FAMVIR
-
famciclovir tablet, film coated
Novartis Pharmaceuticals Corporation
----------
FamvirFamvir®
(famciclovir)
Tablets
Rx only
PRESCRIBING INFORMATION
DESCRIPTION
Famvir® (famciclovir) contains famciclovir, an orally administered prodrug of the antiviral agent penciclovir. Chemically, famciclovir is known as 2-[2-(2-amino-9H-purin-9-yl)ethyl]-1,3-propanediol diacetate. Its molecular formula is C14H19N504; its molecular weight is 321.3. It is a synthetic acyclic guanine derivative and has the following structure
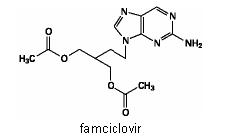
Famciclovir is a white to pale yellow solid. It is freely soluble in acetone and methanol, and sparingly soluble in ethanol and isopropanol. At 25°C famciclovir is freely soluble (>25% w/v) in water initially, but rapidly precipitates as the sparingly soluble (2%-3% w/v) monohydrate. Famciclovir is not hygroscopic below 85% relative humidity. Partition coefficients are: octanol/water (pH 4.8) P=1.09 and octanol/phosphate buffer (pH 7.4) P=2.08.
Tablets for Oral Administration
Each white, film-coated tablet contains famciclovir. The 125-mg and 250-mg tablets are round; the 500-mg tablets are oval. Inactive ingredients consist of hydroxypropyl cellulose, hydroxypropyl methylcellulose, lactose, magnesium stearate, polyethylene glycols, sodium starch glycolate and titanium dioxide.
MICROBIOLOGY
Mechanism of Antiviral Action
Famciclovir undergoes rapid biotransformation to the active antiviral compound penciclovir, which has demonstrated inhibitory activity against herpes simplex virus types 1 (HSV-1) and 2 (HSV-2) and varicella zoster virus (VZV). In cells infected with HSV-1, HSV-2 or VZV, the viral thymidine kinase phosphorylates penciclovir to a monophosphate form that, in turn, is converted to penciclovir triphosphate by cellular kinases. In vitro studies demonstrate that penciclovir triphosphate inhibits HSV-2 DNA polymerase competitively with deoxyguanosine triphosphate. Consequently, herpes viral DNA synthesis and, therefore, replication are selectively inhibited.
Penciclovir triphosphate has an intracellular half-life of 10 hours in HSV-1-, 20 hours in HSV-2- and 7 hours in VZV-infected cells grown in culture; however, the clinical significance is unknown.
Antiviral Activity
In cell culture studies, penciclovir is inhibitory to the following herpes viruses (listed in decreasing order of potency): HSV-1, HSV-2 and VZV. Sensitivity test results, expressed as the concentration of the drug required to inhibit the growth of the virus by 50% (EC50) or 99% (EC99) in cell culture, vary greatly depending upon a number of factors, including the assay protocols, and in particular the cell type used. See Table 1.
| Method of Assay | Virus Type | Cell Type | EC50 | EC99 |
| (mcg/mL) | ||||
| Plaque Reduction | VZV (c.i.) | MRC-5 | 5.0 ± 3.0 | |
| VZV (c.i.) | Hs68 | 0.9 ± 0.4 | ||
| HSV-1 (c.i.) | MRC-5 | 0.2 – 0.6 | ||
| HSV-1 (c.i.) | WISH | 0.04 – 0.5 | ||
| HSV-2 (c.i.) | MRC-5 | 0.9 – 2.1 | ||
| HSV-2 (c.i.) | WISH | 0.1 – 0.8 | ||
| Virus Yield | HSV-1 (c.i.) | MRC-5 | 0.4 – 0.5 | |
| Reduction | HSV-2 (c.i.) | MRC-5 | 0.6 – 0.7 | |
| DNA Synthesis | VZV (Ellen) | MRC-5 | 0.1 | |
| Inhibition | HSV-1 (SC16) | MRC-5 | 0.04 | |
| HSV-2 (MS) | MRC-5 | 0.05 | ||
(c.i.) = clinical isolates.
Resistance
Penciclovir-resistant mutants of HSV and VZV can result from mutations in the viral thymidine kinase (TK) and DNA polymerase genes. Mutations in the viral TK gene may lead to complete loss of TK activity (TK negative), reduced levels of TK activity (TK partial), or alteration in the ability of viral TK to phosphorylate the drug without an equivalent loss in the ability to phosphorylate thymidine (TK altered). The most commonly encountered acyclovir-resistant mutants that are TK negative are also resistant to penciclovir. The possibility of viral resistance to penciclovir should be considered in patients who fail to respond or experience recurrent viral shedding during therapy.
CLINICAL PHARMACOLOGY
Pharmacokinetics
Absorption and Bioavailability
Famciclovir is the diacetyl 6-deoxy analog of the active antiviral compound penciclovir. Following oral administration, little or no famciclovir is detected in plasma or urine.
