fuzeon (enfuvirtide)
[Roche Laboratories Inc.]
DESCRIPTION
FUZEON (enfuvirtide) is an inhibitor of the fusion of HIV-1 with CD4+ cells. Enfuvirtide is a linear 36-amino acid synthetic peptide with the N-terminus acetylated and the C-terminus is a carboxamide. It is composed of naturally occurring L-amino acid residues.
Enfuvirtide is a white to off-white amorphous solid. It has negligible solubility in pure water and the solubility increases in aqueous buffers (pH 7.5) to 85-142 g/100 mL. The empirical formula of enfuvirtide is C204H301N51O64, and the molecular weight is 4492. It has the following primary amino acid sequence:
CH3CO-Tyr-Thr-Ser-Leu-Ile-His-Ser-Leu-Ile-Glu-Glu-Ser-Gln-Asn-Gln-Gln-Glu-Lys-Asn-Glu-Gln-Glu-Leu-Leu-Glu-Leu-Asp-Lys-Trp-Ala-Ser-Leu-Trp-Asn-Trp-Phe-NH2 and the following structural formula:
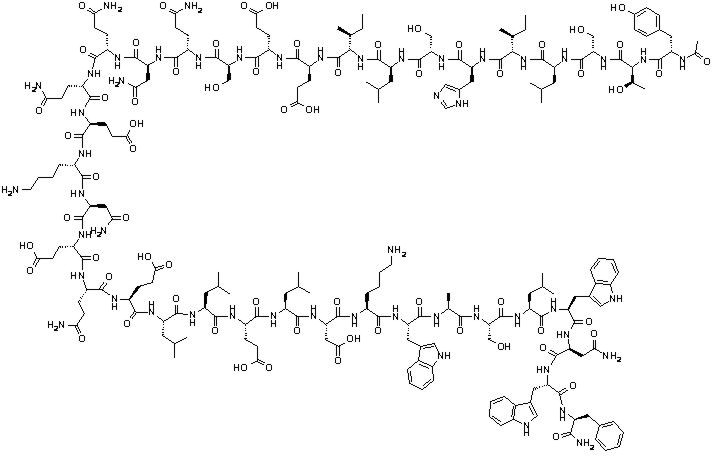
The drug product, FUZEON (enfuvirtide) for Injection, is a white to off-white, sterile, lyophilized powder. Each single-use vial contains 108 mg of enfuvirtide for the delivery of 90 mg. Prior to subcutaneous administration, the contents of the vial are reconstituted with 1.1 mL of Sterile Water for Injection giving a volume of approximately 1.2 mL to provide the delivery of 1 mL of the solution. Each 1 mL of the reconstituted solution contains approximately 90 mg of enfuvirtide with approximate amounts of the following excipients: 22.55 mg of mannitol, 2.39 mg of sodium carbonate (anhydrous), and sodium hydroxide and hydrochloric acid for pH adjustment as needed. The reconstituted solution has an approximate pH of 9.0.
MICROBIOLOGY
Mechanism of Action
Enfuvirtide interferes with the entry of HIV-1 into cells by inhibiting fusion of viral and cellular membranes. Enfuvirtide binds to the first heptad-repeat (HR1) in the gp41 subunit of the viral envelope glycoprotein and prevents the conformational changes required for the fusion of viral and cellular membranes.
Antiviral Activity In Vitro
The in vitro antiviral activity of enfuvirtide was assessed by infecting different CD4+ cell types with laboratory and clinical isolates of HIV-1. The IC50 values for baseline clinical isolates ranged from 0.089 to 107 nM (0.4 to 480 ng/mL) by the cMAGI assay (n=130) and from 1.56 to 1680 nM (7 to 7530 ng/mL) by a recombinant phenotypic entry assay (n=627). Enfuvirtide was similarly active in vitro against clades A, AE, C, D, E, F, and G (range 5.1 to 10.5 nM), and R5, X4, and dual tropic viruses. Enfuvirtide has no activity against HIV-2.
Enfuvirtide exhibited additive to synergistic effects in cell culture assays when combined with individual members of various antiretroviral classes, including lamivudine, zidovudine, indinavir, nelfinavir, and efavirenz.
Drug Resistance
HIV-1 isolates with reduced susceptibility to enfuvirtide have been selected in vitro. Genotypic analysis of the in vitro-selected resistant isolates showed mutations that resulted in amino acid substitutions at the enfuvirtide binding HR1 domain positions 36 to 38 of the HIV-1 envelope glycoprotein gp41. Phenotypic analysis of site-directed mutants in positions 36 to 38 in an HIV-1 molecular clone showed a 5-fold to 684-fold decrease in susceptibility to enfuvirtide.
In clinical trials, HIV-1 isolates with reduced susceptibility to enfuvirtide have been recovered from subjects failing a FUZEON containing regimen. Posttreatment HIV-1 virus from 277 subjects experiencing protocol defined virological failure at 48 weeks exhibited a median decrease in susceptibility to enfuvirtide of 33.4-fold (range 0.4-6318-fold) relative to their respective baseline virus. Of these, 249 had decreases in susceptibility to enfuvirtide of greater than 4-fold and all but 3 of those 249 exhibited genotypic changes in the codons encoding gp41 HR1 domain amino acids 36 to 45. Substitutions in this region were observed with decreasing frequency at amino acid positions 38, 43, 36, 40, 42, and 45.
