTIONS: Post-marketing Experience for more information.)
Cystic Fibrosis:
If a patient with cystic fibrosis does not respond to SPORANOX® Oral Solution, consideration should be given to switching to alternative therapy (see CLINICAL PHARMACOLOGY/Special Populations).
Treatment of Severely Neutropenic Patients:
SPORANOX® Oral Solution as treatment for oropharyngeal and/or esophageal candidiasis was not investigated in severely neutropenic patients. Due to its pharmacokinetic properties, SPORANOX® Oral Solution is not recommended for initiation of treatment in patients at immediate risk of systemic candidiasis.
In febrile neutropenic subjects in whom the likelihood of systemic candidiasis is considered high, therapy should be initiated with Sporanox Intravenous formulation.
PRECAUTIONS
Hepatotoxicity:
Rare cases of serious hepatotoxicity have been observed with SPORANOX® treatment, including some cases within the first week. In patients with elevated or abnormal liver enzymes or active liver disease, or who have experienced liver toxicity with other drugs, treatment with SPORANOX® is strongly discouraged unless there is a serious or life-threatening situation where the expected benefit exceeds the risk. Liver function monitoring should be done in patients with pre-existing hepatic function abnormalities or those who have experienced liver toxicity with other medications and should be considered in all patients receiving SPORANOX®. Treatment should be stopped immediately and liver function testing should be conducted in patients who develop signs and symptoms suggestive of liver dysfunction.
Neuropathy:
If neuropathy occurs that may be attributable to SPORANOX® Oral Solution, the treatment should be discontinued.
Hearing Loss:
Transient or permanent hearing loss has been reported in patients receiving treatment with itraconazole. Several of these reports included concurrent administration of quinidine which is contraindicated (see BOX WARNING: Drug Interactions, CONTRAINDICATIONS: Drug Interactions and PRECAUTIONS: Drug Interactions). The hearing loss usually resolves when treatment is stopped, but can persist in some patients.
Information for Patients:
- Only SPORANOX® Oral Solution has been demonstrated effective for oral and/or esophageal candidiasis. SPORANOX® Oral Solution contains the excipient hydroxypropyl-β-cyclodextrin which produced pancreatic adenocarcinomas in a rat carcinogenicity study. These findings were not observed in a similar mouse carcinogenicity study. The clinical relevance of these findings is unknown. (See Carcinogenesis, Mutagenesis, and Impairment of Fertility.)
- Taking SPORANOX® Oral Solution under fasted conditions improves the systemic availability of itraconazole. Instruct patients to take SPORANOX® Oral Solution without food, if possible.
- SPORANOX® Oral Solution should not be used interchangeably with SPORANOX® Capsules.
- Instruct patients about the signs and symptoms of congestive heart failure, and if these signs or symptoms occur during SPORANOX® administration, they should discontinue SPORANOX® and contact their healthcare provider immediately.
- Instruct patients to stop SPORANOX® treatment immediately and contact their healthcare provider if any signs and symptoms suggestive of liver dysfunction develop. Such signs and symptoms may include unusual fatigue, anorexia, nausea and/or vomiting, jaundice, dark urine or pale stools.
- Instruct patients to contact their physician before taking any concomitant medications with itraconazole to ensure there are no potential drug interactions.
- Instruct patients that hearing loss can occur with the use of itraconazole. The hearing loss usually resolves when treatment is stopped, but can persist in some patients. Advise patients to discontinue therapy and inform their physicians if any hearing loss symptoms occur.
Drug Interactions:
Itraconazole and its major metabolite, hydroxyitraconazole, are inhibitors of CYP3A4. Therefore, the following drug interactions may occur (See Table 1 below and the following drug class subheadings that follow):
- SPORANOX® may decrease the elimination of drugs metabolized by CYP3A4, resulting in increased plasma concentrations of these drugs when they are administered with SPORANOX®. These elevated plasma concentrations may increase or prolong both therapeutic and adverse effects of these drugs. Whenever possible, plasma concentrations of these drugs should be monitored, and dosage adjustments made after concomitant SPORANOX® therapy is initiated. When appropriate, clinical monitoring for signs or symptoms of increased or prolonged pharmacologic effects is advised. Upon discontinuation, depending on the dose and duration of treatment, itraconazole plasma concentrations decline gradually (especially in patients with hepatic cirrhosis or in those receiving CYP3A4 inhibitors). This is particularly important when initiating therapy with drugs whose metabolism is affected by itraconazole.
