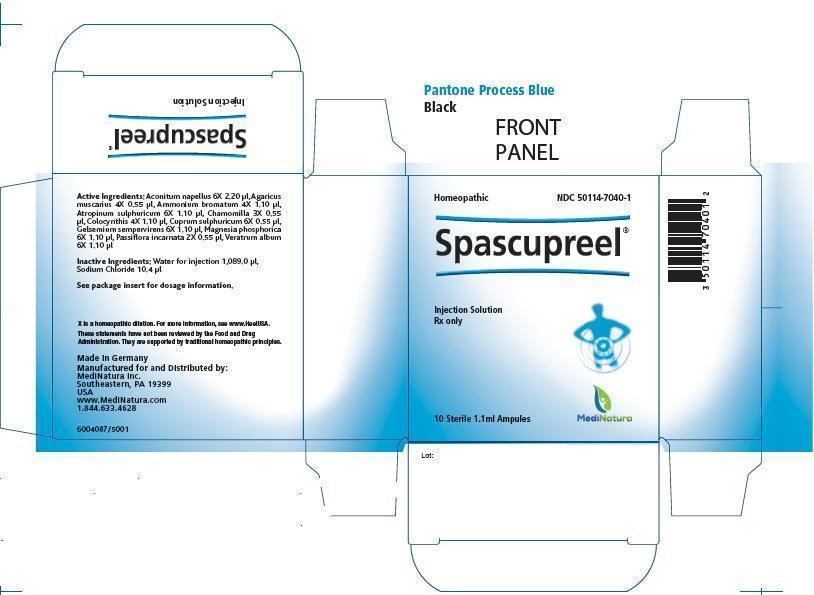
SPASCUPREEL - aconitum napellus, ammonium bromide, citrullus colocynthis fruit pulp, atropine sulfate, gelsemium sempervirens root, magnesium phosphate, dibasic trihydrate, veratrum album root, passiflora incarnata flowering top, matricaria recutita, amanita muscaria fruiting body and cupric sulfate injection
MediNatura Inc
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
Spascupreel 1.1ml Injection
DESCRIPTION
| Each 1.1 ml solution for injection ampule contains: | |||
| Active Ingredients: | |||
| Ingredient name | Potency | Quantity | Final dilution |
| Aconitum napellus | 6X | 2.20 µl | 8.69X |
| Agaricus muscarius | 4X | 0.55 µl | 7.30X |
| Ammonium bromatum | 4X | 1.10 µl | 7.00X |
| Atropinum sulphuricum | 6X | 1.10 µl | 8.99X |
| Chamomilla | 3X | 0.55 µl | 6.30X |
| Colocynthis | 4X | 1.10 µl | 7.00X |
| Cuprum sulphuricum | 6X | 0.55 µl | 9.30X |
| Gelsemium sempervirens | 6x | 1.10 µl | 8.99X |
| Magnesia phosphorica | 6X | 1.10 µl | 8.99X |
| Passiflora incarnata | 2X | 0.55 µl | 5.30X |
| Veratrum album | 6X | 1.10 µl | 8.99X |
Inactive Ingredients:
Water for injection 1,089.0 μl
Sodium Chloride 10.4 μl
INDICATION AND USAGE
Spascupreel® Injection Solution is a homeopathic drug product indicated for the relief of spasms of the smooth musculature of the gastrointestinal and the urogenital tract as well as general muscle spasms.
DOSAGE AND ADMINISTRATION
General Considerations
- The dosage schedules listed below can be used as a general guide for the administration of Spascupreel® Injection Solution.
- Spascupreel® Injection Solution may be administered s.c., i.d., i.m., or i.v.
-
If co-administration with a local anesthetic is desired, Spascupreel®Injection Solution may be mixed with lidocaine or similar agents at the discretion of the physician.
- The interval between injections is left to the discretion of the HCP, but should not exceed 1 ampule in 24 hours.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard any unused ampule contents.
- Draw up required dose into syringe.
- Discard any unused ampule contents. Do not reuse ampule.
Standard Dosage:
Adults and children 12 years and older: 1 ml 1 to 3 times per 7 days.
Children 6 to 11 years: 0.7 ml 1 to 3 times per 7 days.
Children 2 to 5 years: 0.5 ml 1 to 3 times per 7 days.
Acute Dosage:
Adults and children 12 years and older: 1 ml daily, and then continue with standard dosage.
Children 6 to 11 years: 0.7 ml daily, and then continue with standard dosage.
Children 2 to 5 years: 0.5 ml daily, and then continue with standard dosage.
CONTRAINDICATIONS
Spascupreel® Injection Solution is contraindicated in patients with known hypersensitivity to Spascupreel® or any of its ingredients.
ADVERSE REACTIONS
Post-marketing Experience
- No adverse events have been reported with a causal relationship to Spascupreel® injection solution.
| SPASCUPREEL
aconitum napellus, ammonium bromide, citrullus colocynthis fruit pulp, atropine sulfate, gelsemium sempervirens root, magnesium phosphate, dibasic trihydrate, veratrum album root, passiflora incarnata flowering top, matricaria recutita, amanita muscaria fruiting body and cupric sulfate injection |
|||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||
| Labeler - MediNatura Inc (102783016) |