Cross-resistance
HIV-1 clinical isolates resistant to nucleoside analogue reverse transcriptase inhibitors (NRTI), non-nucleoside analogue reverse transcriptase inhibitors (NNRTI), and protease inhibitors (PI) were susceptible to enfuvirtide in cell culture.
CLINICAL PHARMACOLOGY
Pharmacokinetics
The pharmacokinetic properties of enfuvirtide were evaluated in HIV-1 infected adult and pediatric patients.
Absorption
Following a 90-mg single subcutaneous injection of FUZEON into the abdomen in 12 HIV-1 infected subjects, the mean (±SD) Cmax was 4.59 ± 1.5 µg/mL, AUC was 55.8 ± 12.1 µg∙h/mL and the median Tmax was 8 hours (ranged from 3 to 12 h). The absolute bioavailability (using a 90-mg intravenous dose as a reference) was 84.3%± 15.5%. Following 90-mg bid dosing of FUZEON subcutaneously in combination with other antiretroviral agents in 11 HIV-1 infected subjects, the mean (±SD) steady-state Cmax was 5.0 ± 1.7 µg/mL, Ctrough was 3.3 ± 1.6 µg/mL, AUC0-12h was 48.7 ± 19.1 µg∙h/mL, and the median Tmax was 4 hours (ranged from 4 to 8 h).
Absorption of the 90-mg dose was comparable when injected into the subcutaneous tissue of the abdomen, thigh or arm.
Distribution
The mean (±SD) steady-state volume of distribution after intravenous administration of a 90-mg dose of FUZEON (N=12) was 5.5 ± 1.1 L.
Enfuvirtide is approximately 92% bound to plasma proteins in HIV-infected plasma over a concentration range of 2 to 10 µg/mL. It is bound predominantly to albumin and to a lower extent to α-1 acid glycoprotein.
Metabolism/Elimination
As a peptide, enfuvirtide is expected to undergo catabolism to its constituent amino acids, with subsequent recycling of the amino acids in the body pool.
Mass balance studies to determine elimination pathway(s) of enfuvirtide have not been performed in humans.
In vitro studies with human microsomes and hepatocytes indicate that enfuvirtide undergoes hydrolysis to form a deamidated metabolite at the C-terminal phenylalanine residue, M3. The hydrolysis reaction is not NADPH dependent. The M3 metabolite is detected in human plasma following administration of enfuvirtide, with an AUC ranging from 2.4% to 15% of the enfuvirtide AUC.
Following a 90-mg single subcutaneous dose of enfuvirtide (N=12) the mean ±SD elimination half-life of enfuvirtide is 3.8 ± 0.6 h and the mean ±SD apparent clearance was 24.8 ± 4.1 mL/h/kg. Following 90-mg bid dosing of FUZEON subcutaneously in combination with other antiretroviral agents in 11 HIV-1 infected subjects, the mean ±SD apparent clearance was 30.6 ± 10.6 mL/h/kg.
Special Populations
Hepatic Insufficiency
Formal pharmacokinetic studies of enfuvirtide have not been conducted in patients with hepatic impairment.
Renal Insufficiency
Formal pharmacokinetic studies of enfuvirtide have not been conducted in patients with renal insufficiency. However, analysis of plasma concentration data from subjects in clinical trials indicated that the clearance of enfuvirtide is not affected in patients with creatinine clearance greater than 35 mL/min. The effect of creatinine clearance less than 35 mL/min on enfuvirtide clearance is unknown.
Gender and Weight
GENDER
Analysis of plasma concentration data from subjects in clinical trials indicated that the clearance of enfuvirtide is 20% lower in females than males after adjusting for body weight.
WEIGHT
Enfuvirtide clearance decreases with decreased body weight irrespective of gender. Relative to the clearance of a 70-kg male, a 40-kg male will have 20% lower clearance and a 110-kg male will have a 26% higher clearance. Relative to a 70-kg male, a 40-kg female will have a 36% lower clearance and a 110-kg female will have the same clearance.
No dose adjustment is recommended for weight or gender.
Race
Analysis of plasma concentration data from subjects in clinical trials indicated that the clearance of enfuvirtide was not different in Blacks compared to Caucasians. Other pharmacokinetic studies suggest no difference between Asians and Caucasians after adjusting for body weight.
Pediatric Patients
The pharmacokinetics of enfuvirtide have been studied in 23 pediatric subjects aged 6 through 16 years at a dose of 2 mg/kg. Enfuvirtide pharmacokinetics were determined in the presence of concomitant medications including antiretroviral agents. A dose of 2 mg/kg bid (maximum 90 mg bid) provided enfuvirtide plasma concentrations similar to those obtained in adult patients receiving 90 mg bid.