- Inducers of CYP3A4 may decrease the plasma concentrations of itraconazole. SPORANOX® may not be effective in patients concomitantly taking SPORANOX® and one of these drugs. Therefore, administration of these drugs with SPORANOX® is not recommended.
- Other inhibitors of CYP3A4 may increase the plasma concentrations of itraconazole. Patients who must take SPORANOX® concomitantly with one of these drugs should be monitored closely for signs or symptoms of increased or prolonged pharmacologic effects of SPORANOX®.
| 1 This list is not all-inclusive. | |
| 2 Contraindicated with SPORANOX® based on clinical and/or pharmacokinetics studies. (See WARNINGS and below.) | |
| 3 For information on parenterally administered midazolam, see the Benzodiazepine paragraph below. | |
| Drug plasma concentration increased by itraconazole | |
| Antiarrhythmics | digoxin, dofetilide,2 quinidine,2 disopyramide |
| Anticoagulants | warfarin |
| Anticonvulsants | carbamazepine |
| Antimycobacterials | rifabutin |
| Antineoplastics | busulfan, docetaxel, vinca alkaloids |
| Antipsychotics | pimozide2 |
| Benzodiazepines | alprazolam, diazepam, midazolam,2, 3 triazolam2 |
| Calcium Channel Blockers | dihydropyridines (including nisoldipine2), verapamil |
| Gastrointestinal Motility Agents | cisapride2 |
| HMG CoA-Reductase Inhibitors | atorvastatin, cerivastatin, lovastatin,2 simvastatin2 |
| Immunosuppressants | cyclosporine, tacrolimus, sirolimus |
| Oral Hypoglycemics | oral hypoglycemics |
| Protease Inhibitors | indinavir, ritonavir, saquinavir |
| Other | levacetylmethadol (levomethadyl),2 ergot alkaloids,2 halofantrine, alfentanil, buspirone, methylprednisolone, budesonide, dexamethasone, fluticasone, trimetrexate, warfarin, cilostazol, eletriptan, fentanyl |
| Decrease plasma concentration of itraconazole | |
| Anticonvulsants | carbamazepine, phenobarbital, phenytoin |
| Antimycobacterials | isoniazid, rifabutin, rifampin |
| Gastric Acid Suppressors/Neutralizers | antacids, H2-receptor antagonists, proton pump inhibitors |
| Reverse Transcriptase Inhibitors | nevirapine |
| Increase plasma concentration of itraconazole | |
| Macrolide Antibiotics | clarithromycin, erythromycin |
| Protease Inhibitors | indinavir, ritonavir |
Antiarrhythmics:
The class IA antiarrhythmic quinidine and class III antiarrhythmic dofetilide are known to prolong the QT interval. Coadministration of quinidine or dofetilide with SPORANOX® may increase plasma concentrations of quinidine or dofetilide which could result in serious cardiovascular events. Therefore, concomitant administration of SPORANOX® and quinidine or dofetilide is contraindicated. (See BOX WARNING, CONTRAINDICATIONS, and WARNINGS.)
The class IA antiarrhythmic disopyramide has the potential to increase the QT interval at high plasma concentrations. Caution is advised when SPORANOX® and disopyramide are administered concomitantly.
Concomitant administration of digoxin and SPORANOX® has led to increased plasma concentrations of digoxin via inhibition of P-glycoprotein.
Anticoagulants:
SPORANOX® enhances the anticoagulant effect of coumarin-like drugs, such as warfarin.
Anticonvulsants:
Reduced plasma concentrations of itraconazole were reported when SPORANOX® was administered concomitantly with phenytoin. Carbamazepine, phenobarbital, and phenytoin are all inducers of CYP3A4. Although interactions with carbamazepine and phenobarbital have not been studied, concomitant administration of SPORANOX® and these drugs would be expected to result in decreased plasma concentrations of itraconazole. In addition, in vivo studies have demonstrated an increase in plasma carbamazepine concentrations in subjects concomitantly receiving ketoconazole. Although there are no data regarding the effect of itraconazole on carbamazepine metabolism, because of the similarities between ketoconazole and itraconazole, concomitant administration of SPORANOX® and carbamazepine may inhibit the metabolism of carbamazepine.
Antimycobacterials:
Drug interaction studies have demonstrated that plasma concentrations of azole antifungal agents and their metabolites, including itraconazole and hydroxyitraconazole, were significantly decreased when these agents were given concomitantly with rifabutin or rifampin. In vivo data suggest that rifabutin is metabolized in part by CYP3A4. SPORANOX® may inhibit the metabolism of rifabutin. Although no formal study data are available for isoniazid, similar effects should be anticipated. Therefore, the efficacy of SPORANOX® could be substantially reduced if given concomitantly with one of these agents. Coadministration is not recommended.
