LINDANE
-
lindane lotion
Morton Grove Pharmaceuticals, Inc.
----------
LINDANE LOTION, USP 1%Rx only
WARNINGS
Lindane Lotion should only be used in patients who cannot tolerate or have failed first-line treatment with safer medications for the treatment of scabies. (See INDICATIONS AND USAGE.)
Neurologic Toxicity
Seizures and deaths have been reported following Lindane Lotion use with repeat or prolonged application, but also in rare cases following a single application used according to directions. Lindane Lotion should be used with caution for infants, children, the elderly, and individuals with other skin conditions (e.g., atopic dermatitis, psoriasis) and in those who weigh < 110 lbs (50 kg) as they may be at risk of serious neurotoxicity.
Contraindications
Lindane Lotion is contraindicated in premature infants and individuals with known uncontrolled seizure disorders.
Proper Use
Instruct patients on the proper use of Lindane Lotion, the amount to apply, how long to leave it on, and avoiding retreatment. Inform patients that itching occurs after the successful killing of scabies and is not necessarily an indication for retreatment with Lindane Lotion. (See DOSAGE AND ADMINISTRATION.)
DESCRIPTION
Lindane Lotion, USP 1%, is an ectoparasiticide and ovicide. In addition to the active ingredient, lindane, it contains 2-amino-2-methyl-1-propanol, carrageenan, cetyl alcohol, cocoa butter, glyceryl monostearate, methylparaben, propylene glycol, propylparaben, purified water, stearic acid and trolamine to form a lotion. The pH range is between 6.5 and 8.5. Lindane is the gamma isomer of 1,2,3,4,5,6-hexachlorocyclohexane having the following structural formula:
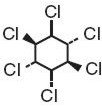
C6H6Cl6 M.W. 290.83
CLINICAL PHARMACOLOGY
Lindane Lotion, USP 1%, is an ectoparasiticide and ovicide effective against Sarcoptes scabiei (scabies). Lindane exerts its parasiticidal action by being directly absorbed into the parasites and their ova. Feldmann and Maibach1 reported approximately 10% systemic absorption of a lindane acetone solution when applied to the forearm of human subjects and left in place for 24 hours. This vehicle was different from the approved product and the percutaneous penetration of lindane is dependent on the vehicle. Therefore, the clinical significance of these observations is unknown. Dale, et al2 reported a blood level of 290 ng/mL associated with convulsions following the accidental ingestion of a lindane-containing product. Ginsburg 3 found the greatest peak blood level of 64 ng/mL, 6 hours after total body application of Lindane Lotion in 1 of 8 non-scabietic pediatric patients. The half-life in blood was determined to be approximately 18 hours.
Data available in the literature suggest that lindane has a rapid distribution phase followed by a longer β-elimination phase.1, 2, 3
INDICATIONS AND USAGE
Lindane Lotion is indicated for the treatment of scabies (infestations of Sarcoptes scabei ) only in patients who:
- cannot tolerate other approved therapies, or
- have failed treatment with other approved therapies.
Lindane Lotion should be used in the context of an overall scabies management program that includes:
- Microscopic evaluation of skin scrapings to confirm the diagnosis.
- Evaluation and treatment of sexual contacts simultaneously. Sexual contacts should be prescribed Lindane Lotion only if they either have failed to respond to adequate doses of other approved therapies or are intolerant of other approved therapies.
- Washing of all recently worn clothing, underwear, pajamas, used sheets, pillowcases, and towels in very hot water or dry-cleaned.
Caregivers applying this product to patients should wear gloves less permeable to Lindane, such as nitrile, latex with neoprene, or sheer vinyl, and thoroughly clean hands after application. Natural latex gloves should be avoided because they are more permeable to Lindane.
Lindane Lotion does not prevent infestation or reinfestation and should not be used to ward off a possible infestation.