In the 23 pediatric subjects receiving the 2 mg/kg bid dose, the mean ±SD steady-state AUC was 56.3 ± 22.3 µg∙h/mL, Cmax was 6.3 ± 2.4 µg/mL, Ctrough was 3.1 ± 1.5 µg/mL, and apparent clearance was 40 ± 17 mL/h/kg.
Geriatric Patients
The pharmacokinetics of enfuvirtide have not been studied in patients over 65 years of age.
Drug Interactions
Influence of FUZEON on the Metabolism of Concomitant Drugs
Based on the results from an in vitro human microsomal study, enfuvirtide is not an inhibitor of CYP450 enzymes. In an in vivo human metabolism study (N=12), FUZEON at the recommended dose of 90 mg bid did not alter the metabolism of CYP3A4, CYP2D6, CYP1A2, CYP2C19 or CYP2E1 substrates.
Influence of Concomitant Drugs on the Metabolism of Enfuvirtide
As indicated in Table 1, pharmacokinetic interaction studies were conducted between FUZEON and the following drugs: ritonavir, saquinavir/ritonavir, and rifampin.
| Coadministered Drug | Dose of Coadministered Drug | N | % Change of Enfuvirtide Pharmacokinetic Parameters†‡ (90% CI) |
||
|---|---|---|---|---|---|
| Cmax | AUC | Ctrough | |||
| Ritonavir | 200 mg, q12h, 4 days |
12 | ↑24 (↑9 to ↑41) |
↑22 (↑8 to ↑37) |
↑14 (↑2 to ↑28) |
| Saquinavir/Ritonavir | 1000/100 mg, q12h, 4 days |
12 | 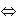 |
↑14 (↑5 to ↑24) |
↑26 (↑17 to ↑35) |
| Rifampin | 600 mg, qd, 10 days |
12 | 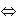 |
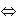 |
↓15 (↓22 to ↓7) |
INDICATIONS AND USAGE
FUZEON in combination with other antiretroviral agents is indicated for the treatment of HIV-1 infection in treatment-experienced patients with evidence of HIV-1 replication despite ongoing antiretroviral therapy.
This indication is based on results from two controlled studies of 48 weeks duration. Subjects enrolled were treatment-experienced adults; many had advanced disease. There are no studies of FUZEON in antiretroviral naive patients.
Description of Clinical Studies
Studies in Antiretroviral Experienced Patients
Studies T20-301 and T20-302 were randomized, controlled, open-label, multicenter trials in HIV-1 infected subjects. Subjects were required to have either (1) viremia despite 3 to 6 months prior therapy with a nucleoside reverse transcriptase inhibitor (NRTI), non-nucleoside reverse transcriptase inhibitor (NNRTI), and protease inhibitor (PI) or (2) viremia and documented resistance or intolerance to at least one member in each of the NRTI, NNRTI, and PI classes.
All subjects received an individualized background regimen consisting of 3 to 5 antiretroviral agents selected on the basis of the subject's prior treatment history and baseline genotypic and phenotypic viral resistance measurements. Subjects were then randomized at a 2:1 ratio to FUZEON 90 mg bid with background regimen or background regimen alone.
After week 8, patients on either treatment arm who met protocol defined criteria for virological failure were permitted to revise their background regimens; those on background regimen alone were also permitted to add FUZEON.
Demographic characteristics for studies T20-301 and T20-302 are shown in Table 2. Subjects had prior exposure to a median of 12 antiretrovirals for a median of 7 years.
| FUZEON+Background Regimen | Background Regimen | |
|---|---|---|
| N=663 | N=334 | |
| Sex | ||
| Male | 90% | 90% |
| Female | 10% | 10% |
| Race | ||
| White | 89% | 89% |
| Black | 8% | 7% |
| Mean Age (yr) | 42 | 43 |
| (range) | (16-67) | (24-82) |
| Median Baseline HIV-1 RNA (log10 copies/mL) (range) |
5.2 (3.5-6.7) |
5.1 (3.7-7.1) |
| Median Baseline CD4+ Cell Count (cells/mm3) (range) |
89 (1-994) |
97 (1-847) |
The disposition and efficacy outcomes of studies T20-301 and T20-302 are shown in Table 3.
| Outcomes | FUZEON+Background Regimen 90 mg bid N=663 |
Background Regimen N=334 |
|
|---|---|---|---|
|
|||
| Virological Responder (at least 1 log10 below baseline) |
304 (46%) | 61 (18%) | |
| Virological Non-responder: | |||
|
0 191 (29%) |
220 (66%) 12 (4%) |
|
| Continued Background Regimen (N=112) |
Switched to FUZEON (N=220) |
||
| Discontinued due to insufficient treatment response† | 37 (5%) | 13 (12%) | 22 (10%) |
| Discontinued due to adverse reactions/intercurrent illness/labs | 46 (7%) | 9 (8%) | 13 (6%) |
| Deaths | 15 (2%) | 5 (4%) | 2 (1%) |
| Discontinued due to injection: | |||
|
27 (4%) | NA | 10 (5%) |
|
18 (3%) | NA | 2 (1%) |
| Discontinued due to other reasons§ | 25 (4%) | 14 (13%) | 6 (3%) |
At 48 weeks, 154 (23%) of subjects in the FUZEON+background regimen and 27 (8%) in the background regimen alone had HIV RNA levels <50 copies/mL, and 225 (34%) of subjects receiving FUZEON+background regimen had HIV RNA levels <400 copies/mL compared to 44 (13%) in the background regimen alone. Subjects achieving HIV RNA levels <50 copies/mL were included in the <400 copies/mL category and both categories were incorporated in the overall virologic responder category of achieving HIV RNA at least 1 log10 below baseline.