Antineoplastics:
SPORANOX® may inhibit the metabolism of busulfan, docetaxel, and vinca alkaloids.
Antipsychotics:
Pimozide is known to prolong the QT interval and is partially metabolized by CYP3A4. Coadministration of pimozide with SPORANOX® could result in serious cardiovascular events. Therefore, concomitant administration of SPORANOX® and pimozide is contraindicated. (See BOX WARNING, CONTRAINDICATIONS, and WARNINGS.)
Benzodiazepines:
Concomitant administration of SPORANOX® and alprazolam, diazepam, oral midazolam, or triazolam could lead to increased plasma concentrations of these benzodiazepines. Increased plasma concentrations could potentiate and prolong hypnotic and sedative effects. Concomitant administration of SPORANOX® and oral midazolam or triazolam is contraindicated. (See CONTRAINDICATIONS and WARNINGS.) If midazolam is administered parenterally, special precaution and patient monitoring is required since the sedative effect may be prolonged.
Calcium Channel Blockers:
Edema has been reported in patients concomitantly receiving SPORANOX® and dihydropyridine calcium channel blockers. Appropriate dosage adjustment may be necessary.
Calcium channel blockers can have a negative inotropic effect which may be additive to those of itraconazole; itraconazole can inhibit the metabolism of calcium channel blockers such as dihydropyridines (e.g., nifedipine and felodipine) and verapamil. Therefore, caution should be used when co-administering itraconazole and calcium channel blockers due to an increased risk of CHF. Concomitant administration of SPORANOX® and nisoldipine results in clinically significant increases in nisoldipine plasma concentrations, which cannot be managed by dosage reduction, therefore the concomitant administration of SPORANOX® and nisoldipine is contraindicated. (See CLINICAL PHARMACOLOGY: Special Populations, CONTRAINDICATIONS, WARNINGS, and ADVERSE REACTIONS: Post-marketing Experience for more information.)
Gastric Acid Suppressors/Neutralizers:
Reduced plasma concentrations of itraconazole were reported when SPORANOX® Capsules were administered concomitantly with H2-receptor antagonists. Studies have shown that absorption of itraconazole is impaired when gastric acid production is decreased. Therefore, SPORANOX® should be administered with a cola beverage if the patient has achlorhydria or is taking H2-receptor antagonists or other gastric acid suppressors. Antacids should be administered at least 1 hour before or 2 hours after administration of SPORANOX® Capsules. In a clinical study, when SPORANOX® Capsules were administered with omeprazole (a proton pump inhibitor), the bioavailability of itraconazole was significantly reduced. However, as itraconazole is already dissolved in SPORANOX® Oral Solution, the effect of H2-antagonists is expected to be substantially less than with the capsules. Nevertheless, caution is advised when the two drugs are coadministered.
Gastrointestinal Motility Agents:
Coadministration of SPORANOX® with cisapride can elevate plasma cisapride concentrations which could result in serious cardiovascular events. Therefore, concomitant administration of SPORANOX® with cisapride is contraindicated. (See BOX WARNING, CONTRAINDICATIONS, and WARNINGS.)
HMG CoA-Reductase Inhibitors:
Human pharmacokinetic data suggest that SPORANOX® inhibits the metabolism of atorvastatin, cerivastatin, lovastatin, and simvastatin, which may increase the risk of skeletal muscle toxicity, including rhabdomyolysis. Concomitant administration of SPORANOX® with HMG CoA-reductase inhibitors, such as lovastatin or simvastatin is contraindicated. (See CONTRAINDICATIONS and WARNINGS.)
Immunosuppressants:
Concomitant administration of SPORANOX® and cyclosporine or tacrolimus has led to increased plasma concentrations of these immunosuppressants. Concomitant administration of SPORANOX® and sirolimus could increase plasma concentrations of sirolimus.
Macrolide Antibiotics:
Erythromycin and clarithromycin are known inhibitors of CYP3A4 (See Table 1) and may increase plasma concentrations of itraconazole. In a small pharmacokinetic study involving HIV infected patients, clarithromycin was shown to increase plasma concentrations of itraconazole. Similarly, following administration of 1 gram of erythromycin ethyl succinate and 200 mg itraconazole as single doses, the mean Cmax and AUC 0-∞ of itraconazole increased by 44% (90% CI: 119-175%) and 36% (90% CI: 108-171%), respectively.
