TRACLEER
-
bosentan tablet, film coated
Actelion Pharmaceuticals US, Inc.
----------
TRACLEER®
[bosentan]
62.5 mg and 125 mg film-coated tablets
Use of TRACLEER® requires attention to two significant concerns: 1) potential for serious liver injury, and 2) potential damage to a fetus.
WARNING: Potential liver injury
TRACLEER® causes at least 3-fold (upper limit of normal; ULN) elevation of liver aminotransferases (ALT and AST) in about 11% of patients, accompanied by elevated bilirubin in a small number of cases. Because these changes are a marker for potential serious liver injury, serum aminotransferase levels must be measured prior to initiation of treatment and then monthly (see WARNINGS: Potential Liver Injury and DOSAGE AND ADMINISTRATION). In the post-marketing period, in the setting of close monitoring, rare cases of unexplained hepatic cirrhosis were reported after prolonged (> 12 months) therapy with TRACLEER® in patients with multiple co-morbidities and drug therapies. There have also been rare reports of liver failure. The contribution of TRACLEER® in these cases could not be excluded.
In at least one case the initial presentation (after > 20 months of treatment) included pronounced elevations in aminotransferases and bilirubin levels accompanied by non-specific symptoms, all of which resolved slowly over time after discontinuation of TRACLEER®. This case reinforces the importance of strict adherence to the monthly monitoring schedule for the duration of treatment and the treatment algorithm, which includes stopping TRACLEER® with a rise of aminotransferases accompanied by signs or symptoms of liver dysfunction. (see DOSAGE AND ADMINISTRATION).
Elevations in aminotransferases require close attention (see DOSAGE AND ADMINISTRATION). TRACLEER® should generally be avoided in patients with elevated aminotransferases (> 3 × ULN) at baseline because monitoring liver injury may be more difficult. If liver aminotransferase elevations are accompanied by clinical symptoms of liver injury (such as nausea, vomiting, fever, abdominal pain, jaundice, or unusual lethargy or fatigue) or increases in bilirubin ≥ 2 × ULN, treatment should be stopped. There is no experience with the re-introduction of TRACLEER® in these circumstances.
CONTRAINDICATION: Pregnancy
TRACLEER® (bosentan) is very likely to produce major birth defects if used by pregnant women, as this effect has been seen consistently when it is administered to animals (see CONTRAINDICATIONS). Therefore, pregnancy must be excluded before the start of treatment with TRACLEER® and prevented thereafter by the use of a reliable method of contraception. Hormonal contraceptives, including oral, injectable, transdermal, and implantable contraceptives should not be used as the sole means of contraception because these may not be effective in patients receiving TRACLEER® (see Precautions: Drug Interactions). Therefore, effective contraception through additional forms of contraception must be practiced. Monthly pregnancy tests should be obtained.
Because of potential liver injury and in an effort to make the chance of fetal exposure to TRACLEER® (bosentan) as small as possible, TRACLEER® may be prescribed only through the TRACLEER® Access Program by calling 1 866 228 3546. Adverse events can also be reported directly via this number.
DESCRIPTION
Bosentan is the first of a new drug class, an endothelin receptor antagonist.
TRACLEER® (bosentan) belongs to a class of highly substituted pyrimidine derivatives, with no chiral centers. It is designated chemically as 4-tert-butyl-N-[6-(2-hydroxy-ethoxy)-5-(2-methoxy-phenoxy)-[2,2']-bipyrimidin-4-yl]- benzenesulfonamide monohydrate and has the following structural formula:
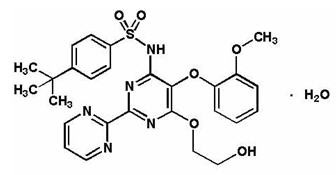
Bosentan has a molecular weight of 569.64 and a molecular formula of C27H29N5O6S•H2O. Bosentan is a white to yellowish powder. It is poorly soluble in water (1.0 mg/100 mL) and in aqueous solutions at low pH (0.1 mg/100 mL at pH 1.1 and 4.0; 0.2 mg/100 mL at pH 5.0). Solubility increases at higher pH values (43 mg/100 mL at pH 7.5). In the solid state, bosentan is very stable, is not hygroscopic and is not light sensitive.
TRACLEER® is available as 62.5 mg and 125 mg film-coated tablets for oral administration, and contains the following excipients: corn starch, pregelatinized starch, sodium starch glycolate, povidone, glyceryl behenate, magnesium stearate, hydroxypropylmethylcellulose, triacetin, talc, titanium dioxide, iron oxide yellow, iron oxide red, and ethylcellulose. Each TRACLEER® 62.5 mg tablet contains 64.541 mg of bosentan, equivalent to 62.5 mg of anhydrous bosentan. Each TRACLEER® 125 mg tablet contains 129.082 mg of bosentan, equivalent to 125 mg of anhydrous bosentan.
CLINICAL PHARMACOLOGY
Mechanism of Action
Endothelin-1 (ET-1) is a neurohormone, the effects of which are mediated by binding to ETA and ETB receptors in the endothelium and vascular smooth muscle. ET-1 concentrations are elevated in plasma and lung tissue of patients with pulmonary arterial hypertension, suggesting a pathogenic role for ET-1 in this disease. Bosentan is a specific and competitive antagonist at endothelin receptor types ETA and ETB. Bosentan has a slightly higher affinity for ETA receptors than for ETB receptors.
Pharmacokinetics
General
After oral administration, maximum plasma concentrations of bosentan are attained within 3–5 hours and the terminal elimination half-life (t1/2) is about 5 hours in healthy adult subjects. The exposure to bosentan after intravenous and oral administration is about 2-fold greater in adult patients with pulmonary arterial hypertension than in healthy adult subjects.
Absorption and Distribution
The absolute bioavailability of bosentan in normal volunteers is about 50% and is unaffected by food. The volume of distribution is about 18 L. Bosentan is highly bound (> 98%) to plasma proteins, mainly albumin. Bosentan does not penetrate into erythrocytes.
Metabolism and Elimination
Bosentan has three metabolites, one of which is pharmacologically active and may contribute 10%–20% of the effect of bosentan. Bosentan is an inducer of CYP2C9 and CYP3A4 and possibly also of CYP2C19. Total clearance after a single intravenous dose is about 4 L/hr in patients with pulmonary arterial hypertension. Upon multiple oral dosing, plasma concentrations in healthy adults decrease gradually to 50-65% of those seen after single dose administration, probably the effect of auto-induction of the metabolizing liver enzymes. Steady-state is reached within 3-5 days. Bosentan is eliminated by biliary excretion following metabolism in the liver. Less than 3% of an administered oral dose is recovered in urine.