CONTRAINDICATIONS
Lindane Lotion is contraindicated for premature infants because their skin may be more permeable than that of full term infants and their liver enzymes may not be sufficiently developed to metabolize Lindane.
Lindane Lotion is also contraindicated for patients with crusted (Norwegian) scabies and other skin conditions (e.g., atopic dermatitis, psoriasis) that may increase systemic absorption of the drug.
Lindane Lotion is contraindicated for patients with known uncontrolled seizure disorders and for individuals with a known sensitivity to the product or any of its components.
WARNINGS
(See boxed WARNINGS.)
Seizures and deaths have been reported following Lindane Lotion's use with repeat or prolonged application, but also in rare cases following a single application reportedly used according to directions. It is not known how soon after application of a single dose of Lindane Lotion that a second dose of Lindane Lotion can be safely applied.
There have been cases of adverse events reported for Lindane Lotion and Lindane Shampoo in which a serious outcome (hospitalization, disability or death) has occurred.4 In approximately 20% of the total reported cases, Lindane Lotion and Shampoo were reported to have been used according to the labeled directions. Of these cases, thirteen deaths were reported, many cases which were remote from the time of actual Lindane use. Lindane toxicity, verified by autopsy was the cause of one infant's death, was the cause of death reported for an adult who ingested it orally in a successful suicide. The direct causes of death for the other cases were attributed to reasons other than Lindane. Most of these adverse events occurred with Lindane Lotion.
Infants, children, the elderly, and individuals with other skin conditions and those who weigh < 110 lbs (50 kg) may be at greater risk of serious neurotoxicity. (See Pediatric Use and Geriatric Use.) Animal studies have shown increased susceptibility to neurologic adverse events in younger animals. Children have a larger body surface area to volume ratio that may result in a proportionately larger systemic exposure.
Careful consideration should be given before prescribing Lindane Lotion to patients with conditions that may increase the risk of seizure, such as HIV infection, history of head trauma or a prior seizure, CNS tumor, the presence of severe hepatic cirrhosis, excessive use of alcohol, abrupt withdrawal from alcohol or sedatives, as well as concomitant use of medications known to lower seizure threshold. (See PRECAUTIONS: Drug Interactions.)
Patients should be instructed on proper use of Lindane Lotion, especially the amount to apply, how long to leave the lotion on, and the need to avoid retreatment. Patients should be informed that itching may occur, and even worsen, after the successful killing of scabies. Repeat treatment is usually not necessary.
A Lindane Lotion Medication Guide must be given to the patient each time Lindane Lotion is dispensed, as required by law.
PRECAUTIONS
General
Care should be taken to avoid contact with the eyes. If such contact occurs, eyes should be immediately flushed with water. If irritation or sensitization occurs, the patient should be advised to consult a physician.
Information for Patients (and Caregivers)
- This product can be poisonous if misused.
- Other important information is found in the Medication Guide, which by law, must be dispensed with Lindane Lotion.
- If someone other than the patient will be applying Lindane Lotion to the patient, they should wear less permeable gloves such as nitrile, latex with neoprene, or sheer vinyl, and thoroughly clean their hands after application. Natural latex gloves should be avoided because they are more permeable to lindane.
- If the person applying Lindane Lotion could be pregnant, contact with Lindane Lotion should be avoided as much as possible.
- If the patient could be pregnant, other treatments may be preferable.
- Lindane Lotion should be used for scabies only.
- The skin should be clean and without any other lotion, cream, or oil on it. Oils can make the Lindane Lotion go through the skin faster and possibly increase the risk of neurotoxicity (e.g., seizures).
- Wait at least 1 hour after bathing or showering before putting Lindane Lotion on the skin. Wet or warm skin can make the Lindane Lotion go through skin faster.
- Information for Use:
- Shake Lindane Lotion well.
- Put Lindane Lotion under fingernails after trimming the fingernails short, because scabies are very likely to remain there. A toothbrush can be used to apply the Lindane Lotion under the fingernails. Immediately after use, the toothbrush should be wrapped in paper and thrown away. Use of the same brush in the mouth could lead to poisoning.