The mean log change in HIV-1 RNA from baseline was -1.4 log10 copies/mL in subjects receiving FUZEON+background and -0.5 in those receiving background alone. The mean change in CD4+ cell count from baseline to week 48 was +91 cells/mm3 in the FUZEON+background arm and +45 cells/mm3 in the background alone arm.
Subjects in the FUZEON+background arm achieved a better virologic and immunologic outcome than subjects in the background alone arm across all subgroups based on baseline CD4+ cell count, baseline HIV-1 RNA, number of prior ARVs or number of active ARVs in the background regimen.
CONTRAINDICATIONS
FUZEON is contraindicated in patients with known hypersensitivity to FUZEON or any of its components (see WARNINGS).
WARNINGS
Local Injection Site Reactions (ISRs)
The majority of patients (98%) receiving FUZEON in the Phase 3 clinical trials had at least one local injection site reaction; ISRs occurred throughout treatment with FUZEON. Manifestations may include pain and discomfort, induration, erythema, nodules and cysts, pruritus, and ecchymosis (see ADVERSE REACTIONS). Reactions are often present at more than one injection site. Patients must be familiar with the FUZEON Injection Instructions in order to know how to inject FUZEON appropriately and how to monitor carefully for signs or symptoms of cellulitis or local infection.
Pneumonia
An increased rate of bacterial pneumonia was observed in subjects treated with FUZEON in the Phase 3 clinical trials compared to the control arm (see ADVERSE REACTIONS). It is unclear if the increased incidence of pneumonia is related to FUZEON use. However, because of this finding, patients with HIV infection should be carefully monitored for signs and symptoms of pneumonia, especially if they have underlying conditions which may predispose them to pneumonia. Risk factors for pneumonia included low initial CD4+ cell count, high initial viral load, intravenous drug use, smoking, and a prior history of lung disease (see ADVERSE REACTIONS).
Hypersensitivity Reactions
Systemic hypersensitivity reactions have been associated with FUZEON therapy and may recur on re-challenge. Hypersensitivity reactions have occurred in <1% of patients studied and have included combinations of: rash, fever, nausea and vomiting, chills, rigors, hypotension, and/or elevated serum liver transaminases. Other adverse events that may be immune mediated and have been reported in subjects receiving FUZEON include primary immune complex reaction, respiratory distress, glomerulonephritis, and Guillain-Barre syndrome. Patients developing signs and symptoms suggestive of a systemic hypersensitivity reaction should discontinue FUZEON and should seek medical evaluation immediately. Therapy with FUZEON should not be restarted following systemic signs and symptoms consistent with a hypersensitivity reaction. Risk factors that may predict the occurrence or severity of hypersensitivity to FUZEON have not been identified (see ADVERSE REACTIONS).
PRECAUTIONS
Non-HIV Infected Individuals
There is a theoretical risk that FUZEON use may lead to the production of anti-enfuvirtide antibodies which cross react with HIV gp41. This could result in a false positive HIV test with an ELISA assay; a confirmatory western blot test would be expected to be negative. FUZEON has not been studied in non-HIV infected individuals.
Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including FUZEON. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia [PCP] or tuberculosis), which may necessitate further evaluation and treatment.
Information for Patients
To assure safe and effective use of FUZEON, the following information and instructions should be given to patients:
- Patients should be informed that injection site reactions occur in almost all patients taking FUZEON. Patients must be familiar with the FUZEON Injection Instructions for instructions on how to appropriately inject FUZEON and how to carefully monitor for signs or symptoms of cellulitis or local infection. Patients should be instructed when to contact their healthcare provider about these reactions.
- Patients should be made aware that an increased rate of bacterial pneumonia was observed in subjects treated with FUZEON in Phase 3 clinical trials compared to the control arm. Patients should be advised to seek medical evaluation immediately if they develop signs or symptoms suggestive of pneumonia (cough with fever, rapid breathing, shortness of breath) (see WARNINGS).
- Patients should be advised of the possibility of a systemic hypersensitivity reaction to FUZEON. Patients should be advised to discontinue therapy and immediately seek medical evaluation if they develop signs/symptoms of systemic hypersensitivity such as combinations of rash, fever, nausea and vomiting, chills, rigors, and/or hypotension (see WARNINGS).
- FUZEON is not a cure for HIV-1 infection and patients may continue to contract illnesses associated with HIV-1 infection. The long-term effects of FUZEON are unknown at this time. FUZEON therapy has not been shown to reduce the risk of transmitting HIV-1 to others through sexual contact or blood contamination.