Special Populations
It is not known whether bosentan's pharmacokinetics is influenced by gender, body weight, race, or age.
Liver Function Impairment
In vitro and in vivo evidence showing extensive hepatic metabolism of bosentan suggests that liver impairment could significantly increase exposure of bosentan. In a study comparing 8 patients with mild liver impairment (as indicated by the Child-Pugh method) to 8 controls, the single- and multiple-dose pharmacokinetics of bosentan were not altered in patients with mild hepatic impairment. The influence of moderate or severe liver impairment on the pharmacokinetics of bosentan has not been evaluated. Bosentan should generally be avoided in patients with moderate or severe liver abnormalities and/or elevated aminotransferases >3 × ULN (See DOSAGE AND ADMINISTRATION and WARNINGS).
Renal Impairment
In patients with severe renal impairment (creatinine clearance 15–30 mL/min), plasma concentrations of bosentan were essentially unchanged and plasma concentrations of the three metabolites were increased about 2-fold compared to people with normal renal function. These differences do not appear to be clinically important (See DOSAGE AND ADMINISTRATION).
Clinical Studies
Pulmonary Arterial Hypertension
Two randomized, double-blind, multi-center, placebo-controlled trials were conducted in 32 and 213 patients. The larger study (BREATHE-1) compared 2 doses (125 mg b.i.d. and 250 mg b.i.d.) of TRACLEER® with placebo. The smaller study (Study 351) compared 125 mg b.i.d. with placebo. Patients had severe (WHO functional Class III–IV) pulmonary arterial hypertension: primary pulmonary hypertension (72%) or pulmonary hypertension secondary to scleroderma or other connective tissue diseases (21%), or to autoimmune diseases (7%). There were no patients with pulmonary hypertension secondary to other conditions such as HIV disease, or recurrent pulmonary emboli.
In both studies, TRACLEER® or placebo was added to patients' current therapy, which could have included a combination of digoxin, anticoagulants, diuretics, and vasodilators (e.g., calcium channel blockers, ACE inhibitors), but not epoprostenol. TRACLEER® was given at a dose of 62.5 mg b.i.d. for 4 weeks and then at 125 mg b.i.d. or 250 mg b.i.d. for either 12 (BREATHE-1) or 8 (Study 351) additional weeks. The primary study endpoint was 6-minute walk distance. In addition, symptoms and functional status were assessed. Hemodynamic measurements were made at 12 weeks in Study 351.
The mean age was about 49 years. About 80% of patients were female, and about 80% were Caucasian. Patients had been diagnosed with pulmonary hypertension for a mean of 2.4 years.
Submaximal Exercise Capacity
Results of the 6-minute walk distance at 3 months (Study 351) or 4 months (BREATHE-1) are shown in Table 1.
| BREATHE-1 | Study 351 | ||||
|---|---|---|---|---|---|
| Bosentan 125 mg b.i.d. (n = 74) | Bosentan 250 mg b.i.d. (n = 70) | Placebo (n = 69) | Bosentan 125 mg b.i.d. (n = 21) | Placebo (n = 11) |
|
| Distance in meters: mean ± standard deviation. Changes are to week 16 for BREATHE-1 and to week 12 for Study 351. | |||||
| Baseline | 326 ± 73 | 333 ± 75 | 344 ± 76 | 360 ± 86 | 355 ± 82 |
| End point | 353 ± 115 | 379 ± 101 | 336 ± 129 | 431 ± 66 | 350 ± 147 |
| Change from baseline | 27 ± 75 | 46 ± 62 | -8 ± 96 | 70 ± 56 | -6 ± 121 |
| Placebo – subtracted | 35 * | 54 † | 76 ‡ |
|
|
In both trials, treatment with TRACLEER® resulted in a significant increase in exercise capacity. The improvement in walk distance was apparent after 1 month of treatment (with 62.5 mg b.i.d.) and fully developed by about 2 months of treatment (Figure 1). It was maintained for up to 7 months of double-blind treatment. Walking distance was somewhat greater with 250 mg b.i.d., but the potential for increased liver injury causes this dose not to be recommended (See DOSAGE AND ADMINISTRATION). There were no apparent differences in treatment effects on walk distance among subgroups analyzed by demographic factors, baseline disease severity, or disease etiology, but the studies had little power to detect such differences.
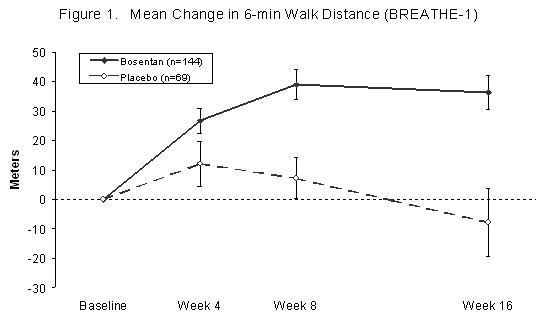
Change from baseline in 6-minute walking distance from start of therapy to week 16 in the placebo and combined bosentan (125 mg b.i.d. and 250 mg b.i.d.) groups. Values are expressed as mean ± standard error of the mean.
Hemodynamic Changes
Invasive hemodynamic parameters were assessed in Study 351. Treatment with TRACLEER® led to a significant increase in cardiac index (CI) associated with a significant reduction in pulmonary artery pressure (PAP), pulmonary vascular resistance (PVR), and mean right atrial pressure (RAP) (Table 2).