- Use only a single application, applied as a very thin layer over all skin from the neck down.
- Close the bottle with the leftover Lindane Lotion and immediately throw it away in a trash can out of the reach of children.
- Do not use any covering over the applied Lindane Lotion that does not breathe, like diapers with plastic lining, plastic clothes, tight clothes, or blankets.
- Wash the Lindane Lotion completely off after 8 to 12 hours. Never leave Lindane Lotion on the skin for more than 12 hours. Warm, but not hot water can be used. Lindane Lotion will not kill any more scabies after 8 to 12 hours, but may cause serious health problems.
- Keep Lindane Lotion away from mouth and eyes. If contact with eyes occurs, immediately flush eyes with water. Do not use if you have open wounds, cuts or sores that are present, unless specifically directed by the prescribing physician.
- All recently worn clothing, underwear, pajamas, used sheets, pillowcases, and towels should be washed in very hot water or dry-cleaned.
- The patient may still itch after using Lindane Lotion. This does not mean the medicine did not work. Even after all the scabies (mites) are dead, they can still make the skin itch for a few weeks. Lindane Lotion sometimes makes this itch even worse. Other treatments can be used to soothe the itch. Do not use more Lindane Lotion.
- The patient should contact the physician with any questions or concerns about his or her condition or use of the Lindane Lotion.
Drug Interactions
Oils may enhance absorption of Lindane, therefore, patients or caregivers applying Lindane Lotion should be warned about simultaneous use of creams, ointments, or oils. In addition, there are many drugs that may lower the seizure threshold, and Lindane Lotion should be prescribed with caution in patients taking these medications. Drugs that may lower the seizure threshold include, but are not limited to the following:
- Antipsychotics
- Antidepressants
- Theophylline
- Cyclosporine, mycophenolate mofetil, tacrolimus capsules
- Penicillins, imipenem, quinolone antibiotics
- Chloroquine sulfate, pyrimethamine
- Isoniazid
- Meperidine
- Radiographic contrast agents
- Centrally active anticholinesterases
- Methocarbamol
Carcinogenesis, Mutagenesis, and Fertility
Although no studies have been conducted with Lindane Lotion, numerous long-term feeding studies have been conducted in mice and rats to evaluate the carcinogenic potential of the technical grade of hexachlorocyclohexane as well as the alpha, beta, gamma (lindane) and delta isomers. Both oral and topical applications have been evaluated. Increased incidences of neoplasms were not clearly related to administration of lindane. The results of mutagenicity tests in bacteria do not indicate that lindane is mutagenic. Lindane did not cause sister chromatid exchange in an in vivo assay. The number of spermatids in the testes of rats 2 weeks after oral administration of a single dose of 30 mg/kg body weight (12 times the estimated human exposure for scabies on a body surface area comparison and assuming 50% rat oral bioavailability and 10% human bioavailability) was significantly reduced compared to the control rats.
Pregnancy
Pregnancy Category C
All pregnancies have a risk of birth defect, loss, or other adverse event regardless of drug exposure. Predictions of fetal risk from drug exposure rely heavily on animal data. However, animal studies may fail to predict effects in humans or may overstate such risks. Even if human data are available, the data may not be sufficient to determine whether there is an increased risk to the fetus, and individual reports of adverse outcomes in pregnancy in association with a drug may not reflect a causal relationship.
Lindane Lotion should be given to pregnant women only if clearly needed. There are no adequate and well-controlled studies of Lindane Lotion in pregnant women. There are no known maternal or fetal health risks if the scabies is not treated. Lindane is lipophilic and may accumulate in the placenta. There has been a single case report of a stillborn infant following multiple maternal exposures to lindane during pregnancy. The relationship of the maternal exposures to the fetal outcome is unknown.