The absolute bioavailability of penciclovir is 77±8% as determined following the administration of a 500-mg famciclovir oral dose and a 400-mg penciclovir intravenous dose to 12 healthy male subjects.
Penciclovir concentrations increased in proportion to dose over a famciclovir dose range of 125 mg to 1000 mg administered as a single dose. Single oral-dose administration of 125-mg, 250-mg, 500-mg, or 1000-mg famciclovir to healthy male volunteers across 17 studies gave the following pharmacokinetic parameters:
| Dose | AUC (0-inf)† (mcg hr/mL) | Cmax‡ (mcg/mL) | Tmax§ (h) |
| 125 mg | 2.24 | 0.8 | 0.9 |
| 250 mg | 4.48 | 1.6 | 0.9 |
| 500 mg | 8.95 | 3.3 | 0.9 |
| 1000 mg | 17.9 | 6.6 | 0.9 |
†AUC (0-inf) (mcg hr/mL)=area under the plasma concentration-time profile extrapolated to infinity.
‡Cmax (mcg/mL)=maximum observed plasma concentration.
§Tmax (h)= time to Cmax.
Following oral single-dose administration of 500-mg famciclovir to seven patients with herpes zoster, the mean ± SD AUC, Cmax, and Tmax were 12.1±1.7 mcg hr/mL, 4.0±0.7 mcg/mL, and 0.7±0.2 hours, respectively. The AUC of penciclovir was approximately 35% greater in patients with herpes zoster as compared to healthy volunteers. Some of this difference may be due to differences in renal function between the two groups.
There is no accumulation of penciclovir after the administration of 500-mg famciclovir t.i.d. for 7 days.
Penciclovir Cmax decreased approximately 50% and Tmax was delayed by 1.5 hours when a capsule formulation of famciclovir was administered with food (nutritional content was approximately 910 Kcal and 26% fat). There was no effect on the extent of availability (AUC) of penciclovir. There was an 18% decrease in Cmax and a delay in Tmax of about 1 hour when famciclovir was given 2 hours after a meal as compared to its administration 2 hours before a meal. Because there was no effect on the extent of systemic availability of penciclovir, it appears that Famvir® (famciclovir) can be taken without regard to meals.
Distribution
The volume of distribution (Vdβ) was 1.08±0.17 L/kg in 12 healthy male subjects following a single intravenous dose of penciclovir at 400 mg administered as a 1-hour intravenous infusion.
Penciclovir is <20% bound to plasma proteins over the concentration range of 0.1 to 20 mcg/mL. The blood/plasma ratio of penciclovir is approximately 1.
Metabolism
Following oral administration, famciclovir is deacetylated and oxidized to form penciclovir. Metabolites that are inactive include 6-deoxy penciclovir, monoacetylated penciclovir, and 6-deoxy monoacetylated penciclovir (5%, <0.5% and <0.5% of the dose in the urine, respectively). Little or no famciclovir is detected in plasma or urine.
An in vitro study using human liver microsomes demonstrated that cytochrome P450 does not play an important role in famciclovir metabolism. The conversion of 6-deoxy penciclovir to penciclovir is catalyzed by aldehyde oxidase.
Elimination
Approximately 94% of administered radioactivity was recovered in urine over 24 hours (83% of the dose was excreted in the first 6 hours) after the administration of 5 mg/kg radiolabeled penciclovir as a 1-hour infusion to three healthy male volunteers. Penciclovir accounted for 91% of the radioactivity excreted in the urine.
Following the oral administration of a single 500-mg dose of radiolabeled famciclovir to three healthy male volunteers, 73% and 27% of administered radioactivity were recovered in urine and feces over 72 hours, respectively. Penciclovir accounted for 82% and 6-deoxy penciclovir accounted for 7% of the radioactivity excreted in the urine. Approximately 60% of the administered radiolabeled dose was collected in urine in the first 6 hours.
After intravenous administration of penciclovir in 48 healthy male volunteers, mean ± S.D. total plasma clearance of penciclovir was 36.6±6.3 L/hr (0.48±0.09 L/hr/kg). Penciclovir renal clearance accounted for 74.5±8.8% of total plasma clearance.
Renal clearance of penciclovir following the oral administration of a single 500-mg dose of famciclovir to 109 healthy male volunteers was 27.7±7.6 L/hr.
The plasma elimination half-life of penciclovir was 2.0±0.3 hours after intravenous administration of penciclovir to 48 healthy male volunteers and 2.3±0.4 hours after oral administration of 500-mg famciclovir to 124 healthy male volunteers. The half-life in 17 patients with herpes zoster was 2.8±1.0 hours and 2.7±1.0 hours after single and repeated doses, respectively.
HIV-Infected Patients
Following oral administration of a single dose of 500-mg famciclovir (the oral prodrug of penciclovir) to HIV-positive patients, the pharmacokinetic parameters of penciclovir were comparable to those observed in healthy subjects.