- FUZEON must be taken as part of a combination antiretroviral regimen. Use of FUZEON alone may lead to rapid development of virus resistant to FUZEON and possibly other agents of the same class.
- Patients and caregivers must be instructed in the use of aseptic technique when administering FUZEON in order to avoid injection site infections. Appropriate training for FUZEON reconstitution and self-injection must be given by a healthcare provider, including a careful review of the FUZEON Patient Package Insert and FUZEON Injection Instructions. The first injection should be performed under the supervision of an appropriately qualified healthcare provider. It is recommended that the patient and/or caregiver's understanding and use of aseptic injection techniques and procedures be periodically re-evaluated.
- Patients and caregivers should be instructed in the proper techniques for preparation, injection and disposal of needles and syringes (including not recapping needles) in order to avoid needle stick injuries. Patients should be told not to reuse needles or syringes, and be instructed in safe disposal procedures including the use of a puncture-resistant container for disposal of used needles and syringes. Patients must be instructed on the safe disposal of full containers as per local requirements. Caregivers who experience an accidental needle stick after patient injection should contact a healthcare provider immediately.
- Patients should contact their healthcare provider for any questions regarding the administration of FUZEON.
- Patients should inform their healthcare provider if they are pregnant, plan to become pregnant or become pregnant while taking this medication.
- Patients should inform their healthcare provider if they are breast-feeding.
- Patients should not change the dose or dosing schedule of FUZEON or any antiretroviral medication without consulting their healthcare provider.
- Patients should contact their healthcare provider immediately if they stop taking FUZEON or any other drug in their antiretroviral regimen.
- Patients should be told that they can obtain more information on the self-administration of FUZEON at www.FUZEON.com or by calling 1-877-4-FUZEON (1-877-438-9366).
Patients should be advised that no studies have been conducted on the ability to drive or operate machinery while taking FUZEON. If patients experience dizziness while taking FUZEON, they should be advised to talk to their healthcare provider before driving or operating machinery.
Drug Interactions
CYP450 Metabolized Drugs
Results from in vitro and in vivo studies suggest that enfuvirtide is unlikely to have significant drug interactions with concomitantly administered drugs metabolized by CYP450 enzymes (see CLINICAL PHARMACOLOGY).
Antiretroviral Agents
No drug interactions with other antiretroviral medications have been identified that would warrant alteration of either the enfuvirtide dose or the dose of the other antiretroviral medication.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long-term animal carcinogenicity studies of enfuvirtide have not been conducted.
Mutagenesis
Enfuvirtide was neither mutagenic nor clastogenic in a series of in vivo and in vitro assays including the Ames bacterial reverse mutation assay, a mammalian cell forward gene mutation assay in AS52 Chinese Hamster ovary cells or an in vivo mouse micronucleus assay.
Impairment of Fertility
Enfuvirtide produced no adverse effects on fertility in male or female rats at doses up to 1.6 times the maximum recommended adult human daily dose on a m2 basis.
Pregnancy
Pregnancy Category B. Reproduction studies have been performed in rats and rabbits at doses up to 27 times and 3.2 times the adult human dose on a m2 basis. The animal studies revealed no evidence of harm to the fetus from enfuvirtide. There are no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Antiretroviral Pregnancy Registry
To monitor maternal-fetal outcomes of pregnant women exposed to FUZEON and other antiretroviral drugs, an Antiretroviral Pregnancy Registry has been established. Physicians are encouraged to register patients by calling 1-800-258-4263.
Nursing Mothers
The Centers for Disease Control and Prevention recommends that HIV-infected mothers not breast-feed their infants to avoid the risk of postnatal transmission of HIV. It is not known whether enfuvirtide is excreted in human milk. Because of both the potential for HIV transmission and the potential for serious adverse reactions in nursing infants, mothers should be instructed not to breast-feed if they are receiving FUZEON.
Studies where radio-labeled 3H-enfuvirtide was administered to lactating rats indicated that radioactivity was present in the milk. It is not known whether the radioactivity in the milk was from radio-labeled enfuvirtide or from radio-labeled metabolites of enfuvirtide (ie, amino acids and peptide fragments).
Pediatric Use
The safety and pharmacokinetics of FUZEON have not been established in pediatric subjects below 6 years of age; limited efficacy data is available in pediatric subjects 6 years of age and older.
Fifty-two HIV-1 infected pediatric subjects ages 6 through 16 years have received FUZEON in two open-label, single-arm clinical trials. Adverse experiences, including ISRs, were similar to those observed in adult patients.
Study T20-204 was an open-label, multicenter trial that evaluated the safety and antiviral activity of FUZEON in treatment-experienced pediatric subjects. Eleven subjects from 6 to 12 years were enrolled (median age of 9 years). Median baseline CD4+ cell count was 495 cells/µL and the median baseline HIV-1 RNA was 4.6 log10 copies/mL.
Ten of the 11 study subjects completed 48 weeks of chronic therapy. By week 48, 6/11 (55%) subjects had ≥1 log10 decline in HIV-1 RNA and 4/11 (36%) subjects were below 400 copies/mL of HIV-1 RNA. The median changes from baseline (for the As Treated population) in HIV-1 RNA and CD4+ cell count were -1.48 log10 copies/mL and +122 cells/µL, respectively.