Animal data suggest that lindane exposure of the fetus may increase the likelihood of neurologic developmental abnormalities (see below), based on findings at systemic exposures close to that expected in humans when Lindane Lotion is used to treat scabies. The immature central nervous system (as in the fetus) may have increased susceptibility to the effects of the drug.
Data
When rats received lindane in the diet from day 6 of gestation through day 10 of lactation, reduced pup survival, decreased pup weight and decreased weight gains during lactation, increased motor activity and decreased motor activity habituation were seen in pups at 5.6 mg/kg (2 times the estimated human exposure) but not at 1.2 mg/kg. An increased number of stillborn pups was seen at 8 mg/kg, and increased pup mortality was seen at 5.6 mg/kg. No gross abnormalities were seen in this study or in a study in which rabbits received up to 20 mg/kg lindane by gavage on gestation day 6–18 (up to 10 times the human exposure on a body surface area comparison and assuming 50% rabbit oral bioavailability and 10% human bioavailability).
Nursing Mothers
Lindane is lipophilic and is present in human breast milk, but exact quantities are not known. There may be a risk of toxicity if lindane is ingested from breast milk, or from skin absorption from mother to baby in the course of breast-feeding when Lindane Lotion is applied topically to the chest area. Nursing mothers who require treatment with Lindane Lotion should be advised of the potential risks and be counseled to avoid large areas of skin-to-skin contact with the infant while Lindane Lotion is applied, as well as to interrupt breast-feeding, with expression and discarding of milk, for at least 24 hours following use.
Pediatric Use
Animal data demonstrated increased risk of adverse events in the young across species. Pediatric patients have a higher surface to volume ratio and may be at risk of greater systemic exposure when Lindane Lotion is applied to the body. Infants and children may be at an even higher risk due to immaturity of organ systems such as skin and liver. Lindane Lotion should be used with extreme caution in patients who weigh less than approximately 110 lbs (50 kg) and especially in infants. Lindane Lotion is indicated only for the treatment of scabies; patients with lice should use Lindane Shampoo according to the labeled instructions.
Geriatric Use
There have been no studies of Lindane Lotion in the elderly. There are four postmarketing reports of deaths in elderly patients who were treated for scabies with Lindane Lotion. Two patients died within 24 hours of Lindane Lotion application, and the third patient died 41 days after application of Lindane Lotion, having suffered a seizure on the day of death. A fourth patient died of an unreported cause of death on the same day that Lindane Lotion treatment for scabies was administered.
ADVERSE REACTIONS
Lindane Lotion has been reported to cause central nervous system stimulation ranging from dizziness to seizures. Although seizures were almost always associated with ingestion or misuse of the product (to include repeat treatment), seizures and deaths have been reported when Lindane Lotion was used according to directions. Irritant dermatitis from contact with this product has also been reported. (See WARNINGS, PRECAUTIONS, and DOSAGE AND ADMINISTRATION.)
Postmarketing Experience
The following adverse reactions reflect additional postmarketing experience of Lindane Lotion. These events include alopecia, dermatitis, headache, pain, paresthesia, pruritus and urticaria. The relationship of some of these events to Lindane therapy is unknown.
OVERDOSAGE
Contact the closest Poison Control Center in the event of suspected overdosage with Lindane Lotion.
If accidental ingestion occurs, prompt gastric lavage should be instituted. However, since oils enhance absorption, saline cathartics for intestinal evacuation should be given rather than oil laxatives. If central nervous system (CNS) manifestations occur, they may be antagonized by the administration of pentobarbital, phenobarbital, or diazepam.
DOSAGE AND ADMINISTRATION
Apply a thin layer of Lindane Lotion over all skin from the neck down. One ounce is sufficient for an average adult. Do not prescribe more than 2 ounces for larger adults. Apply only once. Wash off in 8 to 12 hours. Do not retreat. (See boxed WARNINGS.)