Renal Insufficiency
Apparent plasma clearance, renal clearance, and the plasma-elimination rate constant of penciclovir decreased linearly with reductions in renal function. After the administration of a single 500-mg famciclovir oral dose (n=27) to healthy volunteers and to volunteers with varying degrees of renal insufficiency (CLCR ranged from 6.4 to 138.8 mL/min.), the following results were obtained (Table 3):
| Parameter
(mean ± S.D.) | CLCR† ≥60
(mL/min.) (n=15) | CLCR 40-59
(mL/min.) (n=5) | CLCR 20-39
(mL/min.) (n=4) | CLCR <20
(mL/min.) (n=3) |
| CLCR (mL/min) | 88.1 ± 20.6 | 49.3 ± 5.9 | 26.5 ± 5.3 | 12.7 ± 5.9 |
| CLR (L/hr) | 30.1 ± 10.6 | 13.0 ± 1.3‡ | 4.2 ± 0.9 | 1.6 ± 1.0 |
| CL/F§ (L/hr) | 66.9 ± 27.5 | 27.3 ± 2.8 | 12.8 ± 1.3 | 5.8 ± 2.8 |
| Half-life (hr) | 2.3 ± 0.5 | 3.4 ± 0.7 | 6.2 ± 1.6 | 13.4 ± 10.2 |
†CLCR is measured creatinine clearance.
‡n=4.
§CL/F consists of bioavailability factor and famciclovir to penciclovir conversion factor.
In a multiple-dose study of famciclovir conducted in subjects with varying degrees of renal impairment (n=18), the pharmacokinetics of penciclovir were comparable to those after single doses.
A dosage adjustment is recommended for patients with renal insufficiency (see DOSAGE AND ADMINISTRATION).
Hepatic Insufficiency
Well-compensated chronic liver disease (chronic hepatitis [n=6], chronic ethanol abuse [n=8], or primary biliary cirrhosis [n=1]) had no effect on the extent of availability (AUC) of penciclovir following a single dose of 500-mg famciclovir. However, there was a 44% decrease in penciclovir mean maximum plasma concentration and the time to maximum plasma concentration was increased by 0.75 hours in patients with hepatic insufficiency compared to normal volunteers. No dosage adjustment is recommended for patients with well-compensated hepatic impairment. The pharmacokinetics of penciclovir have not been evaluated in patients with severe uncompensated hepatic impairment.
Elderly Subjects
Based on cross-study comparisons, mean penciclovir AUC was 40% larger and penciclovir renal clearance was 22% lower after the oral administration of famciclovir in elderly volunteers (n=18, age 65 to 79 years) compared to younger volunteers. Some of this difference may be due to differences in renal function between the two groups. No famciclovir dosage adjustment based on age is recommended unless renal function is impaired (see DOSAGE AND ADMINISTRATION).
Gender
The pharmacokinetics of penciclovir were evaluated in 18 healthy male and 18 healthy female volunteers after single-dose oral administration of 500-mg famciclovir. AUC of penciclovir was 9.3±1.9 mcg hr/mL and 11.1±2.1 mcg hr/mL in males and females, respectively. Penciclovir renal clearance was 28.5±8.9 L/hr and 21.8±4.3 L/hr, respectively. These differences were attributed to differences in renal function between the two groups. No famciclovir dosage adjustment based on gender is recommended.
Pediatric Patients
The pharmacokinetics of famciclovir or penciclovir have not been evaluated in patients <18 years of age.
Race
The pharmacokinetics of famciclovir or penciclovir with respect to race have not been evaluated.
Drug Interactions
Effects on Penciclovir
No clinically significant alterations in penciclovir pharmacokinetics were observed following single-dose administration of 500-mg famciclovir after pre-treatment with multiple doses of allopurinol, cimetidine, theophylline, zidovudine, or promethazine, when given shortly after an antacid (magnesium and aluminium hydroxide), or concomitantly with emtricitabine. No clinically significant effect on penciclovir pharmacokinetics was observed following multiple-dose (t.i.d.) administration of famciclovir (500 mg) with multiple doses of digoxin.
Clinical interaction studies of famciclovir with cimetidine and promethazine, in vitro inhibitors of aldehyde oxidase, did not show relevant effects on the formation of penciclovir. Raloxifene, a potent aldehyde oxidase inhibitor in vitro, could decrease the formation of penciclovir. However, a clinical drug-drug interaction study to determine the magnitude of interaction between pencyclovir and raloxifene has not been conducted.
Effects of Famciclovir on Co-administered Drugs
The steady-state pharmacokinetics of digoxin were not altered by concomitant administration of multiple doses of famciclovir (500 mg t.i.d.). No clinically significant effect on the pharmacokinetics of zidovudine, its metabolite zidovudine glucuronide or emtricitabine was observed following a single oral dose of 500-mg famciclovir co-administered with zidovudine or emtricitabine.
An in vitro study using human liver microsomes suggests that famciclovir is not an inhibitor of CYP4503A4 enzymes.