|
Bosentan 125 mg b.i.d | Placebo | ||
|---|---|---|---|
| Values shown are means ± SD | |||
| Mean CI (L/min/m2) | N=20 | N=10 | |
| Baseline | 2.35±0.73 | 2.48±1.03 | |
| Absolute Change | 0.50±0.46 | -0.52±0.48 | |
| Treatment Effect | 1.02* | ||
| Mean PAP (mmHg) | N=20 | N=10 | |
| Baseline | 53.7±13.4 | 55.7±10.5 | |
| Absolute Change | -1.6±5.1 | 5.1±8.8 | |
| Treatment Effect | -6.7† | ||
| Mean PVR (dyn•sec•cm-5) | N=19 | N=10 | |
| Baseline | 896±425 | 942±430 | |
| Absolute Change | -223±245 | 191±235 | |
| Treatment Effect | -415* | ||
| Mean RAP (mmHg) | N=19 | N=10 | |
| Baseline | 9.7±5.6 | 9.9±4.1 | |
| Absolute Change | -1.3±4.1 | 4.9±4.6 | |
| Treatment Effect | -6.2* | ||
Symptoms and Functional Status
Symptoms of pulmonary arterial hypertension were assessed by Borg dyspnea score, WHO functional class, and rate of "clinical worsening." Clinical worsening was assessed as the sum of death, hospitalizations for PAH, discontinuation of therapy because of PAH, and need for epoprostenol. There was a significant reduction in dyspnea during walk tests (Borg dyspnea score), and significant improvement in WHO functional class in TRACLEER®-treated patients. There was a significant reduction in the rate of clinical worsening (Table 3 and Figure 2). Figure 2 shows the Log-rank test reflecting clinical worsening over 28 weeks.
| BREATHE-1 | Study 351 | |||
|---|---|---|---|---|
| Bosentan 125/250 mg b.i.d. (N = 144) | Placebo (N = 69) | Bosentan 125 mg b.i.d. (N = 21) | Placebo (N = 11) |
|
| Note: Patients may have had more than one reason for clinical worsening. | ||||
| Patients with clinical worsening [n (%)] | 9 (6%)* | 14 (20%) | 0 (0%)† | 3 (27%) |
| Death | 1 (1%) | 2 (3%) | 0 (0%) | 0 (0%) |
| Hospitalization for PAH | 6 (4%) | 9 (13%) | 0 (0%) | 3 (27%) |
| Discontinuation due to worsening of PAH | 5 (3%) | 6 (9%) | 0 (0%) | 3 (27%) |
| Receipt of epoprostenol‡ | 4 (3%) | 3 (4%) | 0 (0%) | 3 (27%) |
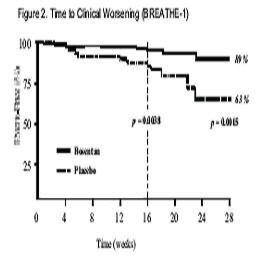
Time from randomization to clinical worsening with Kaplan-Meier estimate of the proportions of failures in BREATHE-1. All patients (N=144 in the bosentan group and N=69 in the placebo group) participated in the first 16 weeks of the study. A subset of this population (N=35 in the bosentan group and 13 in the placebo group) continued double-blind therapy for up to 28 weeks.
Pulmonary Arterial Hypertension related to Congenital Heart Defect
A small study with patients with Eisenmenger physiology demonstrated effects of bosentan on exercise and safety that were similar to those seen in other trials in patients with PAH (WHO Group I).
Congestive Heart Failure (CHF)
In a pair of studies, 1613 subjects with NYHA Class III-IV heart failure, left ventricular ejection fraction <35%, on diuretics, ACE inhibitor, and other therapies, were randomized to placebo or TRACLEER® (62.5 mg b.i.d. titrated as tolerated to 125 mg b.i.d.) and followed for up to 70 weeks. Use of TRACLEER® was associated with no benefit on patient global assessment (the primary end point) or mortality. However, hospitalizations for heart failure were more common during the first 4 to 8 weeks after bosentan was initiated. Based on these results, bosentan is not effective in the treatment of congestive heart failure with left ventricular dysfunction.
Long-term Treatment
The long-term follow-up of the patients who were treated with TRACLEER® in the two pivotal studies and their open-label extensions (N=235) shows that 93% and 84% of patients were still alive at 1 and 2 years, respectively, after the start of treatment with TRACLEER®. These uncontrolled observations do not allow comparison with a group not given TRACLEER® and cannot be used to determine the long-term effect of TRACLEER® on mortality.
INDICATIONS AND USAGE
TRACLEER® is indicated for the treatment of pulmonary arterial hypertension (WHO Group I) in patients with WHO Class III or IV symptoms, to improve exercise ability and decrease the rate of clinical worsening (see Clinical Studies).
CONTRAINDICATIONS
See BOX WARNING for CONTRAINDICATION to use in pregnancy.
Pregnancy Category X
TRACLEER® is expected to cause fetal harm if administered to pregnant women. Bosentan was teratogenic in rats given oral doses ≥ 60 mg/kg/day (twice the maximum recommended human oral dose of 125 mg, b.i.d., on a mg/ m2 basis). In an embryo-fetal toxicity study in rats, bosentan showed dose-dependent teratogenic effects, including malformations of the head, mouth, face and large blood vessels. Bosentan increased stillbirths and pup mortality at oral doses of 60 and 300 mg/kg/day (2 and 10 times, respectively, the maximum recommended human dose on a mg/m2 basis). Although birth defects were not observed in rabbits given oral doses of up to 1500 mg/kg/day, plasma concentrations of bosentan in rabbits were lower than those reached in the rat. The similarity of malformations induced by bosentan and those observed in endothelin-1 knockout mice and in animals treated with other endothelin receptor antagonists indicates that teratogenicity is a class effect of these drugs. There are no data on the use of TRACLEER® in pregnant women.
Pregnancy must be excluded before the start of treatment with TRACLEER® and prevented thereafter by use of reliable contraception. It has been demonstrated that hormonal contraceptives, including oral, injectable, transdermal, and implantable contraceptives may not be reliable in the presence of TRACLEER® and should not be used as the sole contraceptive method in patients receiving TRACLEER® (see Drug Interactions: Hormonal Contraceptives, Including Oral, Injectable, Transdermal and Implantable Contraceptives). Input from a gynecologist or similar expert on adequate contraception should be sought as needed.
TRACLEER® should be started only in patients known not to be pregnant. For female patients of childbearing potential, a prescription for TRACLEER® should not be issued by the prescriber unless the patient assures the prescriber that she is not sexually active or provides negative results from a urine or serum pregnancy test performed during the first 5 days of a normal menstrual period and at least 11 days after the last unprotected act of sexual intercourse.
Follow-up urine or serum pregnancy tests should be obtained monthly in women of childbearing potential taking TRACLEER®. The patient must be advised that if there is any delay in onset of menses or any other reason to suspect pregnancy, she must notify the physician immediately for pregnancy testing. If the pregnancy test is positive, the physician and patient must discuss the risk to the pregnancy and to the fetus.
