EPOGEN
-
epoetin alfa solution
Amgen
----------
EPOGEN®
(Epoetin alfa)
FOR INJECTION
WARNINGS: INCREASED MORTALITY, SERIOUS CARDIOVASCULAR and THROMBOEMBOLIC EVENTS, and INCREASED RISK OF TUMOR PROGRESSION OR RECURRENCE
Renal failure: Patients experienced greater risks for death and serious cardiovascular events when administered erythropoiesis-stimulating agents (ESAs) to target higher versus lower hemoglobin levels (13.5 vs. 11.3 g/dL; 14 vs. 10 g/dL) in two clinical studies. Individualize dosing to achieve and maintain hemoglobin levels within the range of 10 to 12 g/dL.
Cancer:
- ESAs shortened overall survival and/or increased the risk of tumor progression or recurrence in some clinical studies in patients with breast, non-small cell lung, head and neck, lymphoid, and cervical cancers (see WARNINGS: Table 1).
- To decrease these risks, as well as the risk of serious cardio- and thrombovascular events, use the lowest dose needed to avoid red blood cell transfusion.
- Use ESAs only for treatment of anemia due to concomitant myelosuppressive chemotherapy.
- ESAs are not indicated for patients receiving myelosuppressive therapy when the anticipated outcome is cure.
- Discontinue following the completion of a chemotherapy course.
Perisurgery: EPOGEN® increased the rate of deep venous thromboses in patients not receiving prophylactic anticoagulation. Consider deep venous thrombosis prophylaxis.
(See WARNINGS: Increased Mortality, Serious Cardiovascular and Thromboembolic Events, WARNINGS: Increased Mortality and/or Increased Risk of Tumor Progression or Recurrence, INDICATIONS AND USAGE, and DOSAGE AND ADMINISTRATION.)
DESCRIPTION
Erythropoietin is a glycoprotein which stimulates red blood cell production. It is produced in the kidney and stimulates the division and differentiation of committed erythroid progenitors in the bone marrow. EPOGEN® (Epoetin alfa), a 165 amino acid glycoprotein manufactured by recombinant DNA technology, has the same biological effects as endogenous erythropoietin.1 It has a molecular weight of 30,400 daltons and is produced by mammalian cells into which the human erythropoietin gene has been introduced. The product contains the identical amino acid sequence of isolated natural erythropoietin.
EPOGEN® is formulated as a sterile, colorless liquid in an isotonic sodium chloride/sodium citrate buffered solution or a sodium chloride/sodium phosphate buffered solution for intravenous (IV) or subcutaneous (SC) administration.
Single-dose, Preservative-free Vial: Each 1 mL of solution contains 2000, 3000, 4000 or 10,000 Units of Epoetin alfa, 2.5 mg Albumin (Human), 5.8 mg sodium citrate, 5.8 mg sodium chloride, and 0.06 mg citric acid in Water for Injection, USP (pH 6.9 ± 0.3). This formulation contains no preservative.
Single-dose, Preservative-free Vial: 1 mL (40,000 Units/mL). Each 1 mL of solution contains 40,000 Units of Epoetin alfa, 2.5 mg Albumin (Human), 1.2 mg sodium phosphate monobasic monohydrate, 1.8 mg sodium phosphate dibasic anhydrate, 0.7 mg sodium citrate, 5.8 mg sodium chloride, and 6.8 mcg citric acid in Water for Injection, USP (pH 6.9 ± 0.3). This formulation contains no preservative.
Multidose, Preserved Vial: 2 mL (20,000 Units, 10,000 Units/mL). Each 1 mL of solution contains 10,000 Units of Epoetin alfa, 2.5 mg Albumin (Human), 1.3 mg sodium citrate, 8.2 mg sodium chloride, 0.11 mg citric acid, and 1% benzyl alcohol as preservative in Water for Injection, USP (pH 6.1 ± 0.3).
Multidose, Preserved Vial: 1 mL (20,000 Units/mL). Each 1 mL of solution contains 20,000 Units of Epoetin alfa, 2.5 mg Albumin (Human), 1.3 mg sodium citrate, 8.2 mg sodium chloride, 0.11 mg citric acid, and 1% benzyl alcohol as preservative in Water for Injection, USP (pH 6.1 ± 0.3).