CLINICAL TRIALS
Herpes Zoster
Famvir® (famciclovir) was studied in a placebo-controlled, double-blind trial of 419 immunocompetent adults with uncomplicated herpes zoster. Comparisons included Famvir 500 mg t.i.d., Famvir 750 mg t.i.d., or placebo. Treatment was begun within 72 hours of initial lesion appearance and therapy was continued for 7 days.
The median time to full crusting in Famvir-treated patients was 5 days compared to 7 days in placebo-treated patients. The times to full crusting, loss of vesicles, loss of ulcers, and loss of crusts were shorter for Famvir 500 mg-treated patients than for placebo-treated patients in the overall study population. The effects of Famvir were greater when therapy was initiated within 48 hours of rash onset; it was also more pronounced in patients 50 years of age or older. Among the 65.2% of patients with at least one positive viral culture, Famvir-treated patients had a shorter median duration of viral shedding than placebo-treated patients (1 day and 2 days, respectively).
There were no overall differences in the duration of pain before rash healing between Famvir- and placebo-treated groups. In addition, there was no difference in the incidence of pain after rash healing (postherpetic neuralgia) between the treatment groups. In the 186 patients (44.4% of total study population) who did develop postherpetic neuralgia, the median duration of postherpetic neuralgia was shorter in patients treated with Famvir 500 mg than in those treated with placebo (63 days and 119 days, respectively). No additional efficacy was demonstrated with higher doses of Famvir.
A double-blind controlled trial in 545 immunocompetent adults with uncomplicated herpes zoster treated within 72 hours of initial lesion appearance compared three doses of Famvir to acyclovir 800 mg 5 times per day. Times to full lesion crusting and times to loss of acute pain were comparable for all groups and there were no statistically significant differences in the time to loss of postherpetic neuralgia between Famvir- and acyclovir-treated groups.
Herpes Simplex Infections
Recurrent Genital Herpes
In one placebo-controlled trial, 329 immunocompetent adults with a recurrence of genital herpes were treated with Famvir 1000 mg b.i.d. (n=163) or placebo (n=166) for 1 day. Treatment was initiated within 6 hours of either symptom onset or lesion appearance. Among patients with non-aborted lesions, the median time to healing (from start of therapy to re-epithelialization) in Famvir-treated patients (n=125) was 4.3 days compared to 6.1 days in placebo-treated patients (n=145). The median difference in time to healing between placebo and the Famvir-treated groups was 1.2 days (95% CI: 0.5 – 2.0). Twenty-three percent of Famvir-treated patients had aborted lesions (no development beyond erythema) compared to 13% in placebo-treated patients. The median time to loss of all symptoms (e.g., burning, itching, pain, tenderness, tingling), was 3.3 days in Famvir-treated patients vs. 5.4 days in placebo-treated patients.
Suppression of Recurrent Genital Herpes
934 immunocompetent adults with a history of 6 or more recurrences per year were randomized into two double-blind, 1-year, placebo-controlled trials. Comparisons included Famvir 125 mg t.i.d., 250 mg b.i.d., 250 mg t.i.d. and placebo. At one year, 60% to 65% of patients were still receiving Famvir and 25% were receiving placebo treatment. Patient reported recurrence rates for the 250 mg b.i.d. dose at 6 and 12 months as shown in Table 4.
| Recurrence Rates
at 6 Months | Recurrence Rates
at 12 Months |
|||
| Famvir ®
250 mg b.i.d. (n=236) | Placebo
(n=233) | Famvir ®
250 mg b.i.d. (n=236) | Placebo
(n=233) |
|
| Recurrence-free | 39% | 10% | 29% | 6% |
| Recurrences† | 47% | 74% | 53% | 78% |
| Lost to Follow-up‡ | 14% | 16% | 17% | 16% |
† Based on patient reported data; not necessarily confirmed by a physician.
‡ Patients recurrence-free at time of last contact prior to withdrawal.
Famvir-treated patients had approximately 1/5 the median number of recurrences as compared to placebo-treated patients.
Higher doses of Famvir were not associated with an increase in efficacy.
Herpes Labialis (Cold Sores)
In one placebo-controlled trial, 701 immunocompetent adults with recurrent herpes labialis were treated with Famvir 1500 mg as a single dose (n=227), Famvir 750 mg b.i.d. (n=220) or placebo (n=254) for 1 day. Treatment was initiated within 1 hour of symptom onset. Among patients with non-aborted lesions, the median time to healing was 4.4 days in the Famvir 1500 mg single dose group (n=152) compared to 6.2 days in the placebo group (n=168). The median difference in time to healing between the placebo and Famvir treated group was 1.3 days (95% CI: 0.6 – 2.0). No differences in aborted lesions (beyond the papular stage) were observed between subjects receiving Famvir or placebo: 33% for Famvir 1500 mg and 34% for placebo. The median time to loss of pain and tenderness was 1.7 days in Famvir 1500 mg once-treated patients versus 2.9 days in placebo-treated patients.