Cyclosporine A
Co-administration of cyclosporine A and bosentan resulted in markedly increased plasma concentrations of bosentan. Therefore, concomitant use of TRACLEER® and cyclosporine A is contraindicated.
Glyburide
An increased risk of liver enzyme elevations was observed in patients receiving glyburide concomitantly with bosentan. Therefore co-administration of glyburide and TRACLEER® is contraindicated.
Lopinavir/ritonavir or other ritonavir-containing HIV regimens
Co-administration of lopinavir/ritonavir and bosentan resulted in markedly increased plasma concentrations of bosentan. Therefore, concomitant use of TRACLEER® and lopinavir/ritonavir or other ritonavir-containing HIV regimens is contraindicated (see PRECAUTIONS/Drug Interactions).
Hypersensitivity
TRACLEER® is also contraindicated in patients who are hypersensitive to bosentan or any component of the medication.
WARNINGS
Potential Liver Injury
(see BOX WARNING)
Elevations in ALT or AST by more than 3 × ULN were observed in 11% of bosentan-treated patients (N = 658) compared to 2% of placebo-treated patients (N = 280). Three-fold increases were seen in 12% of 95 PAH patients on 125 mg b.i.d. and 14% of 70 PAH patients on 250 mg b.i.d. Eight-fold increases were seen in 2% of PAH patients on 125 mg b.i.d. and 7% of PAH patients on 250 mg b.i.d. Bilirubin increases to ≥3 × ULN were associated with aminotransferase increases in 2 of 658 (0.3%) of patients treated with bosentan.
The combination of hepatocellular injury (increases in aminotransferases of > 3 × ULN) and increases in total bilirubin (≥ 3 × ULN) is a marker for potential serious liver injury.1
Elevations of AST and/or ALT associated with bosentan are dose-dependent, occur both early and late in treatment, usually progress slowly, are typically asymptomatic, and usually have been reversible after treatment interruption or cessation. Aminotransferase elevations also may reverse spontaneously while continuing treatment with TRACLEER®.
Liver aminotransferase levels must be measured prior to initiation of treatment and then monthly. If elevated aminotransferase levels are seen, changes in monitoring and treatment must be initiated (see DOSAGE AND ADMINISTRATION). If liver aminotransferase elevations are accompanied by clinical symptoms of liver injury (such as nausea, vomiting, fever, abdominal pain, jaundice, or unusual lethargy or fatigue) or increases in bilirubin ≥ 2 × ULN, treatment should be stopped. There is no experience with the re-introduction of TRACLEER® in these circumstances.
Pre-existing Liver Impairment
Liver aminotransferase levels must be measured prior to initiation of treatment and then monthly. TRACLEER® should generally be avoided in patients with moderate or severe liver impairment (see Clinical Pharmacology and DOSAGE AND ADMINISTRATION). In addition, TRACLEER® should generally be avoided in patients with elevated aminotransferases (> 3 × ULN) because monitoring liver injury in these patients may be more difficult (see BOX WARNING).
Decreased Sperm Counts
An open-label, single arm, multicenter, safety study evaluated the effect on testicular function of TRACLEER® 62.5 mg b.i.d. for 4 weeks, followed by 125 mg b.i.d for 5 months. Twenty-five male patients with WHO functional class III and IV PAH and normal baseline sperm count were enrolled. Twenty-three completed the study and 2 discontinued due to adverse events not related to testicular function. There was a decline in sperm count of at least 50% in 25% of the patients after 3 or 6 months of treatment with TRACLEER®. Sperm count remained within the normal range in all 22 patients with data after 6 months and no changes in sperm morphology, sperm motility, or hormone levels were observed. One patient developed marked oligospermia at 3 months and the sperm count remained low with 2 follow-up measurements over the subsequent 6 weeks. TRACLEER® was discontinued and after two months the sperm count had returned to baseline levels. Based on these findings and preclinical data from endothelin receptor antagonists, it cannot be excluded that endothelin receptor antagonists such as TRACLEER® have an adverse effect on spermatogenesis.
PRECAUTIONS
Hematologic Changes
Treatment with TRACLEER® caused a dose-related decrease in hemoglobin and hematocrit. Hemoglobin levels should be monitored after 1 and 3 months of treatment and then every 3 months. The overall mean decrease in hemoglobin concentration for bosentan-treated patients was 0.9 g/dL (change to end of treatment). Most of this decrease of hemoglobin concentration was detected during the first few weeks of bosentan treatment and hemoglobin levels stabilized by 4–12 weeks of bosentan treatment. In placebo-controlled studies of all uses of bosentan, marked decreases in hemoglobin (> 15% decrease from baseline resulting in values < 11 g/dL) were observed in 6% of bosentan-treated patients and 3% of placebo-treated patients. In patients with pulmonary arterial hypertension treated with doses of 125 and 250 mg b.i.d., marked decreases in hemoglobin occurred in 3% compared to 1% in placebo-treated patients.
A decrease in hemoglobin concentration by at least 1 g/dL was observed in 57% of bosentan-treated patients as compared to 29% of placebo-treated patients. In 80% of those patients whose hemoglobin decreased by at least 1 g/dL, the decrease occurred during the first 6 weeks of bosentan treatment.
During the course of treatment the hemoglobin concentration remained within normal limits in 68% of bosentan-treated patients compared to 76% of placebo patients. The explanation for the change in hemoglobin is not known, but it does not appear to be hemorrhage or hemolysis.
It is recommended that hemoglobin concentrations be checked after 1 and 3 months, and every 3 months thereafter. If a marked decrease in hemoglobin concentration occurs, further evaluation should be undertaken to determine the cause and need for specific treatment.
Fluid retention
In a placebo-controlled trial of patients with severe chronic heart failure, there was an increased incidence of hospitalization for CHF associated with weight gain and increased leg edema during the first 4-8 weeks of treatment with TRACLEER®. In addition, there have been numerous post-marketing reports of fluid retention in patients with pulmonary hypertension, occurring within weeks after starting TRACLEER®. Patients required intervention with a diuretic, fluid management, or hospitalization for decompensating heart failure (see CLINICAL STUDIES; Congestive Heart Failure).
Pulmonary Veno-Occlusive Disease (PVOD)
Should signs of pulmonary edema occur when TRACLEER® is administered the possibility of associated PVOD should be considered and TRACLEER® should be discontinued.