CLINICAL PHARMACOLOGY
Chronic Renal Failure Patients
Endogenous production of erythropoietin is normally regulated by the level of tissue oxygenation. Hypoxia and anemia generally increase the production of erythropoietin, which in turn stimulates erythropoiesis.2 In normal subjects, plasma erythropoietin levels range from 0.01 to 0.03 Units/mL and increase up to 100- to 1000-fold during hypoxia or anemia.2 In contrast, in patients with chronic renal failure (CRF), production of erythropoietin is impaired, and this erythropoietin deficiency is the primary cause of their anemia.3,4
Chronic renal failure is the clinical situation in which there is a progressive and usually irreversible decline in kidney function. Such patients may manifest the sequelae of renal dysfunction, including anemia, but do not necessarily require regular dialysis. Patients with end-stage renal disease (ESRD) are those patients with CRF who require regular dialysis or kidney transplantation for survival.
EPOGEN® has been shown to stimulate erythropoiesis in anemic patients with CRF, including both patients on dialysis and those who do not require regular dialysis.4-12 The first evidence of a response to the three times weekly (TIW) administration of EPOGEN® is an increase in the reticulocyte count within 10 days, followed by increases in the red cell count, hemoglobin, and hematocrit, usually within 2 to 6 weeks.4,5 Because of the length of time required for erythropoiesis — several days for erythroid progenitors to mature and be released into the circulation — a clinically significant increase in hematocrit is usually not observed in less than 2 weeks and may require up to 6 weeks in some patients. Once the hematocrit reaches the suggested target range (30% to 36%), that level can be sustained by EPOGEN® therapy in the absence of iron deficiency and concurrent illnesses.
The rate of hematocrit increase varies between patients and is dependent upon the dose of EPOGEN®, within a therapeutic range of approximately 50 to 300 Units/kg TIW.4 A greater biologic response is not observed at doses exceeding 300 Units/kg TIW.6 Other factors affecting the rate and extent of response include availability of iron stores, the baseline hematocrit, and the presence of concurrent medical problems.
Zidovudine-treated HIV-infected Patients
Responsiveness to EPOGEN® in HIV-infected patients is dependent upon the endogenous serum erythropoietin level prior to treatment. Patients with endogenous serum erythropoietin levels ≤ 500 mUnits/mL, and who are receiving a dose of zidovudine ≤ 4200 mg/week, may respond to EPOGEN® therapy. Patients with endogenous serum erythropoietin levels > 500 mUnits/mL do not appear to respond to EPOGEN® therapy. In a series of four clinical trials involving 255 patients, 60% to 80% of HIV-infected patients treated with zidovudine had endogenous serum erythropoietin levels ≤ 500 mUnits/mL.
Response to EPOGEN® in zidovudine-treated HIV-infected patients is manifested by reduced transfusion requirements and increased hematocrit.
Cancer Patients on Chemotherapy
A series of clinical trials enrolled 131 anemic cancer patients who received EPOGEN® TIW and who were receiving cyclic cisplatin- or non cisplatin-containing chemotherapy. Endogenous baseline serum erythropoietin levels varied among patients in these trials with approximately 75% (n = 83/110) having endogenous serum erythropoietin levels ≤ 132 mUnits/mL, and approximately 4% (n = 4/110) of patients having endogenous serum erythropoietin levels > 500 mUnits/mL. In general, patients with lower baseline serum erythropoietin levels responded more vigorously to EPOGEN® than patients with higher baseline erythropoietin levels. Although no specific serum erythropoietin level can be stipulated above which patients would be unlikely to respond to EPOGEN® therapy, treatment of patients with grossly elevated serum erythropoietin levels (eg, > 200 mUnits/mL) is not recommended.
Pharmacokinetics
In adult and pediatric patients with CRF, the elimination half-life of plasma erythropoietin after intravenously administered EPOGEN® ranges from 4 to 13 hours.13-15 The half-life is approximately 20% longer in CRF patients than that in healthy subjects. After SC administration, peak plasma levels are achieved within 5 to 24 hours. The half-life is similar between adult patients with serum creatinine level greater than 3 and not on dialysis and those maintained on dialysis. The pharmacokinetic data indicate no apparent difference in EPOGEN® half-life among adult patients above or below 65 years of age.
The pharmacokinetic profile of EPOGEN® in children and adolescents appears to be similar to that of adults. Limited data are available in neonates.16 A study of 7 preterm very low birth weight neonates and 10 healthy adults given IV erythropoietin suggested that distribution volume was approximately 1.5 to 2 times higher in the preterm neonates than in the healthy adults, and clearance was approximately 3 times higher in the preterm neonates than in the healthy adults.39
The pharmacokinetics of EPOGEN® have not been studied in HIV-infected patients.