Recurrent Mucocutaneous Herpes Simplex Infection in HIV-Infected Patients
A randomized, double-blind, multicenter study compared famciclovir 500 mg twice daily for 7 days (n=150) with oral acyclovir 400 mg 5 times daily for 7 days (n=143) in HIV-infected patients with recurrent mucocutaneous HSV infection treated within 48 hours of lesion onset. Approximately 40% of patients had a CD4 count below 200 cells/mm3, 54% of patients had anogenital lesions and 35% had orolabial lesions. Famciclovir therapy was comparable to oral acyclovir in reducing new lesion formation and in time to complete healing.
INDICATIONS AND USAGE
Herpes Zoster
Famvir® (famciclovir) is indicated for the treatment of acute herpes zoster (shingles).
Herpes Simplex Infections
Famvir is indicated for:
- treatment or suppression of recurrent genital herpes in immunocompetent patients.
- treatment of recurrent herpes labialis (cold sores) in immunocompetent patients.
- treatment of recurrent mucocutaneous herpes simplex infections in HIV-infected patients.
CONTRAINDICATIONS
Famvir® (famciclovir) is contraindicated in patients with known hypersensitivity to the product, its components, and Denavir® (penciclovir cream).
PRECAUTIONS
General
The efficacy of Famvir® (famciclovir) has not been established for initial episode genital herpes infection, ophthalmic zoster, disseminated zoster or in immunocompromised patients with herpes zoster.
Dosage adjustment is recommended when administering Famvir to patients with creatinine clearance values < 60 mL/min. (see DOSAGE AND ADMINISTRATION). In patients with underlying renal disease who have received inappropriately high doses of Famvir for their level of renal function, acute renal failure has been reported.
Famvir 125 mg, 250 mg and 500 mg tablets contain lactose (26.9 mg, 53.7 mg and 107.4 mg, respectively). Patients with rare hereditary problems of galactose intolerance, a severe lactase deficiency or glucose-galactose malabsorption should not take Famvir 125 mg, 250 mg and 500 mg tablets.
Information for Patients
Patients should be informed that Famvir is not a cure for genital herpes. There are no data evaluating whether Famvir will prevent transmission of infection to others. As genital herpes is a sexually transmitted disease, patients should avoid contact with lesions or intercourse when lesions and/or symptoms are present to avoid infecting partners. Genital herpes can also be transmitted in the absence of symptoms through asymptomatic viral shedding. If medical management of recurrent episodes is indicated, patients should be advised to initiate therapy at the first sign or symptom.
There is no evidence that Famvir will affect the ability of a patient to drive or to use machines. However, patients who experience dizziness, somnolence, confusion or other central nervous system disturbances while taking Famvir should refrain from driving or operating machinery.
Drug Interactions
Concurrent use with probenecid or other drugs significantly eliminated by active renal tubular secretion may result in increased plasma concentrations of penciclovir.
The conversion of 6-deoxy penciclovir to penciclovir is catalyzed by aldehyde oxidase. Interactions with other drugs metabolized by this enzyme and/or inhibiting this enzyme could potentially occur.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Famciclovir was administered orally unless otherwise stated.
Carcinogenesis
Two-year dietary carcinogenicity studies with famciclovir were conducted in rats and mice. An increase in the incidence of mammary adenocarcinoma (a common tumor in animals of this strain) was seen in female rats receiving the high dose of 600 mg/kg/day (1.1 to 4.5x the human systemic exposure at the recommended total daily oral dose ranging between 2000 mg and 500 mg, based on area under the plasma concentration curve comparisons [24 hr AUC] for penciclovir). No increases in tumor incidence were reported in male rats treated at doses up to 240 mg/kg/day (0.7 to 2.7x the human AUC), or in male and female mice at doses up to 600 mg/kg/day (0.3 to 1.2x the human AUC).
Mutagenesis
Famciclovir and penciclovir (the active metabolite of famciclovir) were tested for genotoxic potential in a battery of in vitro and in vivo assays. Famciclovir and penciclovir were negative in in vitro tests for gene mutations in bacteria (S. typhimurium and E. coli) and unscheduled DNA synthesis in mammalian HeLa 83 cells (at doses up to 10,000 and 5,000 mcg/plate, respectively). Famciclovir was also negative in the L5178Y mouse lymphoma assay (5000 mcg/mL), the in vivo mouse micronucleus test (4800 mg/kg), and rat dominant lethal study (5000 mg/kg). Famciclovir induced increases in polyploidy in human lymphocytes in vitro in the absence of chromosomal damage (1200 mcg/mL). Penciclovir was positive in the L5178Y mouse lymphoma assay for gene mutation/chromosomal aberrations, with and without metabolic activation (1000 mcg/mL). In human lymphocytes, penciclovir caused chromosomal aberrations in the absence of metabolic activation (250 mcg/mL). Penciclovir caused an increased incidence of micronuclei in mouse bone marrow in vivo when administered intravenously at doses highly toxic to bone marrow (500 mg/kg), but not when administered orally.