Study T20-310 is an ongoing, open-label, multicenter trial evaluating the pharmacokinetics, safety, and antiviral activity of FUZEON in treatment-experienced pediatric subjects and adolescents. Forty-one subjects from 6 through 16 years were enrolled (median age of 12 years). Median baseline CD4+ cell count was 117 cells/µL and the median baseline HIV-1 RNA was 5.1 log10 copies/mL. The evaluation of the antiviral activity is ongoing.
Geriatric Use
Clinical studies of FUZEON did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
ADVERSE REACTIONS
The overall safety profile of FUZEON is based on 2120 subjects who received at least 1 dose of FUZEON during various clinical trials. This includes 2051 adults, 658 of whom received the recommended dose for greater than 48 weeks, and 52 pediatric subjects.
Assessment of treatment-emergent adverse events is based on the pooled data from the two Phase 3 studies T20-301 and T20-302.
Local Injection Site Reactions
Local injection site reactions were the most frequent adverse events associated with the use of FUZEON. In Phase 3 clinical studies (T20-301 and T20-302), 98% of subjects had at least one local injection site reaction (ISR). A total of 7% of subjects discontinued treatment with FUZEON because of ISRs (4%) or difficulties with injecting FUZEON (3%) such as injection fatigue and inconvenience. Eighty-five percent of subjects experienced their first ISR during the initial week of treatment; ISRs continued to occur throughout treatment with FUZEON. For most subjects the severity of signs and symptoms associated with ISRs did not change during the 48 weeks of treatment. The majority of ISRs were associated with erythema, induration, the presence of nodules or cysts, and mild to moderate pain at the injection site (Table 4). In addition, the average duration of individual ISRs was between three and seven days in 41% of subjects and more than seven days in 24% of subjects. Also, the numbers of ISRs per subject at any one time was between six to 14 ISRs in 26% of subjects and more than 14 ISRs in 1.3% of subjects. Infection at the injection site (including abscess and cellulitis) was reported in 1.7% of subjects.
| N=663 | |||
|---|---|---|---|
| Event Category | Any Severity Grade | % of Patients with Grade 3 Reactions | % of Patients with Grade 4 Reactions |
|
|||
| Pain/Discomfort* | 96% | 11% | 0% |
| Induration | 90% | 39% | 18% |
| >25 but <50 mm | ≥50 mm | ||
| Erythema | 91% | 22% | 10% |
| >50 but <85 mm | ≥85 mm | ||
| Nodules and Cysts | 80% | 23% | 0.2% |
| >3 cm average diameter | draining | ||
| Pruritus† | 65% | 3% | NA |
| Ecchymosis | 52% | 5% | 2% |
| >3 but ≤5 cm | >5 cm | ||
Other Adverse Events
Systemic hypersensitivity reactions have been attributed to FUZEON (≤1%) and in some cases have recurred upon re-challenge (see WARNINGS).
In the T20-301 and T20-302 studies, after study week 8, patients on background alone who met protocol defined criteria for virological failure were permitted to revise their background regimens and add FUZEON. Exposure on FUZEON+background was 557 patient-years, and to background alone 162 patient-years. Due to this difference in exposure, safety results are expressed as the number of patients with an adverse event per 100 patient-years of exposure. For FUZEON+background, adverse events are also displayed by percent of subjects.
The events most frequently reported in subjects receiving FUZEON+background regimen, excluding injection site reactions, were diarrhea (38 per 100 patient-years or 31.7%), nausea (27 per 100 patient-years or 22.8%), and fatigue (24 per 100 patient-years or 20.2%). These events were also commonly observed in subjects that received background regimen alone: diarrhea (73 per 100 patient-years), nausea (50 per 100 patient-years), and fatigue (38 per 100 patient-years).
Treatment-emergent adverse events, regardless of causality and excluding ISRs, from Phase 3 studies are summarized for adult subjects, in Table 5. Any Grade 2 or above events occurring at ≥2 percent of subjects and at a higher rate in subjects treated with FUZEON are summarized in Table 5; events that occurred at a higher rate in the control arms are not displayed.
Rates of adverse events for patients who switched to FUZEON after virological failure were similar.