Patients should be provided specific information on use of product. (See PRECAUTIONS: Information for Patients and the Medication Guide.) Patients should be instructed on proper use of Lindane Lotion, especially the amount to apply, how long to leave on and the need to avoid retreatment. Patients should be informed that itching occurs after the successful killing of scabies (mites) and continued itching is not necessarily an indication for retreatment with Lindane Lotion.
A Lindane Lotion Medication Guide must be given to the patient each time Lindane Lotion is dispensed, as required by law. The Lindane Lotion Medication Guide is an important part of the risk management program for the patient.
HOW SUPPLIED
Lindane Lotion, USP 1% is supplied in patient-size 2 fl oz (60 mL) bottles.
SHAKE WELL BEFORE USING
Store at controlled room temperature, 15 ° – 30 °C (59 ° – 86 °F) [see USP].
REFERENCES
- Feldmann, R.J. and Maibach, H.I., Toxicol. Applied. Pharmacol., 28:126, 1974.
- Dale, W.E., Curly, A. and Cueto, C. Life Sci 5:47, 1966.
- Ginsburg, C.M., et al., J. Pediatr. 91:6, 998–1000, 1977.
- FDA AERS database search, January 2003
Rx Only
Product No.: 8833
Manufactured By: Morton Grove Pharmaceuticals, Inc.
Morton Grove, IL 60053
A50-8833-60
REV. 01-09
PHARMACIST—PATIENT MEDICATION GUIDE PROVIDED BELOW
MEDICATION GUIDE
Lindane (LIHN-dane) Lotion, USP 1%
You must read and follow all instructions before using Lindane Lotion. Read the information you get every time you or a family member get Lindane Lotion. There may be new information. This Medication Guide does not take the place of talking with your doctor about your medical condition or treatment. If you have any questions about Lindane Lotion, ask your doctor or pharmacist.
What is the most important information I should know about Lindane Lotion?
Lindane Lotion is a poison if you do not use it the right way. Lindane Lotion goes through your skin and may affect your brain and nerves. Lindane Lotion can cause seizures, also called convulsions, "fits" or epilepsy.
- Seizures and death can happen in people who use Lindane Lotion too much or too often.
- Seizures can happen in some people even if they use Lindane Lotion exactly as directed.
If you or a family member has a seizure while using Lindane Lotion, get emergency help right away.
- Do not use Lindane Lotion unless:
- You have scabies and were treated with another medicine that did not work for you, or
- You cannot use other safer medicines to treat your scabies.
- Do not use Lindane Lotion more than 1 time to treat an attack of scabies. Do not use Lindane Lotion to treat a second attack of scabies that comes soon after the first episode. Using it more than 1 time can cause seizures and death. No one knows a safe time to reuse Lindane Lotion. Even if you still itch after using Lindane Lotion, do not use more or use it again. Scabies (mites) can make your skin itch for a few weeks even after all of the bugs (mites) are dead.
- Do not use more Lindane Lotion than your doctor tells you.
- Do not keep Lindane Lotion on your skin for more than 8 to 12 hours.
- Do not put Lindane Lotion in your mouth because it is a poison if taken by mouth. If you get Lindane Lotion in your mouth or swallow Lindane Lotion, call your area Poison Control Center right away and get emergency help.
What is Lindane Lotion?
Lindane Lotion is a medicine that is used to treat scabies. It kills scabies and their eggs. Scabies are very small bugs (mites) that crawl under your skin, lay eggs, and cause severe itching. Lindane Lotion goes through your skin and kills the scabies and their eggs. Lindane Lotion is used only after safer medicines have not made your scabies go away. The only time Lindane Lotion is used first is when someone cannot use safer medicines, which may include permethrin and crotamiton.
Lindane Lotion is mainly for adults and children who weigh at least 110 pounds. If you weigh less than 110 pounds, use Lindane Lotion only if your doctor thinks it is really needed. People who weigh less than 110 pounds and the elderly have higher chances for side effects because more lindane may go through their skin.