Impairment of Fertility
Testicular toxicity was observed in rats, mice, and dogs following repeated administration of famciclovir or penciclovir. Testicular changes included atrophy of the seminiferous tubules, reduction in sperm count, and/or increased incidence of sperm with abnormal morphology or reduced motility. The degree of toxicity to male reproduction was related to dose and duration of exposure. In male rats, decreased fertility was observed after 10 weeks of dosing at 500 mg/kg/day (1.4 to 5.7x the human AUC). The no observable effect level for sperm and testicular toxicity in rats following chronic administration (26 weeks) was 50 mg/kg/day (0.15 to 0.6x the human systemic exposure based on AUC comparisons). Testicular toxicity was observed following chronic administration to mice (104 weeks) and dogs (26 weeks) at doses of 600 mg/kg/day (0.3 to 1.2x the human AUC) and 150 mg/kg/day (1.3 to 5.1x the human AUC), respectively.
Famciclovir had no effect on general reproductive performance or fertility in female rats at doses up to 1000 mg/kg/day (2.7 to 10.8x the human AUC).
Two placebo-controlled studies in a total of 130 otherwise healthy men with a normal sperm profile over an 8-week baseline period and recurrent genital herpes receiving oral Famvir (250 mg b.i.d.) (n=66) or placebo (n=64) therapy for 18 weeks showed no evidence of significant effects on sperm count, motility or morphology during treatment or during an 8-week follow-up.
Pregnancy
Teratogenic Effects–Pregnancy Category B
Famciclovir was tested for effects on embryo-fetal development in rats and rabbits at oral doses up to 1000 mg/kg/day (approximately 2.7 to 10.8x and 1.4 to 5.4x the human systemic exposure to penciclovir based on AUC comparisons for the rat and rabbit, respectively) and intravenous doses of 360 mg/kg/day in rats (1.5 to 6x the human dose based on body surface area [BSA] comparisons) or 120 mg/kg/day in rabbits (1.1 to 4.5x the human dose [BSA]). No adverse effects were observed on embryo-fetal development. Similarly, no adverse effects were observed following intravenous administration of penciclovir to rats (80 mg/kg/day, 0.3 to 1.3x the human dose [BSA]) or rabbits (60 mg/kg/day, 0.5 to 2.1x the human dose [BSA]). There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, famciclovir should be used during pregnancy only if the benefit to the patient clearly exceeds the potential risk to the fetus.
Pregnancy Exposure Registry
To monitor maternal-fetal outcomes of pregnant women exposed to Famvir, Novartis Pharmaceuticals Corporation maintains a Famvir Pregnancy Registry. Physicians are encouraged to register their patients by calling (888) 669-6682.
Nursing Mothers
Following oral administration of famciclovir to lactating rats, penciclovir was excreted in breast milk at concentrations higher than those seen in the plasma. It is not known whether it is excreted in human milk. There are no data on the safety of Famvir in infants. Famciclovir should not be used in nursing mothers unless the potential benefits are considered to outweigh the potential risks associated with treatment.
Usage in Children
Safety and efficacy in children under the age of 18 years have not been established.
Geriatric Use
Of 816 patients with herpes zoster in clinical studies who were treated with Famvir, 248 (30.4%) were ≥65 years of age and 103 (13%) were ≥75 years of age. No overall differences were observed in the incidence or types of adverse events between younger and older patients.
Of 610 patients with recurrent herpes simplex (type 1 or type 2) in clinical studies who were treated with Famvir, 26 (4.3%) were >65 years of age and 7 (1.1%) were >75 years of age. Clinical studies of Famvir did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
In general, appropriate caution should be exercised in the administration and monitoring of Famvir in elderly patients reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
ADVERSE REACTIONS
Immunocompetent Patients
The safety of Famvir® (famciclovir) has been evaluated in clinical studies involving 816 Famvir-treated patients with herpes zoster (Famvir, 250 mg t.i.d. to 750 mg t.i.d.); 163 Famvir-treated patients with recurrent genital herpes (Famvir, 1000 mg b.i.d.); 1,197 patients with recurrent genital herpes treated with Famvir as suppressive therapy (125 mg q.d. to 250 mg t.i.d.) of which 570 patients received Famvir (open-labeled and/or double-blind) for at least 10 months; and 447 Famvir-treated patients with herpes labialis (Famvir, 1500 mg once or 750 mg b.i.d.). Table 5 lists selected adverse events.