A pharmacokinetic study comparing 150 Units/kg SC TIW to 40,000 Units SC weekly dosing regimen was conducted for 4 weeks in healthy subjects (n = 12) and for 6 weeks in anemic cancer patients (n = 32) receiving cyclic chemotherapy. There was no accumulation of serum erythropoietin after the 2 dosing regimens during the study period. The 40,000 Units weekly regimen had a higher Cmax (3- to 7-fold), longer Tmax (2- to 3-fold), higher AUC0-168h (2- to 3-fold) of erythropoietin and lower clearance (50%) than the 150 Units/kg TIW regimen. In anemic cancer patients, the average t1/2 was similar (40 hours with range of 16 to 67 hours) after both dosing regimens. After the 150 Units/kg TIW dosing, the values of Tmax and clearance are similar (13.3 ± 12.4 vs. 14.2 ± 6.7 hours, and 20.2 ± 15.9 vs. 23.6 ± 9.5 mL/h/kg) between Week 1 when patients were receiving chemotherapy (n = 14) and Week 3 when patients were not receiving chemotherapy (n = 4). Differences were observed after the 40,000 Units weekly dosing with longer Tmax (38 ± 18 hours) and lower clearance (9.2 ± 4.7 mL/h/kg) during Week 1 when patients were receiving chemotherapy (n = 18) compared with those (22 ± 4.5 hours, 13.9 ± 7.6 mL/h/kg) during Week 3 when patients were not receiving chemotherapy (n = 7).
The bioequivalence between the 10,000 Units/mL citrate-buffered Epoetin alfa formulation and the 40,000 Units/mL phosphate-buffered Epoetin alfa formulation has been demonstrated after SC administration of single 750 Units/kg doses to healthy subjects.
INDICATIONS AND USAGE
Treatment of Anemia of Chronic Renal Failure Patients
EPOGEN® is indicated for the treatment of anemia associated with CRF, including patients on dialysis and patients not on dialysis. EPOGEN® is indicated to elevate or maintain the red blood cell level (as manifested by the hematocrit or hemoglobin determinations) and to decrease the need for transfusions in these patients.
Non-dialysis patients with symptomatic anemia considered for therapy should have a hemoglobin less than 10 g/dL.
EPOGEN® is not intended for patients who require immediate correction of severe anemia. EPOGEN® may obviate the need for maintenance transfusions but is not a substitute for emergency transfusion.
Prior to initiation of therapy, the patient’s iron stores should be evaluated. Transferrin saturation should be at least 20% and ferritin at least 100 ng/mL. Blood pressure should be adequately controlled prior to initiation of EPOGEN® therapy, and must be closely monitored and controlled during therapy.
Treatment of Anemia in Zidovudine-treated HIV-infected Patients
EPOGEN® is indicated for the treatment of anemia related to therapy with zidovudine in HIV-infected patients. EPOGEN® is indicated to elevate or maintain the red blood cell level (as manifested by the hematocrit or hemoglobin determinations) and to decrease the need for transfusions in these patients. EPOGEN® is not indicated for the treatment of anemia in HIV-infected patients due to other factors such as iron or folate deficiencies, hemolysis, or gastrointestinal bleeding, which should be managed appropriately. EPOGEN® use has not been demonstrated in controlled clinical trials to improve symptoms of anemia, quality of life, fatigue, or patient well-being.
EPOGEN®, at a dose of 100 Units/kg TIW, is effective in decreasing the transfusion requirement and increasing the red blood cell level of anemic, HIV-infected patients treated with zidovudine, when the endogenous serum erythropoietin level is ≤ 500 mUnits/mL and when patients are receiving a dose of zidovudine ≤ 4200 mg/week.
Treatment of Anemia in Cancer Patients on Chemotherapy
EPOGEN® is indicated for the treatment of anemia due to the effect of concomitantly administered chemotherapy based on studies that have shown a reduction in the need for RBC transfusions in patients with metastatic, non-myeloid malignancies receiving chemotherapy for a minimum of 2 months. Studies to determine whether EPOGEN® increases mortality or decreases progression-free/recurrence-free survival are ongoing.
- EPOGEN® is not indicated for use in patients receiving hormonal agents, therapeutic biologic products, or radiotherapy unless receiving concomitant myelosuppressive chemotherapy.
- EPOGEN® is not indicated for patients receiving myelosuppressive therapy when the anticipated outcome is cure due to the absence of studies that adequately characterize the impact of EPOGEN® on progression-free and overall survival (see WARNINGS: Increased Mortality and/or Increased Risk of Tumor Progression or Recurrence).