Who should not use Lindane Lotion?
Do not use Lindane Lotion:
- if you do not have scabies. Lindane Lotion does not stop you from getting scabies. Lindane Lotion only kills the scabies you already have.
- if you have or have ever had seizures, also called convulsions, "fits" or epilepsy, especially if they have been hard to control.
- if you used Lindane Lotion in the past few months. You should see your doctor if you think you need another treatment.
- unless it is the only medicine you can use for scabies.
- if you had a bad reaction to Lindane Lotion before. Do not use Lindane Lotion again.
- if you have open sores or crusted (scabby) sores on your skin, or lots of broken skin.
- if you have head or body lice. These need a different medicine that you use in a different way.
- if you are allergic to Lindane Lotion or any of its ingredients. The active ingredient is lindane. See the end of this Medication Guide for a list of all the ingredients in Lindane Lotion.
- if you need to treat a premature or young baby. More lindane can go through the skin of babies and go to their brains where it can harm them.
- while you are breast-feeding. Lindane Lotion can get in your milk and be fed to your baby. Lindane Lotion on your skin can also go to your baby. Your baby may get sick. Ask your doctor for a safer medicine. If you use Lindane Lotion, pump your breast milk and throw away the milk for at least 24 hours after using the medicine. During this time, feed your baby formula or breast milk you stored from before you used Lindane Lotion.
Tell your doctor if you:
- used Lindane Lotion in the past few months.
- ever had a seizure or problem that could increase your chances of getting a seizure (like a head injury, tumor in your brain or spinal cord, cirrhosis of the liver, or heavy alcohol drinking.)
- have HIV or AIDS. Lindane Lotion may cause seizures even if you never had them before.
- are pregnant. Lindane Lotion can reach your baby and may harm it. Ask your doctor for a safer medicine. Use Lindane Lotion only if needed.
- have a sexual partner. Your partner should get checked and treated for scabies so they don't give them back to you. Don't share your Lindane Lotion with your partner.
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Some medicines may increase your chances of having a seizure if you take them while using Lindane Lotion. Especially, tell your doctor if you take medicines called sedatives (drugs to help you sleep).
How do I use Lindane Lotion?
Before you put it on:
- Make sure you know how to use it exactly as your doctor prescribes.
- If you are putting Lindane Lotion on another person, wear special gloves made of nitrile, latex with neoprene, or sheer vinyl. Do not use natural latex gloves because more lindane can go through that kind of glove. Wash your hands well when you are done.
- Make sure your skin is clean and does not have any other lotion, cream, or oil on it. Oils can make Lindane Lotion go through your skin faster and may increase the risk of seizures.
- Wait for at least 1 hour after bathing or showering before you put Lindane Lotion on your skin. Wet or warm skin can make the Lindane Lotion go through your skin faster and may increase the risk of seizures.
When you put it on:
- Shake the bottle of Lindane Lotion well.
- Put Lindane Lotion under your fingernails, because the scabies really like to stay there. Trim your fingernails short. Use a toothbrush to get the Lindane Lotion under your fingernails. Wrap this toothbrush in paper and throw this toothbrush away. Do not use it in your mouth.
- Put a very thin layer of the Lindane Lotion on your skin from the neck down. You may have some Lindane Lotion left in the bottle.
- Do not cover over the Lindane Lotion on your skin with anything that does not breathe, like diapers with a plastic lining, plastic clothes, tight clothes, or blankets.
- Close the bottle with the leftover Lindane Lotion and throw it away in a trash can out of the reach of children.
When you are supposed to wash it off:
- Wash the Lindane Lotion off your skin after 8 to 12 hours. You must wash the Lindane Lotion off your body completely. Use warm, but not hot water. Lindane Lotion will not kill any more scabies after 8 to 12 hours. After 8 to 12 hours, Lindane Lotion can cause serious health problems, such as seizures and death.