| Incidence | ||||||||
| Herpes Zoster† | Recurrent
Genital Herpes‡ | Genital Herpes-
Suppression§ | Herpes Labialis‡ | |||||
| Event | Famvir® | Placebo | Famvir ® | Placebo | Famvir® | Placebo | Famvir® | Placebo |
| 500 mg | 1 gram | 250 mg | 1500 mg | |||||
| t.i.d* | b.i.d* | b.i.d* | single dose* | |||||
| (n=273) | (n=146) | (n=163) | (n=166) | (n=458) | (n=63) | (n=227) | (n=254) | |
| % | % | % | % | % | % | % | % | |
| Nervous System | ||||||||
| Headache | 22.7 | 17.8 | 13.5 | 5.4 | 39.3 | 42.9 | 9.7 | 6.7 |
| Paresthesia | 2.6 | 0.0 | 0.0 | 0.0 | 0.9 | 0.0 | 0.0 | 0.0 |
| Migraine | 0.7 | 0.7 | 0.6 | 0.6 | 3.1 | 0.0 | 0.0 | 0.0 |
| Gastrointestinal | ||||||||
| Nausea | 12.5 | 11.6 | 2.5 | 3.6 | 7.2 | 9.5 | 2.2 | 3.9 |
| Diarrhea | 7.7 | 4.8 | 4.9 | 1.2 | 9.0 | 9.5 | 1.8 | 0.8 |
| Vomiting | 4.8 | 3.4 | 1.2 | 0.6 | 3.1 | 1.6 | 0.0 | 0.0 |
| Flatulence | 1.5 | 0.7 | 0.6 | 0.0 | 4.8 | 1.6 | 0.0 | 0.0 |
| Abdominal Pain | 1.1 | 3.4 | 0.0 | 1.2 | 7.9 | 7.9 | 0.0 | 0.4 |
| Body as a Whole | ||||||||
| Fatigue | 4.4 | 3.4 | 0.6 | 0.0 | 4.8 | 3.2 | 1.3 | 0.4 |
| Skin and Appendages | ||||||||
| Pruritus | 3.7 | 2.7 | 0.0 | 0.6 | 2.2 | 0.0 | 0.0 | 0.0 |
| Rash | 0.4 | 0.7 | 0.0 | 0.0 | 3.3 | 1.6 | 0.0 | 0.0 |
| Reproductive Female | ||||||||
| Dysmenorrhea | 0.0 | 0.7 | 1.8 | 0.6 | 7.6 | 6.3 | 0.9 | 0.0 |
*Patients may have entered into more than one clinical trial.
†7 days of treatment
‡1 day of treatment
§daily treatment
Table 6 lists selected laboratory abnormalities in genital herpes suppression trials.
| Parameter | Famvir®
(n = 660)† % | Placebo
(n = 210)† % |
| Anemia (<0.8 x NRL) | 0.1 | 0.0 |
| Leukopenia (<0.75 x NRL) | 1.3 | 0.9 |
| Neutropenia (<0.8 x NRL) | 3.2 | 1.5 |
| AST (SGOT) (>2 x NRH) | 2.3 | 1.2 |
| ALT (SGPT) (>2 x NRH) | 3.2 | 1.5 |
| Total Bilirubin (>1.5 x NRH) | 1.9 | 1.2 |
| Serum Creatinine (>1.5 x NRH) | 0.2 | 0.3 |
| Amylase (>1.5 x NRH) | 1.5 | 1.9 |
| Lipase (>1.5 x NRH) | 4.9 | 4.7 |
*Percentage of patients with laboratory abnormalities that were increased or decreased from baseline and were outside of specified ranges.
†n values represent the minimum number of patients assessed for each laboratory parameter.
NRH = Normal Range High.
NRL = Normal Range Low.
HIV-Infected Patients
In HIV-infected patients, the most frequently reported adverse events for famciclovir (500 mg twice daily; n=150) and acyclovir (400 mg, 5x/day; n=143), respectively, were headache (16.7% vs. 15.4%), nausea (10.7% vs. 12.6%), diarrhea (6.7% vs. 10.5%), vomiting (4.7% vs. 3.5%), fatigue (4.0% vs. 2.1%), and abdominal pain (3.3% vs. 5.6%).
Post Marketing Experience
The following adverse events have been reported during post-approval use of Famvir: urticaria, serious skin reactions (e.g. erythema multiforme,Stevens-Johnson syndrome, toxic epidermal necrolysis), cholestatic jaundice, abnormal liver function tests, thrombocytopenia, hallucinations, dizziness, somnolence and confusion (including delirium, disorientation, confusional state, occurring predominantly in the elderly). Because these adverse events are reported voluntarily from a population of unknown size, estimates of frequency cannot be made.
OVERDOSAGE
Appropriate symptomatic and supportive therapy should be given. Penciclovir is removed by hemodialysis (see PRECAUTIONS, General).
DOSAGE AND ADMINISTRATION
Herpes Zoster
The recommended dosage is 500 mg every 8 hours for 7 days. Therapy should be initiated promptly as soon as herpes zoster is diagnosed. No data are available on efficacy of treatment started greater than 72 hours after rash onset.