| Adverse Event (by System Organ Class) | FUZEON+Background Regimen (N=663) |
FUZEON+Background Regimen (N=663) |
Background Regimen (N=334) |
|---|---|---|---|
| 663 patients total | 557 total patient-years | 162 total patient-years | |
| % frequency | rate/100 patient-years | rate/100 patient-years | |
| Weight Decreased | 6.6% | 7.9 | 6.2 |
| Sinusitis | 6.0% | 7.2 | 4.9 |
| Abdominal Pain | 3.9% | 4.7 | 3.7 |
| Cough | 3.9% | 4.7 | 2.5 |
| Herpes Simplex | 3.5% | 4.1 | 3.7 |
| Appetite Decreased | 3.2% | 3.8 | 2.5 |
| Pancreatitis | 3.0% | 3.6 | 2.5 |
| Pain in Limb | 2.9% | 3.4 | 3.1 |
| Pneumonia (see text below) | 2.7% | 3.2 | 0.6 |
| Myalgia | 2.7% | 3.2 | 1.2 |
| Influenza-Like Illness | 2.4% | 2.9 | 1.9 |
| Folliculitis | 2.4% | 2.9 | 2.5 |
| Anorexia | 2.3% | 2.7 | 1.9 |
| Dry Mouth | 2.1% | 2.5 | 1.9 |
| Conjunctivitis | 2.0% | 2.3 | 1.9 |
The incidence of pneumonia was 2.7% or 3.2 events/100 patient-years in subjects receiving FUZEON+background regimen. On analysis of all diagnoses of pneumonia (pneumonia, bacterial pneumonia, bronchopneumonia, and related terms) in the Phase 3 clinical trials, an increased rate of bacterial pneumonia was observed in subjects treated with FUZEON compared to the control arm (6.9%, 6.7 pneumonia events per 100 patient-years versus 0.6 events per 100 patient-years, respectively). Approximately half of the study subjects with pneumonia required hospitalization. Three subject deaths in the FUZEON arm were attributed to pneumonia; all three had serious concomitant AIDS-related illnesses that contributed to their deaths. Risk factors for pneumonia included low initial CD4+ lymphocyte count, high initial viral load, intravenous drug use, smoking, and a prior history of lung disease. It is unclear if the increased incidence of pneumonia was related to FUZEON use. However, because of this, finding patients with HIV infection should be carefully monitored for signs and symptoms of pneumonia, especially if they have underlying conditions which may predispose them to pneumonia (see WARNINGS).
Less Common Events
The following adverse events have been reported in 1 or more subjects; however, a causal relationship to FUZEON has not been established.
Immune System Disorders: worsening abacavir hypersensitivity reaction
Renal and Urinary Disorders: glomerulonephritis; tubular necrosis; renal insufficiency; renal failure (including fatal cases)
Blood and Lymphatic Disorders: thrombocytopenia; neutropenia; fever; lymphadenopathy
Endocrine and Metabolic: hyperglycemia
Infections: sepsis; herpes simplex
Nervous System Disorders: taste disturbance; Guillain-Barre syndrome (fatal); sixth nerve palsy; peripheral neuropathy
Cardiac Disorders: unstable angina pectoris
Gastrointestinal Disorders: constipation; abdominal pain upper
General: asthenia
Hepatobiliary Disorders: toxic hepatitis; hepatic steatosis
Investigations: increased amylase; increased lipase; increased AST; increased GGT; increased triglycerides
Psychiatric Disorders: insomnia; depression; anxiety; suicide attempt
Respiratory, Thoracic, and Mediastinal Disorders: pneumopathy; respiratory distress; cough
Skin and Subcutaneous Tissue Disorders: pruritus
Laboratory Abnormalities
Table 6 shows the treatment-emergent laboratory abnormalities that occurred in at least 2 subjects per 100 patient-years and more frequently in those receiving FUZEON+background regimen than background regimen alone from studies T20-301 and T20-302.
| Laboratory Parameters | Grading | FUZEON+Background Regimen (N=663) |
FUZEON+Background Regimen (N=663) |
Background Regimen (N=334) |
|---|---|---|---|---|
|
||||
| 663 patients total | 557 total patient-years | 162 total patient-years | ||
| % frequency | rate/100 patient-years | rate/100 patient-years | ||
| Eosinophilia | ||||
| 1-2 × ULN (0.7 × 109/L) | 0.7-1.4 × 109/L | 9.1% | 10.8 | 3.7 |
| >2 × ULN (0.7 × 109/L) | >1.4× 109/L | 1.8% | 2.2 | 1.8 |
| ALT | ||||
| Grade 3 | >5-10× ULN | 4.1% | 4.8 | 4.3 |
| Grade 4 | >10 × ULN | 1.2% | 1.4 | 1.2 |
| Creatine Phosphokinase (U/L) | ||||
| Grade 3 | >5-10 × ULN | 6.9% | 8.3 | 8.0 |
| Grade 4 | >10 × ULN | 2.6% | 3.1 | 8.6 |
Adverse Events in Pediatric Patients
FUZEON has been studied in 52 pediatric subjects 6 through 16 years of age with duration of FUZEON exposure ranging from 1 dose to 134 weeks. Adverse experiences seen during clinical trials were similar to those observed in adult subjects.
OVERDOSAGE
There are no reports of human experience of acute overdose with FUZEON. The highest dose administered to 12 subjects in a clinical trial was 180 mg as a single dose subcutaneously. There is no specific antidote for overdose with FUZEON. Treatment of overdose should consist of general supportive measures.
DOSAGE AND ADMINISTRATION
Adults
The recommended dose of FUZEON is 90 mg (1 mL) twice daily injected subcutaneously into the upper arm, anterior thigh or abdomen. Each injection should be given at a site different from the preceding injection site, and only where there is no current injection site reaction from an earlier dose. FUZEON should not be injected into moles, scar tissue, bruises or the navel. Additional detailed information regarding the administration of FUZEON is described in the FUZEON Injection Instructions.