After you wash off the Lindane Lotion:
- All recently worn clothing, underwear, pajamas, used sheets, pillowcases, and towels should be washed in very hot water or dry-cleaned.
- Do not use Lindane Lotion again. If you think you need to use it again, you must check with your doctor to find out when it is most safe.
You may still itch after you use Lindane Lotion. This does not mean you need more Lindane Lotion. Even after all the scabie bugs (mites) are dead, they can still make your skin itch for a few weeks. Lindane Lotion sometimes makes this itch even worse. Talk to your doctor about things you can do to soothe the itch.
What should I avoid while using Lindane Lotion?
- Do not get Lindane Lotion in your eyes. If you do, rinse your eyes with water right away. Get medical help if your eyes keep hurting.
- Do not let your skin touch other people's skin while you have Lindane Lotion on. Make sure your skin does not touch your baby or small child.
- If you are pregnant, do not use Lindane Lotion, or apply Lindane Lotion to others unless it is needed and you have talked to your doctor about using it. See the special glove advice below if you have to put Lindane Lotion on others.
- Do not use oils on your skin or hair just before or after using Lindane Lotion. Oils include skin lotions and moisturizers, and oil-based hair products and conditioners.
- Do not get Lindane Lotion on your hands if you are putting it on someone else.Wear special gloves made of nitrile, latex with neoprene, or sheer vinyl. Do not use natural latex gloves. Wash your hands well when you are done.
What are the possible side effects of Lindane Lotion?
Lindane Lotion may cause serious side effects such as seizures (convulsions, fits) or death (See the section, "What is the most important information I should know about Lindane Lotion?"). Lindane Lotion can also make you feel sleepy, dizzy, or can cause body shaking that you cannot control.
The most common side effects of Lindane Lotion are:
- Itching skin
- Burning skin
- Dry skin
- A skin rash
These are not all of the possible side effects of Lindane Lotion. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General Information about Lindane Lotion:
Medicines are sometimes prescribed for purposes other than those listed in Medication Guides. Do not use Lindane Lotion for any condition for which it was not prescribed. Do not give Lindane Lotion to other people, even if they have the same symptoms that you have. It may harm them. Keep Lindane Lotion and all medicines out of the reach of children.
This Medication Guide summarizes the most important information about Lindane Lotion. If you want more information, talk with your doctor. You can ask your doctor or pharmacist for information about Lindane Lotion that is written for health professionals.
What are the ingredients in Lindane Lotion?
Active Ingredient: Lindane.
Inactive Ingredients: 2-amino-2-methyl-1-propanol, carrageenan, cetyl alcohol, cocoa butter, glyceryl monostearate, methylparaben, propylene glycol, propylparaben, purified water, stearic acid and trolamine.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
This medication is available only by a prescription from your doctor.
Product No.: 8833
Manufactured By:
Morton Grove Pharmaceuticals, Inc.
Morton Grove, IL 60053
A50-8833-60
REV. 01-09
PRINCIPAL DISPLAY PANEL - 60 mL Bottle
MGP
NDC 60432-833-60
LINDANE
LOTION,
USP 1%
POISON: FOR EXTERNAL USE ONLY
HARMFUL OR FATAL IF SWALLOWED
WARNING: KEEP OUT OF
REACH OF CHILDREN
Rx Only
NET: 2 fl oz (60 mL)

PRINCIPAL DISPLAY PANEL - 60 mL Bottle Carton
MGP
NDC 60432-833-60
LINDANE
LOTION,
USP 1%
POISON: FOR EXTERNAL USE ONLY
HARMFUL OR FATAL IF SWALLOWED
WARNING: KEEP OUT OF
REACH OF CHILDREN
Rx Only
NET: 2 fl oz (60 mL)

| LINDANE
lindane lotion |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA088190 | 08/16/1984 | |
| Labeler - Morton Grove Pharmaceuticals, Inc. (801897505) |