Herpes Simplex Infections
Recurrent Genital Herpes
The recommended dosage is 1000 mg twice daily for 1 day. Initiate therapy at the first sign or symptom if medical management of a genital herpes recurrence is indicated. The efficacy of Famvir® (famciclovir) has not been established when treatment is initiated more than 6 hours after onset of symptoms or lesions.
Suppression of Recurrent Genital Herpes
The recommended dosage is 250 mg twice daily for up to 1 year. The safety and efficacy of Famvir therapy beyond 1 year of treatment have not been established.
Recurrent Herpes Labialis (Cold Sores)
The recommended dosage is 1500 mg as a single dose. Initiate therapy at the earliest sign or symptom of a cold sore (e.g. tingling, itching or burning).
HIV-Infected Patients
For recurrent orolabial or genital herpes simplex infection, the recommended dosage is 500 mg twice daily for 7 days.
Patients with Reduced Renal Function
In patients with reduced renal function, dosage reduction is recommended (see PRECAUTIONS, General).
| Indication and
Normal Dosage Regimen | Creatinine
Clearance (mL/min.) | Adjusted
Dosage Regimen Dose (mg) | Dosing Interval |
| Single-Day Dosing Regimens | |||
| Recurrent Genital Herpes 1000 mg every 12 hours for 1 day | ≥60 | 1000 | every 12 hours for 1 day |
| 40-59 | 500 | every 12 hours for 1 day | |
| 20-39 | 500 | single dose | |
| <20 | 250 | single dose | |
| HD* | 250 | single dose following dialysis |
|
| Recurrent Herpes Labialis 1500 mg single dose | ≥60 | 1500 | single dose |
| 40-59 | 750 | single dose | |
| 20-39 | 500 | single dose | |
| <20 | 250 | single dose | |
| HD* | 250 | single dose following dialysis |
|
| Multiple-Day Dosing Regimens | |||
| Herpes Zoster 500 mg every 8 hours | ≥60 | 500 | every 8 hours |
| 40-59 | 500 | every 12 hours | |
| 20-39 | 500 | every 24 hours | |
| <20 | 250 | every 24 hours | |
| HD* | 250 | following each dialysis | |
| Suppression of Recurrent Genital Herpes 250 mg every 12 hours | ≥40 | 250 | every 12 hours |
| 20-39 | 125 | every 12 hours | |
| <20 | 125 | every 24 hours | |
| HD* | 125 | following each dialysis | |
| Recurrent Orolabial and Genital Herpes Simplex Infection in HIV-Infected Patients 500 mg every 12 hours | ≥40 | 500 | every 12 hours |
| 20-39 | 500 | every 24 hours | |
| <20 | 250 | every 24 hours | |
| HD* | 250 | following each dialysis | |
*Hemodialysis
Administration with Food
When famciclovir was administered with food, penciclovir Cmax decreased approximately 50%. Because the systemic availability of penciclovir (AUC) was not altered, it appears that Famvir may be taken without regard to meals.
HOW SUPPLIED
Famvir® (famciclovir) is supplied as film-coated tablets as follows: 125 mg in bottles of 30; 250 mg in bottles of 30; and 500 mg in bottles of 30 and Single Unit Packages of 50 (intended for institutional use only).
Famvir 125 mg tablet:
White, round film-coated, biconvex, beveled edges, debossed with “FAMVIR” on one side and “125” on the other.
125 mg 30’s…………………………………………………………...……NDC 0078-0366-15
Famvir 250 mg tablet:
White, round film-coated, biconvex, beveled edges, debossed with “FAMVIR” on one side and “250” on the other.
250 mg 30’s………………………………………………………………...NDC 0078-0367-15
Famvir 500 mg tablet:
White, oval film-coated, biconvex, debossed with “FAMVIR” on one side and “500” on the other.
500 mg 30’s…………………………………………………………….......NDC 0078-0368-15
500 mg SUP 50’s…………………………………………………………...NDC 0078-0368-64
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
REV: DECEMBER 2008 T2009-99
Distributed by:
Novartis Pharmaceuticals Corp.
East Hanover, NJ 07936
©Novartis
PRINCIPAL DISPLAY PANEL
Package Label – 125 mg
Rx Only NDC 0078-0366-15
FAMVIR® (famciclovir) Tablets
125 mg per tablet
30 Tablets

PRINCIPAL DISPLAY PANEL
Package Label – 250 mg
Rx Only NDC 0078-0367-15
FAMVIR® (famciclovir) Tablets
250 mg per tablet
30 Tablets

PRINCIPAL DISPLAY PANEL
Package Label – 500 mg
Rx Only NDC 0078-0368-15
FAMVIR® (famciclovir) Tablets
500 mg per tablet
30 Tablets

| FAMVIR
famciclovir tablet, film coated |
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA020363 | 06/29/1994 | |
| FAMVIR
famciclovir tablet, film coated |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA020363 | 06/29/1994 | |
| FAMVIR
famciclovir tablet, film coated |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA020363 | 06/29/1994 | |
| Labeler - Novartis Pharmaceuticals Corporation (002147023) |