Oral Hypoglycemic Agents:
Severe hypoglycemia has been reported in patients concomitantly receiving azole antifungal agents and oral hypoglycemic agents. Blood glucose concentrations should be carefully monitored when SPORANOX® and oral hypoglycemic agents are coadministered.
Polyenes:
Prior treatment with itraconazole, like other azoles, may reduce or inhibit the activity of polyenes such as amphotericin B. However, the clinical significance of this drug effect has not been clearly defined.
Protease Inhibitors:
Concomitant administration of SPORANOX® and protease inhibitors metabolized by CYP3A4, such as indinavir, ritonavir, and saquinavir, may increase plasma concentrations of these protease inhibitors. In addition, concomitant administration of SPORANOX® and indinavir and ritonavir (but not saquinavir) may increase plasma concentrations of itraconazole. Caution is advised when SPORANOX® and protease inhibitors must be given concomitantly.
Reverse Transcriptase Inhibitors:
Nevirapine is an inducer of CYP3A4. In vivo studies have shown that nevirapine induces the metabolism of ketoconazole, significantly reducing the bioavailability of ketoconazole. Studies involving nevirapine and itraconazole have not been conducted. However, because of the similarities between ketoconazole and itraconazole, concomitant administration of SPORANOX® and nevirapine is not recommended. In a clinical study, when 8 HIV-infected subjects were treated concomitantly with SPORANOX® Capsules 100 mg twice daily and the nucleoside reverse transcriptase inhibitor zidovudine 8 ± 0.4 mg/kg/day, the pharmacokinetics of zidovudine were not affected. Other nucleoside reverse transcriptase inhibitors have not been studied.
Other:
- Levacetylmethadol (levomethadyl) is known to prolong the QT interval and is metabolized by CYP3A4. Co-administration of levacetylmethadol with SPORANOX® could result in serious cardiovascular events. Therefore, concomitant administration of SPORANOX® and levacetylmethadol is contraindicated.
- Elevated concentrations of ergot alkaloids can cause ergotism, i.e., a risk for vasospasm potentially leading to cerebral ischemia and/or ischemia of the extremities. Concomitant administration of ergot alkaloids such as dihydroergotamine, ergometrine (ergonovine), ergotamine and methylergometrine (methylergonovine) with SPORANOX® is contraindicated.
- Halofantrine has the potential to prolong the QT interval at high plasma concentrations. Caution is advised when SPORANOX® and halofantrine are administered concomitantly.
- In vitro data suggest that alfentanil is metabolized by CYP3A4. Administration with SPORANOX® may increase plasma concentrations of alfentanil.
- Human pharmacokinetic data suggest that concomitant administration of SPORANOX® and buspirone results in significant increases in plasma concentrations of buspirone.
- SPORANOX® may inhibit the metabolism of certain glucocorticosteroids such as budesonide, dexamethasone, fluticasone, and methylprednisolone.
- In vitro data suggest that trimetrexate is extensively metabolized by CYP3A4. In vitro animal models have demonstrated that ketoconazole potently inhibits the metabolism of trimetrexate. Although there are no data regarding the effect of itraconazole on trimetrexate metabolism, because of the similarities between ketoconazole and itraconazole, concomitant administration of SPORANOX® and trimetrexate may inhibit the metabolism of trimetrexate.
- Cilostazol and eletriptan are CYP3A4 metabolized drugs that should be used with caution when co-administered with SPORANOX®.
- Fentanyl plasma concentrations could be increased or prolonged by concomitant use of SPORANOX® and may cause potentially fatal respiratory depression.
Carcinogenesis, Mutagenesis, and Impairment of Fertility:
Itraconazole showed no evidence of carcinogenicity potential in mice treated orally for 23 months at dosage levels up to 80 mg/kg/day (approximately 10x the maximum recommended human dose [MRHD]). Male rats treated with 25 mg/kg/day (3.1x MRHD) had a slightly increased incidence of soft tissue sarcoma. These sarcomas may have been a consequence of hypercholesterolemia, which is a response of rats, but not dogs or humans, to chronic itraconazole administration. Female rats treated with 50 mg/kg/day (6.25x MRHD) had an increased incidence of squamous cell carcinoma of the lung (2/50) as compared to the untreated group. Although the occurrence of squamous cell carcinoma in the lung is extremely uncommon in untreated rats, the increase in this study was not statistically significant.