- EPOGEN® is not indicated for the treatment of anemia in cancer patients due to other factors such as iron or folate deficiencies, hemolysis, or gastrointestinal bleeding (see PRECAUTIONS: Lack or Loss of Response).
- EPOGEN® use has not been demonstrated in controlled clinical trials to improve symptoms of anemia, quality of life, fatigue, or patient well-being.
Reduction of Allogeneic Blood Transfusion in Surgery Patients
EPOGEN® is indicated for the treatment of anemic patients (hemoglobin > 10 to ≤ 13 g/dL) who are at high risk for perioperative blood loss from elective, noncardiac, nonvascular surgery to reduce the need for allogeneic blood transfusions.17-19 EPOGEN® is not indicated for anemic patients who are willing to donate autologous blood (see BOXED WARNINGS and DOSAGE AND ADMINISTRATION).
CLINICAL EXPERIENCE: RESPONSE TO EPOGEN®
Chronic Renal Failure Patients
When dosed with EPOGEN®, patients responded with an increase in hematocrit.5 After 3 months on study, more than 95% of patients were transfusion-independent.
In the presence of adequate iron stores (see IRON EVALUATION), the time to reach the target hematocrit is a function of the baseline hematocrit and the rate of hematocrit rise.
The rate of increase in hematocrit is dependent upon the dose of EPOGEN® administered and individual patient variation. In clinical trials at starting doses of 50 to 150 Units/kg TIW, adult patients responded with an average rate of hematocrit rise of:
| Starting Dose | Hematocrit Increase | |
| (TIW IV) | Points/Day | Points/2 Weeks |
| 50 Units/kg | 0.11 | 1.5 |
| 100 Units/kg | 0.18 | 2.5 |
| 150 Units/kg | 0.25 | 3.5 |
In a 26 week, double-blind, placebo-controlled trial, 118 anemic dialysis patients with an average hemoglobin of approximately 7 g/dL were randomized to either EPOGEN® or placebo. By the end of the study, average hemoglobin increased to approximately 11 g/dL in the EPOGEN®-treated patients and remained unchanged in patients receiving placebo. EPOGEN®-treated patients experienced improvements in exercise tolerance and patient-reported physical functioning at month 2 that was maintained throughout the study.
Adult Patients on Dialysis: Thirteen clinical studies were conducted, involving IV administration to a total of 1010 anemic patients on dialysis for 986 patient-years of EPOGEN® therapy. In the three largest of these clinical trials, the median maintenance dose necessary to maintain the hematocrit between 30% to 36% was approximately 75 Units/kg TIW. In the US multicenter phase 3 study, approximately 65% of the patients required doses of 100 Units/kg TIW, or less, to maintain their hematocrit at approximately 35%. Almost 10% of patients required a dose of 25 Units/kg, or less, and approximately 10% required a dose of more than 200 Units/kg TIW to maintain their hematocrit at this level.
A multicenter unit dose study was also conducted in 119 patients receiving peritoneal dialysis who self-administered EPOGEN® subcutaneously for approximately 109 patient-years of experience. Patients responded to EPOGEN® administered SC in a manner similar to patients receiving IV administration.20
Pediatric Patients on Dialysis: One hundred twenty-eight children from 2 months to 19 years of age with CRF requiring dialysis were enrolled in 4 clinical studies of EPOGEN®. The largest study was a placebo-controlled, randomized trial in 113 children with anemia (hematocrit ≤ 27%) undergoing peritoneal dialysis or hemodialysis. The initial dose of EPOGEN® was 50 Units/kg IV or SC TIW. The dose of study drug was titrated to achieve either a hematocrit of 30% to 36% or an absolute increase in hematocrit of 6 percentage points over baseline.
At the end of the initial 12 weeks, a statistically significant rise in mean hematocrit (9.4% vs 0.9%) was observed only in the EPOGEN® arm. The proportion of children achieving a hematocrit of 30%, or an increase in hematocrit of 6 percentage points over baseline, at any time during the first 12 weeks was higher in the EPOGEN® arm (96% vs 58%). Within 12 weeks of initiating EPOGEN® therapy, 92.3% of the pediatric patients were transfusion-independent as compared to 65.4% who received placebo. Among patients who received 36 weeks of EPOGEN®, hemodialysis patients required a higher median maintenance dose (167 Units/kg/week [n = 28] vs 76 Units/kg/week [n = 36]) and took longer to achieve a hematocrit of 30% to 36% (median time to response 69 days vs 32 days) than patients undergoing peritoneal dialysis.