Information for Patients
Patients are advised to consult the TRACLEER® Medication Guide on the safe use of TRACLEER®.
The physician should discuss with the patient the importance of monthly monitoring of serum aminotransferases and urine or serum pregnancy testing and of avoidance of pregnancy. The physician should discuss options for effective contraception and measures to prevent pregnancy with their female patients. Input from a gynecologist or similar expert on adequate contraception should be sought as needed.
Drug Interactions
Bosentan is metabolized by CYP2C9 and CYP3A4. Inhibition of these enzymes may increase the plasma concentration of bosentan (see ketoconazole). Concomitant administration of both a CYP2C9 inhibitor (such as fluconazole or amiodarone) and a strong CYP3A4 inhibitor (e.g., ketoconazole, itraconazole, ritonavir) or a moderate CYP3A4 inhibitor (e.g., amprenavir, erythromycin, fluconazole, diltiazem) with bosentan will likely lead to large increases in plasma concentrations of bosentan. Co-administration of such combinations of a CYP2C9 inhibitor plus a strong or moderate CYP3A4 inhibitor with TRACLEER® is not recommended.
Bosentan is an inducer of CYP3A4 and CYP2C9. Consequently plasma concentrations of drugs metabolized by these two isozymes will be decreased when TRACLEER® is co-administered. Bosentan had no relevant inhibitory effect on any CYP isozyme in vitro (CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A4). Consequently, TRACLEER® is not expected to increase the plasma concentrations of drugs metabolized by these enzymes.
Hormonal Contraceptives, Including Oral, Injectable, Transdermal, and Implantable Contraceptives
An interaction study demonstrated that co-administration of bosentan and the oral hormonal contraceptive Ortho-Novum® produced average decreases of norethindrone and ethinyl estradiol levels of 14% and 31%, respectively. However, decreases in exposure were as much as 56% and 66%, respectively, in individual subjects. Therefore, hormonal contraceptives, including oral, injectable, transdermal, and implantable forms, may not be reliable when TRACLEER® is co-administered. Women should practice additional methods of contraception and not rely on hormonal contraception alone when taking TRACLEER®.
Specific interaction studies have demonstrated the following:
Cyclosporine A
During the first day of concomitant administration, trough concentrations of bosentan were increased by about 30-fold. Steady-state bosentan plasma concentrations were 3- to 4-fold higher than in the absence of cyclosporine A. The concomitant administration of bosentan and cyclosporine A is contraindicated (see CONTRAINDICATIONS). Co-administration of bosentan decreased the plasma concentrations of cyclosporine A (a CYP3A4 substrate) by approximately 50%.
Tacrolimus
Co-administration of tacrolimus and bosentan has not been studied in man. Co-administration of tacrolimus and bosentan resulted in markedly increased plasma concentrations of bosentan in animals. Caution should be exercised if tacrolimus and bosentan are used together.
Glyburide
An increased risk of elevated liver aminotransferases was observed in patients receiving concomitant therapy with glyburide. Therefore, the concomitant administration of TRACLEER® and glyburide is contraindicated, and alternative hypoglycemic agents should be considered (see CONTRAINDICATIONS).
Co-administration of bosentan decreased the plasma concentrations of glyburide by approximately 40%. The plasma concentrations of bosentan were also decreased by approximately 30%. Bosentan is also expected to reduce plasma concentrations of other oral hypoglycemic agents that are predominantly metabolized by CYP2C9 or CYP3A4. The possibility of worsened glucose control in patients using these agents should be considered.
Ketoconazole
Co-administration of bosentan 125 mg b.i.d. and ketoconazole, a potent CYP3A4 inhibitor, increased the plasma concentrations of bosentan by approximately 2-fold. No dose adjustment of bosentan is necessary, but increased effects of bosentan should be considered.
Simvastatin and Other Statins
Co-administration of bosentan decreased the plasma concentrations of simvastatin (a CYP3A4 substrate), and its active β-hydroxy acid metabolite, by approximately 50%. The plasma concentrations of bosentan were not affected. Bosentan is also expected to reduce plasma concentrations of other statins that have significant metabolism by CYP3A4, such as lovastatin and atorvastatin. The possibility of reduced statin efficacy should be considered. Patients using CYP3A4 metabolized statins should have cholesterol levels monitored after TRACLEER® is initiated to see whether the statin dose needs adjustment.
Warfarin
Co-administration of bosentan 500 mg b.i.d. for 6 days decreased the plasma concentrations of both S-warfarin (a CYP2C9 substrate) and R-warfarin (a CYP3A4 substrate) by 29 and 38%, respectively. Clinical experience with concomitant administration of bosentan and warfarin in patients with pulmonary arterial hypertension did not show clinically relevant changes in INR or warfarin dose (baseline vs. end of the clinical studies), and the need to change the warfarin dose during the trials due to changes in INR or due to adverse events was similar among bosentan- and placebo-treated patients.
Digoxin, Nimodipine and Losartan
Bosentan has no significant pharmacokinetic interactions with digoxin and nimodipine, and losartan has no significant effect on plasma levels of bosentan.
Sildenafil
In healthy subjects, co-administration of multiple doses of 125 mg b.i.d bosentan and 80 mg t.i.d. sildenafil resulted in a reduction of sildenafil plasma concentrations by 63% and increased bosentan plasma concentrations by 50%. A dose adjustment of neither drug is necessary. This recommendation holds true when sildenafil is used for the treatment of pulmonary arterial hypertension or erectile dysfunction.
Iloprost
In a small, randomized, double-blind, placebo-controlled study (the STEP trial), 34 patients treated with bosentan 125 mg bid for at least 16 weeks tolerated the addition of inhaled iloprost (up to 5 mcg 6 to 9 times per day during waking hours). The mean daily inhaled dose was 27 mcg and the mean number of inhalations per day was 5.6.
Rifampicin
Coadministration of bosentan and rifampicin in normal volunteers resulted in a mean 6-fold increase in bosentan trough levels after the first concomitant dose, but about a 60% decrease in bosentan levels at steady-state. The effect of bosentan on rifampicin levels has not been assessed. When consideration of the potential benefits and known and unknown risks leads to concomitant use, measure LFTs weekly for the first 4 weeks before reverting to normal monitoring.