Pediatric Patients
No data are available to establish a dose recommendation of FUZEON in pediatric patients below the age of 6 years. In pediatric patients 6 years through 16 years of age, the recommended dosage of FUZEON is 2 mg/kg twice daily up to a maximum dose of 90 mg twice daily injected subcutaneously into the upper arm, anterior thigh or abdomen. Each injection should be given at a site different from the preceding injection site and only where there is no current injection site reaction from an earlier dose. FUZEON should not be injected into moles, scar tissue, bruises or the navel. Table 7 contains dosing guidelines for FUZEON based on body weight. Weight should be monitored periodically and the FUZEON dose adjusted accordingly.
| Weight | Dose per bid Injection (mg/dose) |
Injection Volume (90 mg enfuvirtide per mL) |
|
|---|---|---|---|
| Kilograms (kg) | Pounds (lbs) | ||
| 11.0 to 15.5 | 24 to 34 | 27 | 0.3 mL |
| 15.6 to 20.0 | >34 to 44 | 36 | 0.4 mL |
| 20.1 to 24.5 | >44 to 54 | 45 | 0.5 mL |
| 24.6 to 29.0 | >54 to 64 | 54 | 0.6 mL |
| 29.1 to 33.5 | >64 to 74 | 63 | 0.7 mL |
| 33.6 to 38.0 | >74 to 84 | 72 | 0.8 mL |
| 38.1 to 42.5 | >84 to 94 | 81 | 0.9 mL |
| ≥42.6 | >94 | 90 | 1.0 mL |
Directions for Use
For more detailed instructions, see FUZEON Injection Instructions.
Subcutaneous Administration
FUZEON must only be reconstituted with 1.1 mL of Sterile Water for Injection. After adding sterile water, the vial should be gently tapped for 10 seconds and then gently rolled between the hands to avoid foaming and to ensure all particles of drug are in contact with the liquid and no drug remains on the vial wall. The vial should then be allowed to stand until the powder goes completely into solution, which could take up to 45 minutes. Reconstitution time can be reduced by gently rolling the vial between the hands until the product is completely dissolved. Before the solution is withdrawn for administration, the vial should be inspected visually to ensure that the contents are fully dissolved in solution, and that the solution is clear, colorless and without bubbles or particulate matter. If the FUZEON is foamy or jelled, allow more time for it to dissolve. If there is evidence of particulate matter, the vial must not be used and should be returned to the pharmacy.
FUZEON contains no preservatives. Once reconstituted, FUZEON should be injected immediately or kept refrigerated in the original vial until use. Reconstituted FUZEON must be used within 24 hours. The subsequent dose of FUZEON can be reconstituted in advance and must be stored in the refrigerator in the original vial and used within 24 hours. Refrigerated reconstituted solution should be brought to room temperature before injection and the vial should be inspected visually again to ensure that the contents are fully dissolved in solution and that the solution is clear, colorless, and without bubbles or particulate matter.
The reconstituted solution should be injected subcutaneously in the upper arm, abdomen or anterior thigh. The injection should be given at a site different from the preceding injection site and only where there is no current injection site reaction. Also, do not inject into moles, scar tissue, bruises or the navel. A vial is suitable for single use only; unused portions must be discarded (see FUZEON Injection Instructions).
Patients should contact their healthcare provider for any questions regarding the administration of FUZEON. Information about the self-administration of FUZEON may also be obtained by calling the toll-free number 1-877-4-FUZEON (1-877-438-9366) or at the FUZEON website, www.FUZEON.com. Patients should be taught to recognize the signs and symptoms of injection site reactions and instructed when to contact their healthcare provider about these reactions.
HOW SUPPLIED
FUZEON (enfuvirtide) for Injection is a white to off-white, sterile, lyophilized powder and it is packaged in a single-use clear glass vial containing 108 mg of enfuvirtide for the delivery of approximately 90 mg/1 mL when reconstituted with 1.1 mL of Sterile Water for Injection.
FUZEON is available in a Convenience Kit containing 60 single-use vials of FUZEON (90 mg strength), 60 vials (2 cartons of 30 each) of Sterile Water for Injection (1.1 mL per vial), 60 reconstitution syringes (3 cc), 60 administration syringes (1 cc), alcohol wipes, Package Insert, Patient Package Insert, and Injection Instruction Guide (NDC 0004-0380-39).
Storage Conditions
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Reconstituted solution should be stored under refrigeration at 2° to 8°C (36° to 46°F) and used within 24 hours.
Roche and FUZEON are trademarks of Hoffmann-La Roche Inc.
FUZEON has been jointly developed by Trimeris, Inc. and Hoffmann-La Roche Inc. FUZEON is manufactured by Hoffmann-La Roche Inc.
| Distributed by: |
Licensed from: |
|
|
|
|
|
27899075
Copyright © 2003-2005 by Roche Laboratories Inc. and Trimeris, Inc. All rights reserved.
| Fuzeon (enfuvirtide) | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
Revised: 05/2006Roche Laboratories Inc.