Patients With CRF Not Requiring Dialysis
Four clinical trials were conducted in patients with CRF not on dialysis involving 181 patients treated with EPOGEN® for approximately 67 patient-years of experience. These patients responded to EPOGEN® therapy in a manner similar to that observed in patients on dialysis. Patients with CRF not on dialysis demonstrated a dose-dependent and sustained increase in hematocrit when EPOGEN® was administered by either an IV or SC route, with similar rates of rise of hematocrit when EPOGEN® was administered by either route. Moreover, EPOGEN® doses of 75 to 150 Units/kg per week have been shown to maintain hematocrits of 36% to 38% for up to 6 months.21-22
Zidovudine-treated HIV-infected Patients
Efficacy in HIV-infected patients with anemia related to therapy with zidovudine was demonstrated based on reduction in the requirement for RBC transfusions.
EPOGEN® has been studied in four placebo-controlled trials enrolling 297 anemic (hematocrit < 30%) HIV-infected (AIDS) patients receiving concomitant therapy with zidovudine (all patients were treated with Epoetin alfa manufactured by Amgen Inc). In the subgroup of patients (89/125 EPOGEN® and 88/130 placebo) with prestudy endogenous serum erythropoietin levels ≤ 500 mUnits/mL, EPOGEN® reduced the mean cumulative number of units of blood transfused per patient by approximately 40% as compared to the placebo group.24 Among those patients who required transfusions at baseline, 43% of patients treated with EPOGEN® versus 18% of placebo-treated patients were transfusion-independent during the second and third months of therapy. EPOGEN® therapy also resulted in significant increases in hematocrit in comparison to placebo. When examining the results according to the weekly dose of zidovudine received during month 3 of therapy, there was a statistically significant (p < 0.003) reduction in transfusion requirements in patients treated with EPOGEN® (n = 51) compared to placebo treated patients (n = 54) whose mean weekly zidovudine dose was ≤ 4200 mg/week.23
Approximately 17% of the patients with endogenous serum erythropoietin levels ≤ 500 mUnits/mL receiving EPOGEN® in doses from 100 to 200 Units/kg TIW achieved a hematocrit of 38% without administration of transfusions or significant reduction in zidovudine dose. In the subgroup of patients whose prestudy endogenous serum erythropoietin levels were > 500 mUnits/mL, EPOGEN® therapy did not reduce transfusion requirements or increase hematocrit, compared to the corresponding responses in placebo-treated patients.
In a 6 month open-label EPOGEN® study, patients responded with decreased transfusion requirements and sustained increases in hematocrit and hemoglobin with doses of EPOGEN® up to 300 Units/kg TIW.23-25
Responsiveness to EPOGEN® therapy may be blunted by intercurrent infectious/inflammatory episodes and by an increase in zidovudine dosage. Consequently, the dose of EPOGEN® must be titrated based on these factors to maintain the desired erythropoietic response.
Cancer Patients on Chemotherapy
Adult Patients
Efficacy in patients with anemia due to concomitant chemotherapy was demonstrated based on reduction in the requirement for RBC transfusions.
Three-Times Weekly (TIW) Dosing
EPOGEN® administered TIW has been studied in a series of six placebo-controlled, double-blind trials that enrolled 131 anemic cancer patients receiving EPOGEN® or matching placebo. Across all studies, 72 patients were treated with concomitant non cisplatin-containing chemotherapy regimens and 59 patients were treated with concomitant cisplatin-containing chemotherapy regimens. Patients were randomized to EPOGEN® 150 Units/kg or placebo subcutaneously TIW for 12 weeks in each study.
The results of the pooled data from these six studies are shown in the table below. Because of the length of time required for erythropoiesis and red cell maturation, the efficacy of EPOGEN® (reduction in proportion of patients requiring transfusions) is not manifested until 2 to 6 weeks after initiation of EPOGEN®.
| Chemotherapy Regimen | On Study† | During Months 2 and 3‡ | ||
| EPOGEN® | Placebo | EPOGEN® | Placebo | |
|
||||
| Regimens without cisplatin | 44% (15/34) | 44% (16/36) | 21% (6/29) | 33% (11/33) |
| Regimens containing cisplatin | 50% (14/28) | 63% (19/30) | 23% (5/22)§ | 56% (14/25) |
| Combined | 47% (29/62) | 53% (35/66) | 22% (11/51)§ | 43% (25/58) |
Intensity of chemotherapy in the above trials was not directly assessed, however the degree and timing of neutropenia was comparable across all trials. Available evidence suggests that patients with lymphoid and solid cancers respond similarly to EPOGEN® therapy, and that patients with or without tumor infiltration of the bone marrow respond similarly to EPOGEN® therapy.