Lopinavir/ritonavir or other ritonavir-containing HIV regimens
Co-administration of TRACLEER® and lopinavir/rotinavir in healthy subjects resulted in initial and steady state trough plasma concentrations of bosentan that were approximately 48-fold and 5-fold higher, respectively, than those measured after TRACLEER® administered alone. This is most likely explained by inhibition by ritonavir of OATP-mediated uptake into hepatocytes, but the potential for increased toxicity cannot be excluded. Co-administration of bosentan and lopinavir/ritonavir or other ritonavir-containing HIV regimens is contraindicated (see CONTRAINDICATIONS).
Carcinogenesis, Mutagenesis, Impairment of Fertility
Two years of dietary administration of bosentan to mice produced an increased incidence of hepatocellular adenomas and carcinomas in males at doses as low as 450 mg/kg/day (about 8 times the maximum recommended human dose [MRHD] of 125 mg b.i.d., on a mg/m2 basis). In the same study, doses greater than 2000 mg/kg/day (about 32 times the MRHD) were associated with an increased incidence of colon adenomas in both males and females. In rats, dietary administration of bosentan for two years was associated with an increased incidence of brain astrocytomas in males at doses as low as 500 mg/kg/day (about 16 times the MRHD). In a comprehensive battery of in vitro tests (the microbial mutagenesis assay, the unscheduled DNA synthesis assay, the V-79 mammalian cell mutagenesis assay, and human lymphocyte assay) and an in vivo mouse micronucleus assay, there was no evidence for any mutagenic or clastogenic activity of bosentan.
Impairment of Fertility/Testicular Function
The development of testicular tubular atrophy and impaired fertility has been linked with the chronic administration of certain endothelin receptor antagonists in rodents.
Treatment with bosentan at oral doses of up to 1500 mg/kg/day (50 times the MRHD on a mg/m2 basis) or intravenous doses up to 40 mg/kg/day had no effects on sperm count, sperm motility, mating performance or fertility in male and female rats. An increased incidence of testicular tubular atrophy was observed in rats given bosentan orally at doses as low as 125 mg/kg/ day (about 4 times the MRHD and the lowest doses tested) for two years but not at doses as high as 1500 mg/kg/day (about 50 times the MRHD) for 6 months. Effects on sperm count and motility were evaluated only in the much shorter duration fertility studies in which males had been exposed to the drug for 4-6 weeks. An increased incidence of tubular atrophy was not observed in mice treated for 2 years at doses up to 4500 mg/kg/day (about 75 times the MRHD) or in dogs treated up to 12 months at doses up to 500 mg/kg/day (about 50 times the MRHD).
Pregnancy, Teratogenic Effects: Category X (See CONTRAINDICATIONS).
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, breastfeeding while taking TRACLEER® is not recommended.
Pediatric Use
Safety and efficacy in pediatric patients have not been established. (See DOSAGE AND ADMINISTRATION).
Use in Elderly Patients
Clinical experience with TRACLEER® in subjects aged 65 or older has not included a sufficient number of such subjects to identify a difference in response between elderly and younger patients (see DOSAGE AND ADMINISTRATION).
ADVERSE REACTIONS
Adverse Events
See BOX WARNING for discussion of liver injury and PRECAUTIONS for discussion of hemoglobin and hematocrit abnormalities.
Safety data on bosentan were obtained from 12 clinical studies (8 placebo-controlled and 4 open-label) in 777 patients with pulmonary arterial hypertension, and other diseases. Doses up to 8 times the currently recommended clinical dose (125 mg b.i.d.) were administered for a variety of durations. The exposure to bosentan in these trials ranged from 1 day to 4.1 years (N=89 for 1 year; N=61 for 1.5 years and N=39 for more than 2 years). Exposure of pulmonary arterial hypertension patients (N=235) to bosentan ranged from 1 day to 1.7 years (N=126 more than 6 months and N=28 more than 12 months).
Treatment discontinuations due to adverse events other than those related to pulmonary hypertension during the clinical trials in patients with pulmonary arterial hypertension were more frequent on bosentan (5%; 8/165 patients) than on placebo (3%; 2/80 patients). In this database the only cause of discontinuations > 1%, and occurring more often on bosentan was abnormal liver function.
The adverse drug reactions that occurred in ≥ 3% of the bosentan-treated patients and were more common on bosentan in placebo-controlled trials in pulmonary arterial hypertension at doses of 125 or 250 mg b.i.d. are shown in Table 4:
| Adverse Event | Bosentan N = 165 | Placebo N = 80 |
||
|---|---|---|---|---|
| No. | % | No. | % | |
|
||||
| Headache | 36 | 22% | 16 | 20% |
| Nasopharyngitis | 18 | 11% | 6 | 8% |
| Flushing | 15 | 9% | 4 | 5% |
| Hepatic function abnormal | 14 | 8% | 2 | 3% |
| Edema, lower limb | 13 | 8% | 4 | 5% |
| Hypotension | 11 | 7% | 3 | 4% |
| Palpitations | 8 | 5% | 1 | 1% |
| Dyspepsia | 7 | 4% | 0 | 0% |
| Edema | 7 | 4% | 2 | 3% |
| Fatigue | 6 | 4% | 1 | 1% |
| Pruritus | 6 | 4% | 0 | 0% |
In placebo-controlled studies of bosentan in pulmonary arterial hypertension and for other diseases (primarily chronic heart failure), a total of 677 patients were treated with bosentan at daily doses ranging from 100 mg to 2000 mg and 288 patients were treated with placebo. The duration of treatment ranged from 4 weeks to 6 months. For the adverse drug reactions that occurred in ≥ 3% of bosentan-treated patients, the only ones that occurred more frequently on bosentan than on placebo (≥ 2% difference) were headache (16% vs. 13%), flushing (7% vs. 2%), abnormal hepatic function (6% vs. 2%), leg edema (5% vs. 1%), and anemia (3% vs. 1%).
Post-Marketing Experience
Hypersensitivity, Rash, Thrombocytopenia, Jaundice, Anemia requiring transfusion
There have been several post-marketing reports of angioneurotic edema associated with the use of bosentan. The onset of the reported cases occurred within a range of 8 hours to 21 days after starting therapy. Some patients were treated with an antihistamine and their signs of angioedema resolved without discontinuing TRACLEER®.
In the post-marketing period, in the setting of close monitoring, rare cases of unexplained hepatic cirrhosis were reported after prolonged (> 12 months) therapy with TRACLEER® in patients with multiple co-morbidities and drug therapies. There have also been rare reports of liver failure. The contribution of TRACLEER® in these cases could not be excluded (see BOX WARNING).