Hydroxypropyl-β-cyclodextrin (HP-β-CD), the solubilizing excipient used in SPORANOX® Oral Solution, was found to produce pancreatic exocrine hyperplasia and neoplasia when administered orally to rats at doses of 500, 2000 or 5000 mg/kg/day for 25 months. Adenocarcinomas of the exocrine pancreas produced in the treated animals were not seen in the untreated group and are not reported in the historical controls. Development of these tumors may be related to a mitogenic action of cholecystokinin. This finding was not observed in the mouse carcinogenicity study at doses of 500, 2000 or 5000 mg/kg/day for 22-23 months; however, the clinical relevance of these findings is unknown. Based on body surface area comparisons, the exposure to humans of HP-β-CD at the recommended clinical dose of SPORANOX® Oral Solution, is approximately equivalent to 1.7 times the exposure at the lowest dose in the rat study.
Itraconazole produced no mutagenic effects when assayed in a DNA repair test (unscheduled DNA synthesis) in primary rat hepatocytes, in Ames tests with Salmonella typhimurium (6 strains) and Escherichia coli, in the mouse lymphoma gene mutation tests, in a sex-linked recessive lethal mutation (Drosophila melanogaster) test, in chromosome aberration tests in human lymphocytes, in a cell transformation test with C3H/10T½ C18 mouse embryo fibroblasts cells, in a dominant lethal mutation test in male and female mice, and in micronucleus tests in mice and rats.
Itraconazole did not affect the fertility of male or female rats treated orally with dosage levels of up to 40 mg/kg/day (5x MRHD), even though parental toxicity was present at this dosage level. More severe signs of parental toxicity, including death, were present in the next higher dosage level, 160 mg/kg/day (20x MRHD).
Pregnancy: Teratogenic Effects. Pregnancy Category C:
Itraconazole was found to cause a dose-related increase in maternal toxicity, embryotoxicity, and teratogenicity in rats at dosage levels of approximately 40-160 mg/kg/day (5-20x MRHD), and in mice at dosage levels of approximately 80 mg/kg/day (10x MRHD). In rats, the teratogenicity consisted of major skeletal defects; in mice, it consisted of encephaloceles and/or macroglossia.
There are no studies in pregnant women. SPORANOX® should be used in pregnancy only if the benefit outweighs the potential risk.
During post-marketing experience, cases of congenital abnormalities have been reported. (See ADVERSE REACTIONS, Post-marketing Experience.)
Nursing Mothers:
Itraconazole is excreted in human milk; therefore, the expected benefits of SPORANOX® therapy for the mother should be weighed against the potential risk from exposure of itraconazole to the infant. The U.S. Public Health Service Centers for Disease Control and Prevention advises HIV-infected women not to breast-feed to avoid potential transmission of HIV to uninfected infants.
Pediatric Use:
The efficacy and safety of SPORANOX® have not been established in pediatric patients. A pharmacokinetic study was conducted with SPORANOX® Oral Solution in 26 pediatric patients, ages 6 months to 12 years, requiring systemic antifungal treatment. Itraconazole was dosed at 5 mg/kg once daily for two weeks and no serious unexpected adverse events were reported. (See CLINICAL PHARMACOLOGY.)
The long-term effects of itraconazole on bone growth in children are unknown. In three toxicology studies using rats, itraconazole induced bone defects at dosage levels as low as 20 mg/kg/day (2.5x MRHD). The induced defects included reduced bone plate activity, thinning of the zona compacta of the large bones, and increased bone fragility. At a dosage level of 80 mg/kg/day (10x MRHD) over 1 year or 160 mg/kg/day (20x MRHD) for 6 months, itraconazole induced small tooth pulp with hypocellular appearance in some rats. No such bone toxicity has been reported in adult patients.
Geriatric Use:
Transient or permanent hearing loss has been reported in elderly patients receiving treatment with itraconazole. Several of these reports included concurrent administration of quinidine which is contraindicated (see BOX WARNING: Drug Interactions, CONTRAINDICATIONS: Drug Interactions and PRECAUTIONS: Drug Interactions). Itraconazole should be used with care in elderly patients (see PRECAUTIONS).
Renal Impairment:
Limited data are available on the use of oral itraconazole in patients with renal impairment. Caution should be exercised when this drug is administered in this patient population. (See CLINICAL PHARMACOLOGY: Special Populations and DOSAGE AND ADMINISTRATION.)
Hepatic Impairment:
Limited data are available on the use of oral itraconazole in patients with hepatic impairment. Caution should be exercised when this drug is administered in this patient population. (See CLINICAL PHARMACOLOGY: Special Populations and DOSAGE AND ADMINISTRATION.)