Weekly (QW) Dosing
EPOGEN® was also studied in a placebo-controlled, double-blind trial utilizing weekly dosing in a total of 344 anemic cancer patients. In this trial, 61 (35 placebo arm and 26 in the EPOGEN® arm) patients were treated with concomitant cisplatin containing regimens and 283 patients received concomitant chemotherapy regimens that did not contain cisplatinum. Patients were randomized to EPOGEN® 40,000 Units weekly (n = 174) or placebo (n = 170) SC for a planned treatment period of 16 weeks. If hemoglobin had not increased by > 1 g/dL after 4 weeks of therapy or the patient received RBC transfusion during the first 4 weeks of therapy, study drug was increased to 60,000 Units weekly. Forty-three percent of patients in the Epoetin alfa group required an increase in EPOGEN® dose to 60,000 Units weekly. 23
Results demonstrated that EPOGEN® therapy reduced the proportion of patients transfused in day 29 through week 16 of the study as compared to placebo. Twenty-five patients (14%) in the EPOGEN® group received transfusions compared to 48 patients (28%) in the placebo group (p = 0.0010) between day 29 and week 16 or the last day on study.
Comparable intensity of chemotherapy for patients enrolled in the two study arms was suggested by similarities in mean dose and frequency of administration for the 10 most commonly administered chemotherapy agents, and similarity in the incidence of changes in chemotherapy during the trial in the two arms.
Pediatric Patients
The safety and effectiveness of EPOGEN® were evaluated in a randomized, double-blind, placebo-controlled, multicenter study in anemic patients ages 5 to 18 receiving chemotherapy for the treatment of various childhood malignancies. Two hundred twenty-two patients were randomized (1:1) to EPOGEN® or placebo. EPOGEN® was administered at 600 Units/kg (maximum 40,000 Units) intravenously once per week for 16 weeks. If hemoglobin had not increased by 1g/dL after the first 4-5 weeks of therapy, EPOGEN® was increased to 900 Units/kg (maximum 60,000 Units). Among the EPOGEN®-treated patients 60% required dose escalation to 900 Units/kg/week.
The effect of EPOGEN® on transfusion requirements is shown in the table below:
| On Study* | After 28 Days
Post-Randomization |
||
| EPOGEN®
(n=111) | Placebo
(n=111) | EPOGEN®
(n= 111) | Placebo
(n=111) |
| 65% (72) | 77% (86) | 51%(57)† | 69% (77) |
There was no evidence of an improvement in health-related quality of life, including no evidence of an effect on fatigue, energy or strength, in patients receiving EPOGEN® as compared to those receiving placebo.
Surgery Paients
EPOGEN® has been studied in a placebo-controlled, double-blind trial enrolling 316 patients scheduled for major, elective orthopedic hip or knee surgery who were expected to require ≥ 2 units of blood and who were not able or willing to participate in an autologous blood donation program. Based on previous studies which demonstrated that pretreatment hemoglobin is a predictor of risk of receiving transfusion,19,26 patients were stratified into one of three groups based on their pretreatment hemoglobin [≤ 10 (n = 2), > 10 to ≤ 13 (n = 96), and > 13 to ≤ 15 g/dL (n = 218)] and then randomly assigned to receive 300 Units/kg EPOGEN®, 100 Units/kg EPOGEN® or placebo by SC injection for 10 days before surgery, on the day of surgery, and for 4 days after surgery.17 All patients received oral iron and a low-dose post-operative warfarin regimen.17
Treatment with EPOGEN® 300 Units/kg significantly (p = 0.024) reduced the risk of allogeneic transfusion in patients with a pretreatment hemoglobin of > 10 to ≤ 13; 5/31 (16%) of EPOGEN® 300 Units/kg, 6/26 (23%) of EPOGEN® 100 Units/kg, and 13/29 (45%) of placebo-treated patients were transfused.17 There was no significant difference in the number of patients transfused between EPOGEN® (9% 300 Units/kg, 6% 100 Units/kg) and placebo (13%) in the > 13 to ≤ 15 g/dL hemoglobin stratum. There were too few patients in the ≤ 10 g/dL group to determine if EPOGEN® is useful in this hemoglobin strata. In the > 10 to ≤ 13 g/dL pretreatment stratum, the mean number of units transfused per EPOGEN®-treated patient (0.45 units blood for 300 Units/kg, 0.42 units blood for 100 Units/kg) was less than the mean transfused per placebo-treated patient (1.14 units) (overall p = 0.028). In addition, mean hemoglobin, hematocrit, and reticulocyte counts increased significantly during the presurgery period in patients treated with EPOGEN®.17
EPOGEN® was also studied in an open-label, parallel-group trial enrolling 145 subjects with a pretreatment hemoglobin level of ≥ 10 to ≤ 13 g/dL who were scheduled for major orthopedic hip or knee surgery and who were not participating in an autologous program.18 Subjects were randomly assigned to receive one of two SC dosing regimens of EPOGEN® (600 Units/kg once weekly for 3 weeks prior to surgery and on the day of surgery, or 300 Units/kg once daily for 10 days prior to surgery, on the day of surgery and for 4 days after surgery). All subjects received oral iron and appropriate pharmacologic anticoagulation therapy.