Laboratory Abnormalities
Increased Liver Aminotransferases (see BOX WARNING and WARNINGS).
Decreased Hemoglobin and Hematocrit (see PRECAUTIONS)
OVERDOSAGE
Bosentan has been given as a single dose of up to 2400 mg in normal volunteers, or up to 2000 mg/day for 2 months in patients, without any major clinical consequences. The most common side effect was headache of mild to moderate intensity. In the cyclosporine A interaction study, in which doses of 500 and 1000 mg b.i.d. of bosentan were given concomitantly with cyclosporine A, trough plasma concentrations of bosentan increased 30-fold, resulting in severe headache, nausea, and vomiting, but no serious adverse events. Mild decreases in blood pressure and increases in heart rate were observed.
In the postmarketing period, there was one reported overdose of 10,000 mg of bosentan taken by an adolescent male patient. He had symptoms of nausea, vomiting, hypotension, dizziness, sweating, and blurred vision. He recovered within 24 hours with blood pressure support.
Bosentan is unlikely to be effectively removed by dialysis due to the high molecular weight and extensive plasma protein binding.
DOSAGE AND ADMINISTRATION
General
TRACLEER® treatment should be initiated at a dose of 62.5 mg b.i.d. for 4 weeks and then increased to the maintenance dose of 125 mg b.i.d. Doses above 125 mg b.i.d. did not appear to confer additional benefit sufficient to offset the increased risk of liver injury.
Tablets should be administered morning and evening with or without food.
| ALT/AST levels | Treatment and monitoring recommendations |
|---|---|
| > 3 and ≤ 5 × ULN | Confirm by another aminotransferase test; if confirmed, reduce the daily dose or interrupt treatment, and monitor aminotransferase levels at least every 2 weeks. If the aminotransferase levels return to pre-treatment values, continue or re-introduce the treatment as appropriate (see below). |
| > 5 and ≤ 8 × ULN | Confirm by another aminotransferase test; if confirmed, stop treatment and monitor aminotransferase levels at least every 2 weeks. Once the aminotransferase levels return to pre-treatment values, consider re-introduction of the treatment (see below). |
| > 8× ULN | Treatment should be stopped and re-introduction of TRACLEER® should not be considered. There is no experience with re-introduction of TRACLEER® in these circumstances. |
If TRACLEER® is re-introduced it should be at the starting dose; aminotransferase levels should be checked within 3 days and thereafter according to the recommendations above.
If liver aminotransferase elevations are accompanied by clinical symptoms of liver injury (such as nausea, vomiting, fever, abdominal pain, jaundice, or unusual lethargy or fatigue) or increases in bilirubin ≥ 2 × ULN, treatment should be stopped. There is no experience with the re-introduction of TRACLEER® in these circumstances.
Use in Women of Child-bearing Potential
TRACLEER® treatment should only be initiated in women of child-bearing potential following a negative pregnancy test and only in those who practice adequate contraception that does not rely solely upon hormonal contraceptives, including oral, injectable, transdermal, or implantable contraceptives (see DRUG INTERACTIONS: Hormonal Contraceptives, Including Oral, Injectable, Transdermal, and Implantable Contraceptives). Input from a gynecologist or similar expert on adequate contraception should be sought as needed. Urine or serum pregnancy tests should be obtained monthly in women of childbearing potential taking TRACLEER®.
Dosage Adjustment in Renally Impaired Patients
The effect of renal impairment on the pharmacokinetics of bosentan is small and does not require dosing adjustment.
Dosage Adjustment in Geriatric Patients
Clinical studies of TRACLEER® did not include sufficient numbers of subjects aged 65 and older to determine whether they respond differently from younger subjects. Clinical experience has not identified differences in responses between elderly and younger patients. In general, caution should be exercised in dose selection for elderly patients given the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy in this age group.
Dosage Adjustment in Hepatically Impaired Patients
Because there is in vitro and in vivo evidence that the main route of excretion of bosentan is biliary, liver impairment could be expected to increase exposure (Cmax and AUC) of bosentan. Mild liver impairment was shown not to impact the pharmacokinetics of bosentan. The influence of moderate or severe liver impairment on the pharmacokinetics of TRACLEER® has not been evaluated. There are no specific data to guide dosing in hepatically impaired patients (See WARNINGS); caution should be exercised in patients with mildly impaired liver function. TRACLEER® should generally be avoided in patients with moderate or severe liver impairment.
Dosage Adjustment in Children
Safety and efficacy in pediatric patients have not been established.
Dosage Adjustment in Patients with Low Body Weight
In patients with a body weight below 40 kg but who are over 12 years of age the recommended initial and maintenance dose is 62.5 mg b.i.d.
Discontinuation of Treatment
There is limited experience with abrupt discontinuation of TRACLEER®. No evidence for acute rebound has been observed. Nevertheless, to avoid the potential for clinical deterioration, gradual dose reduction (62.5 mg b.i.d. for 3 to 7 days) should be considered.
HOW SUPPLIED
62.5 mg film-coated, round, biconvex, orange-white tablets, embossed with identification marking "62,5", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap.
NDC 66215-101-06: Bottle containing 60 tablets.
125 mg film-coated, oval, biconvex, orange-white tablets, embossed with identification marking "125", packaged in a white high-density polyethylene bottle and a white polypropylene child-resistant cap.
NDC 66215-102-06: Bottle containing 60 tablets.
Rx only.
STORAGE
Store at 20°C – 25°C (68°F – 77°F). Excursions are permitted between 15°C and 30°C (59°F and 86°F). [See USP Controlled Room Temperature].
Distributed by:
Actelion Pharmaceuticals US, Inc.
South San Francisco, CA 94080, USA
Reference:
1. Zimmerman HJ. Hepatotoxicity - The adverse effects of drugs and other chemicals on the liver. Second ed. Philadelphia: Lippincott, 1999.
March 2009
ACT20090319
Medication Guide
Tracleer (tra-KLEER) Tablets
(bosentan)
Read this information carefully before you start taking Tracleer tablets. Read the information you get with Tracleer each time you refill your prescription. There may be new information. This information does not take the place of talking with your doctor.
What is the most important information I should know about Tracleer?
•Liver damage.