ADVERSE REACTIONS
SPORANOX® has been associated with rare cases of serious hepatotoxicity, including liver failure and death. Some of these cases had neither pre-existing liver disease nor a serious underlying medical condition. If clinical signs or symptoms develop that are consistent with liver disease, treatment should be discontinued and liver function testing performed. The risks and benefits of SPORANOX® use should be reassessed. (See WARNINGS: Hepatic Effects and PRECAUTIONS: Hepatotoxicity and Information for Patients.)
Adverse Events Reported in Empiric Therapy in Febrile Neutropenic (ETFN) Patients
Adverse events considered at least possibly drug related in a clinical trial of empiric therapy in 384 febrile, neutropenic patients (192 treated with SPORANOX® and 192 with amphotericin B) with suspected fungal infections are listed in Table 2 below. Patients received a regimen of SPORANOX® Injection followed by SPORANOX® Oral Solution. The dose of SPORANOX® Injection was 200 mg twice daily for the first two days followed by a single daily dose of 200 mg for the remainder of the intravenous treatment period. The majority of patients received between 7 and 14 days of SPORANOX® Injection. The dose of SPORANOX® Oral Solution was 200 mg (20 mL) b.i.d. for the remainder of therapy.
| Adverse Event | SPORANOX®
(N=192) % | Amphotericin B
(N=192) % |
|---|---|---|
| Gastrointestinal system disorders | ||
| Nausea | 11 | 15 |
| Diarrhea | 10 | 9 |
| Vomiting | 7 | 10 |
| Abdominal pain | 3 | 3 |
| Metabolic and nutritional disorders | ||
| Hypokalemia | 9 | 28 |
| Serum creatinine increased | 3 | 25 |
| LDH increased | 2 | 0 |
| Alkaline phosphatase increased | 2 | 2 |
| Hypomagnesemia | 2 | 4 |
| Blood urea nitrogen increased | 1 | 6 |
| Fluid overload | 1 | 3 |
| Hypocalcemia | 1 | 2 |
| Liver and biliary system disorders | ||
| Bilirubinemia | 6 | 3 |
| Hepatic function abnormal | 3 | 2 |
| SGPT/ALT increased | 3 | 1 |
| Jaundice | 2 | 1 |
| SGOT/AST increased | 2 | 1 |
| Skin and appendage disorders | ||
| Rash | 5 | 3 |
| Sweating increased | 2 | 1 |
| CNS and peripheral nervous system | ||
| Headache | 2 | 2 |
| Body as a whole | ||
| Edema | 2 | 2 |
| Rigors | 1 | 34 |
| Fever | 0 | 7 |
| Respiratory system disorder | ||
| Dyspnea | 1 | 3 |
| Urinary system disorder | ||
| Renal function abnormal | 1 | 12 |
| Cardiovascular disorders, general | ||
| Hypotension | 1 | 3 |
| Hypertension | 0 | 2 |
| Heart rate and rhythm disorders | ||
| Tachycardia | 1 | 3 |
The following additional adverse events considered at least possibly related occurred in between 1 and 2% of patients who received SPORANOX® Injection and Oral Solution: constipation, hypophosphatemia, gamma-GT increased, erythematous rash, pruritus, dizziness, tremor, and pulmonary infiltration.
Adverse Events Reported in Oropharyngeal or Esophageal Candidiasis Trials
U.S. adverse experience data are derived from 350 immunocompromised patients (332 HIV seropositive/AIDS) treated for oropharyngeal or esophageal candidiasis. Table 3 below lists adverse events reported by at least 2% of patients treated with SPORANOX® Oral Solution in U.S. clinical trials. Data on patients receiving comparator agents in these trials are included for comparison.