From pretreatment to presurgery, the mean increase in hemoglobin in the 600 Units/kg weekly group (1.44 g/dL) was greater than observed in the 300 Units/kg daily group.18 The mean increase in absolute reticulocyte count was smaller in the weekly group (0.11 x 106/mm3) compared to the daily group (0.17 x 106/mm3). Mean hemoglobin levels were similar for the two treatment groups throughout the postsurgical period.
The erythropoietic response observed in both treatment groups resulted in similar transfusion rates [11/69 (16%) in the 600 Units/kg weekly group and 14/71 (20%) in the 300 Units/kg daily group].18 The mean number of units transfused per subject was approximately 0.3 units in both treatment groups.
CONTRAINDICATIONS
EPOGEN® is contraindicated in patients with:
- Uncontrolled hypertension.
- Known hypersensitivity to mammalian cell-derived products.
- Known hypersensitivity to Albumin (Human).
WARNINGS
Pediatrics
Risk in Premature Infants
The multidose preserved formulation contains benzyl alcohol. Benzyl alcohol has been reported to be associated with an increased incidence of neurological and other complications in premature infants which are sometimes fatal.
Adults
Increased Mortality, Serious Cardiovascular and Thromboembolic Events
Patients with chronic renal failure experienced greater risks for death and serious cardiovascular events when administered erythropoiesis-stimulating agents (ESAs) to target higher versus lower hemoglobin levels (13.5 vs.11.3 g/dL; 14 vs. 10 g/dL) in two clinical studies. Patients with chronic renal failure and an insufficient hemoglobin response to ESA therapy may be at even greater risk for cardiovascular events and mortality than other patients. EPOGEN® and other ESAs increased the risks for death and serious cardiovascular events in controlled clinical trials of patients with cancer. These events included myocardial infarction, stroke, congestive heart failure, and hemodialysis vascular access thrombosis. A rate of hemoglobin rise of > 1 g/dL over 2 weeks may contribute to these risks.
In a randomized prospective trial, 1432 anemic chronic renal failure patients who were not undergoing dialysis were assigned to Epoetin alfa (rHuEPO) treatment targeting a maintenance hemoglobin concentration of 13.5 g/dL or 11.3 g/dL. A major cardiovascular event (death, myocardial infarction, stroke, or hospitalization for congestive heart failure) occurred among 125 (18%) of the 715 patients in the higher hemoglobin group compared to 97 (14%) among the 717 patients in the lower hemoglobin group (HR 1.3, 95% CI: 1.0, 1.7, p = 0.03).40
Increased risk for serious cardiovascular events was also reported from a randomized, prospective trial of 1265 hemodialysis patients with clinically evident cardiac disease (ischemic heart disease or congestive heart failure). In this trial, patients were assigned to EPOGEN® treatment targeted to a maintenance hematocrit of either 42 ± 3% or 30 ± 3%.37 Increased mortality was observed in 634 patients randomized to a target hematocrit of 42% [221 deaths (35% mortality)] compared to 631 patients targeted to remain at a hematocrit of 30% [185 deaths (29% mortality)]. The reason for the increased mortality observed in this study is unknown, however, the incidence of non-fatal myocardial infarctions (3.1% vs. 2.3%), vascular access thromboses (39% vs. 29%), and all other thrombotic events (22% vs. 18%) were also higher in the group randomized to achieve a hematocrit of 42%. An increased incidence of thrombotic events has also been observed in patients with cancer treated with erythropoietic agents.