- Tracleer can cause liver damage if liver problems are not found early. Therefore, you must have a blood test to check your liver function before you start Tracleer and each month after that. (See "What are the possible side effects of Tracleer?" for information about the signs of liver problems.)
•Major birth defects.
- Tracleer can cause major birth defects if taken during pregnancy. Therefore, women must not be pregnant when they start taking Tracleer or during Tracleer treatment. Women who are sexually active must have a negative pregnancy test before beginning treatment. A negative test means you are not pregnant. The test should be during the first five days of a normal menstrual period and at least 11 days after the last unprotected sexual intercourse. Pregnancy tests must be done each month during Tracleer treatment, if you are sexually active.
- Women who are able to get pregnant must use effective birth control while taking Tracleer. Birth control pills, shots, patches, implants, or other hormone-based birth control may not be enough when Tracleer is used. Talk with your doctor and, if needed, with a gynecologist (a doctor who specializes in female reproduction) or another doctor who knows about birth control, to find out how to avoid pregnancy. Tell your doctor right away if you miss a period or think you may be pregnant.
What is Tracleer?
Tracleer is a medicine to treat pulmonary arterial hypertension, which is high blood pressure in the lung arteries. You take it by mouth.
Tracleer can improve your ability to exercise and can slow the worsening of your physical condition and symptoms. Tracleer lowers high blood pressure in your lungs and lets your heart pump blood more effectively.
Who should not take Tracleer?
Do not take Tracleer if:
- you are pregnant, plan to become pregnant, or become pregnant during Tracleer treatment. Tracleer can cause major birth defects. All women should read the birth defects section of "What is the most important information I should know about Tracleer?" Severe birth defects from Tracleer happen early in pregnancy. Therefore, you must not be pregnant while taking Tracleer.
- your blood test shows possible liver injury
- you are taking cyclosporine-A, (used for psoriasis and rheumatoid arthritis, and to prevent rejection of heart or kidney transplants), glyburide (used for diabetes), or lopinavir/ritonavir or other ritonavir-containing HIV regimens.
- you are allergic to any ingredients in Tracleer. The active ingredient is bosentan. Ask your doctor or pharmacist if you need to know the inactive ingredients.
Tell your doctor if you have moderate or severe liver problems. Tracleer may not be right for you.
Tell your doctor about all the medicines you use. They may affect how Tracleer works, or Tracleer may affect how the other medicines work. Be sure to tell your doctor if you take
- ketoconazole, fluconazole, itraconazole or voriconazole (used for fungal infections)
- hormone-based birth control, such as pills, shots, patches, and implants
- cyclosporine A (used for psoriasis and rheumatoid arthritis, and to prevent rejection of heart or kidney transplants)
- tacrolimus (used to prevent rejection of liver or kidney transplants)
- rifampicin (used for tuberculosis)
- glyburide (used for diabetes)
- cholesterol lowering medicines
- warfarin (used to prevent blood clots)
- lopinavir/ritonavir or other ritonavir-containing HIV regimens.
How should I take Tracleer?
Tracleer will be mailed to you by a central pharmacy. Your doctor will give you complete details:
- In most cases, you will take 1 tablet in the morning and 1 in the evening.
- You can take it with or without food.
- Your doctor will tell you how much to take.
- It will be easier to remember to take Tracleer if you do it at the same time each morning and evening. If you have trouble remembering, ask a family member to remind you, or put written notes where you will be sure to see them.
- If you take more than the prescribed dose of Tracleer, call your doctor right away.
- If you miss a dose, take your tablet as soon as you remember. However, do not take 2 doses to make up for a missed dose. Take your next tablet at the regular time.
- Do not stop taking Tracleer unless your doctor tells you to do so. Suddenly stopping your treatment may cause your symptoms to get worse. If you need to stop taking Tracleer, your doctor may tell you to reduce the dose over a few days before stopping completely.
During treatment your doctor will test your blood for signs of side effects to your liver and red blood cells.
What should I avoid while taking Tracleer?
- Do not get pregnant while taking Tracleer. (See the birth defect section of "What is the most important information I should know about Tracleer?") If you miss a period, call your doctor.
- Breast feeding is not recommended while taking Tracleer. It is not known if Tracleer can pass through your milk and harm the baby.
- Do not use hormone-based birth control (pills, shots, patches, implants) as your only method of birth control. These may not work when used with Tracleer. Ask your doctor about effective birth control choices.
- Do not take cyclosporine-A. This medicine can cause too much Tracleer in your blood and increase your chance of side effects.
- Do not take glyburide. This medicine can increase your chance of liver damage.
- Do not take lopinavir/ritonavir or other ritonavir-containing HIV regimens. They can cause too much Tracleer in your blood and increase your chance of side effects.
What are the possible side effects of Tracleer?
Tracleer can have serious side effects:
-
Liver damage. Tracleer can cause liver damage if it is not found early. Because this side effect may not cause symptoms at first, only a blood test can show that you have early liver damage. Regular blood tests let your doctor change or stop your therapy before there is permanent damage. Therefore, it is very important that you have a liver function blood test before you start treatment and every month after that.
Call your doctor right away if you have any of these symptoms of liver problems: nausea, vomiting, fever, unusual tiredness, abdominal (stomach area) pain, or yellowing of the skin or the whites of your eyes (jaundice). - Major birth defects. All females should read the birth defects section of "What is the most important information I should know about Tracleer?"
- Sperm Count Reduction. Reduced sperm counts have been observed in some men on Tracleer, which might impair ability to father a child. Tell your doctor if remaining fertile is important to you.
Other possible side effects
The most common side effects of Tracleer are:
- low red blood cell levels (anemia)
- headache
- inflamed throat and irritated nose passages
- flushing (hot flashes)
- ankle and leg swelling
- low blood pressure
- irregular heart beats
- upset stomach
- tiredness
- rash
- itching
General advice about prescription medicines
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. If you have any concerns or questions about Tracleer, ask your doctor or other healthcare provider. This Medication Guide is only a summary of some important information about Tracleer. Your doctor can give you information about Tracleer that was written for healthcare professionals. Do not use Tracleer for a condition for which it was not prescribed. Do not share Tracleer with other people.
This Medication Guide has been approved by the US Food and Drug Administration.
March 2009
©2009 Actelion Pharmaceuticals US, Inc.
| TRACLEER
bosentan tablet, film coated |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
| TRACLEER
bosentan tablet, film coated |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
Revised: 04/2009Actelion Pharmaceuticals US, Inc.