|
Body System/ Adverse Event | Itraconazole | |||
|---|---|---|---|---|
| Total (n = 350*) % | All
controlled studies (n = 272) % | Fluconazole (n = 125†) % | Clotrimazole (n = 81‡) % | |
| * Of the 350 patients, 209 were treated for oropharyngeal candidiasis in controlled studies, 63 were treated for esophageal candidiasis in controlled studies and 78 were treated for oropharyngeal candidiasis in an open study. | ||||
| † Of the 125 patients, 62 were treated for oropharyngeal candidiasis and 63 were treated for esophageal candidiasis. | ||||
| ‡ All 81 patients were treated for oropharyngeal candidiasis. | ||||
| Gastrointestinal disorders | ||||
| Nausea Diarrhea Vomiting Abdominal pain Constipation | 11 11 7 6 2 | 10 10 6 4 2 | 11 10 8 7 1 | 5 4 1 7 0 |
| Body as a whole | ||||
| Fever Chest pain Pain Fatigue | 7 3 2 2 | 6 3 2 1 | 8 2 4 2 | 5 0 0 0 |
| Respiratory disorders | ||||
| Coughing Dyspnea Pneumonia Sinusitis Sputum increased | 4 2 2 2 2 | 4 3 2 2 3 | 10 5 0 4 3 | 0 1 0 0 1 |
| Skin and appendages disorders | ||||
| Rash Increased sweating Skin disorder unspecified | 4 3 2 | 5 4 2 | 4 6 2 | 6 1 1 |
| Central/peripheral nervous system | ||||
| Headache Dizziness | 4 2 | 4 2 | 6 4 | 6 1 |
| Resistance mechanism disorders | ||||
| Pneumocystis carinii infection | 2 | 2 | 2 | 0 |
| Psychiatric disorders | ||||
| Depression | 2 | 1 | 0 | 1 |
Adverse events reported by less than 2% of patients in U.S. clinical trials with SPORANOX® included: adrenal insufficiency, asthenia, back pain, dehydration, dyspepsia, dysphagia, flatulence, gynecomastia, hematuria, hemorrhoids, hot flushes, implantation complication, infection unspecified, injury, insomnia, male breast pain, myalgia, pharyngitis, pruritus, rhinitis, rigors, stomatitis ulcerative, taste perversion, tinnitus, upper respiratory tract infection, vision abnormal, and weight decrease. Edema, hypokalemia and menstrual disorders have been reported in clinical trials with itraconazole capsules.
Post-marketing Experience
Adverse drug reactions that have been identified during post-approval use of SPORANOX® (all formulations) are listed in the table below. Because these reactions are reported voluntarily from a population of uncertain size, reliably estimating their frequency or establishing a causal relationship to drug exposure is not always possible.
| Blood and lymphatic system disorders: | Leukopenia, neutropenia, thrombocytopenia |
| Immune system disorders: | Anaphylaxis; anaphylactic, anaphylactoid and allergic reactions; serum sickness; angioneurotic edema |
| Metabolism and nutrition disorders: | Hypertriglyceridemia, hypokalemia |
| Nervous system disorders: | Peripheral neuropathy, paresthesia, hypoesthesia, headache, dizziness |
| Eye disorders: | Visual disturbances, including vision blurred and diplopia |
| Ear and labyrinth disorders: | Transient or permanent hearing loss, tinnitus |
| Cardiac disorders: | Congestive heart failure |
| Respiratory, thoracic and mediastinal disorders: | Pulmonary edema |
| Gastrointestinal disorders: | Pancreatitis, abdominal pain, vomiting, dyspepsia, nausea, diarrhea, constipation, dysgeusia |
| Hepato-biliary disorders: | Serious hepatotoxicity (including some cases of fatal acute liver failure), hepatitis, reversible increases in hepatic enzymes |
| Skin and subcutaneous tissue disorders: | Toxic epidermal necrolysis, Stevens-Johnson syndrome, exfoliative dermatitis, leukocytoclastic vasculitis, erythema multiforme, alopecia, photosensitivity, rash, urticaria, pruritus |
| Musculoskeletal and connective tissue disorders: | Myalgia, arthralgia |
| Renal and urinary disorders: | Urinary incontinence, pollakiuria |
| Reproductive system and breast disorders: | Menstrual disorders, erectile dysfunction |
| General disorders and administration site conditions: | Peripheral edema, pyrexia |
There is limited information on the use of SPORANOX® during pregnancy. Cases of congenital abnormalities including skeletal, genitourinary tract, cardiovascular and ophthalmic malformations as well as chromosomal and multiple malformations have been reported during post-marketing experience. A causal relationship with SPORANOX® has not been established. (See CLINICAL PHARMACOLOGY: Special Populations, CONTRAINDICATIONS, WARNINGS, and PRECAUTIONS: Drug Interactions for more information.)
OVERDOSAGE
Itraconazole is not removed by dialysis. In the event of accidental overdosage, supportive measures, including gastric lavage with sodium bicarbonate, should be employed.
There are limited data on the outcomes of patients ingesting high doses of itraconazole. In patients taking either 1000 mg of SPORANOX® (itraconazole) Oral Solution or up to 3000 mg of SPORANOX® Capsules, the adverse event profile was similar to that observed at recommended doses.
DOSAGE AND ADMINISTRATION
Empiric Therapy in Febrile, Neutropenic Patients with Suspected Fungal Infections (ETFN):
The recommended dose of SPORANOX® Injection is 200 mg b.i.d. for four doses, followed by 200 mg once daily for up to 14 days. Each intravenous dose should be infused over 1 hour. Treatment should be continued with SPORANOX® Oral Solution 200 mg (20 mL) b.i.d. until resolution of clinically significant neutropenia. The safety and efficacy of SPORANOX® use exceeding 28 days in ETFN is not known.
SPORANOX® Oral Solution is a different preparation than SPORANOX® Capsules and should not be used interchangeably.
Treatment of Oropharyngeal and Esophageal Candidiasis:
The solution should be vigorously swished in the mouth (10 mL at a time) for several seconds and swallowed.
The recommended dosage of SPORANOX® (itraconazole) Oral Solution for oropharyngeal candidiasis is 200 mg (20 mL) daily for 1 to 2 weeks. Clinical signs and symptoms of oropharyngeal candidiasis generally resolve within several days.
For patients with oropharyngeal candidiasis unresponsive/refractory to treatment with fluconazole tablets, the recommended dose is 100 mg (10 mL) b.i.d. For patients responding to therapy, clinical response will be seen in 2 to 4 weeks. Patients may be expected to relapse shortly after discontinuing therapy. Limited data on the safety of long-term use (>6 months) of SPORANOX® Oral Solution are available at this time.
The recommended dosage of SPORANOX® Oral Solution for esophageal candidiasis is 100 mg (10 mL) daily for a minimum treatment of three weeks. Treatment should continue for 2 weeks following resolution of symptoms. Doses up to 200 mg (20 mL) per day may be used based on medical judgment of the patient's response to therapy.
SPORANOX® Oral Solution and SPORANOX® Capsules should not be used interchangeably. Patients should be instructed to take SPORANOX® Oral Solution without food, if possible. Only SPORANOX® Oral Solution has been demonstrated effective for oral and/or esophageal candidiasis.
Use in Patients with Renal Impairment:
Limited data are available on the use of oral itraconazole in patients with renal impairment. Caution should be exercised when this drug is administered in this patient population. (See CLINICAL PHARMACOLOGY: Special Populations and PRECAUTIONS.)
Use in Patients with Hepatic Impairment:
Limited data are available on the use of oral itraconazole in patients with hepatic impairment. Caution should be exercised when this drug is administered in this patient population. (See CLINICAL PHARMACOLOGY: Special Populations and PRECAUTIONS.)
HOW SUPPLIED
SPORANOX® (itraconazole) Oral Solution is available in 150 mL amber glass bottles (NDC 50458-295-15) containing 10 mg of itraconazole per mL.
Store at or below 25°C (77°F). Do not freeze.
Keep out of reach of children.
U.S. Patent No. 4,267,179; 5,707,975; 4,727,064
Revised November 2009
©Centocor Ortho Biotech Products, L.P. 2003
Manufactured by:
Janssen Pharmaceutica N.V.
Beerse, Belgium
Manufactured for:
Centocor Ortho Biotech Products, L.P.
Raritan, NJ 08869
Sporanox Oral Solution - Front Panel of Bottle
10mg of itraconazole in an aqueous solution.
Dosage: For information concerning dosage and
administration of SPORANOX® (itraconazole) oral
solution, please read accompanying Package Insert.
Store bottle at or below
25ºC (77ºF ). Do not freeze.
U.S. Patent No. 4,267,179
U.S. Patent No. 4,791,111
U.S. Patent No. 5,707,975
Distributed by:
Ortho Biotech Inc.
Raritan, NJ 08869
Manufactured by:
Janssen Pharmaceutica N.V.
Beerse, Belgium
Sporanox Oral Solution - Front Panel of Carton
SPORANOX®
(itraconazole) Oral Solution
Each 1mL contains:
10 mg of itraconazole in an aqueous solution.

| SPORANOX
itraconazole solution |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA020567 | 02/21/1997 | |
| Labeler - Ortho-McNeil-Janssen Pharmaceuticals Inc. (063137772) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Janssen Pharmaceutica, NV | 374747970 | API MANUFACTURE | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Janssen Pharmaceutical, Ltd | 989673884 | API MANUFACTURE | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Catalent | 806746405 | RELABEL | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Janssen Pharmaceutica, NV | 370005019 | MANUFACTURE | |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Ortho-McNeil-Janssen Pharmaceuticals, Inc | 063137772 | RELABEL | |