In a randomized controlled study (referred to as Cancer Study 1 - the ‘BEST’ study) with another ESA in 939 women with metastatic breast cancer receiving chemotherapy, patients received either weekly Epoetin alfa or placebo for up to a year. This study was designed to show that survival was superior when an ESA was administered to prevent anemia (maintain hemoglobin levels between 12 and 14 g/dL or hematocrit between 36% and 42%). The study was terminated prematurely when interim results demonstrated that a higher mortality at 4 months (8.7% vs. 3.4%) and a higher rate of fatal thrombotic events (1.1% vs. 0.2%) in the first 4 months of the study were observed among patients treated with Epoetin alfa. Based on Kaplan-Meier estimates, at the time of study termination, the 12-month survival was lower in the Epoetin alfa group than in the placebo group (70% vs. 76%; HR 1.37, 95% CI: 1.07, 1.75; p = 0.012).43
A systematic review of 57 randomized controlled trials (including Cancer Studies 1 and 5 - the 'BEST' and 'ENHANCE' studies) evaluating 9353 patients with cancer compared ESAs plus red blood cell transfusion with red blood cell transfusion alone for prophylaxis or treatment of anemia in cancer patients with or without concurrent antineoplastic therapy. An increased relative risk of thromboembolic events (RR 1.67, 95% CI: 1.35, 2.06; 35 trials and 6769 patients) was observed in ESA-treated patients. An overall survival hazard ratio of 1.08 (95% CI: 0.99, 1.18; 42 trials and 8167 patients) was observed in ESA-treated patients.41
An increased incidence of deep vein thrombosis (DVT) in patients receiving Epoetin alfa undergoing surgical orthopedic procedures has been observed (see ADVERSE REACTIONS, Surgery Patients: Thrombotic/Vascular Events). In a randomized controlled study (referred to as the ‘SPINE’ study), 681 adult patients, not receiving prophylactic anticoagulation and undergoing spinal surgery, received either 4 doses of 600 U/kg Epoetin alfa (7, 14, and 21 days before surgery, and the day of surgery) and standard of care (SOC) treatment, or SOC treatment alone. Preliminary analysis showed a higher incidence of DVT, determined by either Color Flow Duplex Imaging or by clinical symptoms, in the Epoetin alfa group [16 patients (4.7%)] compared to the SOC group [7 patients (2.1%)]. In addition, 12 patients in the Epoetin alfa group and 7 patients in the SOC group had other thrombotic vascular events. Deep venous thrombosis should be strongly considered when ESAs are used for the reduction of allogeneic RBC transfusions in surgical patients (see BOXED WARNINGS and DOSAGE AND ADMINISTRATION).
Increased mortality was also observed in a randomized placebo-controlled study of EPOGEN® in adult patients who were undergoing coronary artery bypass surgery (7 deaths in 126 patients randomized to EPOGEN® versus no deaths among 56 patients receiving placebo). Four of these deaths occurred during the period of study drug administration and all four deaths were associated with thrombotic events.42 ESAs are not approved for reduction of allogeneic red blood cell transfusions in patients scheduled for cardiac surgery.
Increased Mortality and/or Increased Risk of Tumor Progression or Recurrence
Erythropoiesis-stimulating agents resulted in decreased locoregional control/progression-free survival and/or overall survival (see Table 1). These findings were observed in studies of patients with advanced head and neck cancer receiving radiation therapy (Cancer Studies 5 and 6), in patients receiving chemotherapy for metastatic breast cancer (Cancer Study 1) or lymphoid malignancy (Cancer Study 2), and in patients with non-small cell lung cancer or various malignancies who were not receiving chemotherapy or radiotherapy (Cancer Studies 7 and 8).
| Study / Tumor / (n) | Hemoglobin Target |
Achieved Hemoglobin (Median Q1,Q3) | Primary Endpoint | Adverse Outcome for ESA-containing Arm |
| Chemotherapy | ||||
|
Cancer Study 1 Metastatic breast cancer (n=939) | 12-14 g/dL |
12.9 g/dL 12.2, 13.3 g/dL | 12-month overall survival |
Decreased 12-month survival |
|
Cancer Study 2 Lymphoid malignancy (n=344) |
13-15 g/dL (M) 13-14 g/dL (F) |
11.0 g/dL 9.8, 12.1 g/dL | Proportion of patients achieving a hemoglobin response |
Decreased overall survival |
|
Cancer Study 3 Early breast cancer (n=733) | 12.5-13 g/dL |
13.1 g/dL 12.5, 13.7 g/dL | Relapse-free and overall survival | Decreased 3 yr. relapse-free and overall survival |
|
Cancer Study 4 Cervical Cancer (n=114) | 12-14 g/dL |
12.7 g/dL 12.1, 13.3 g/dL | Progression-free and overall survival and locoregional control | Decreased 3 yr. progression-free and overall survival and locoregional control |
| Radiotherapy Alone | ||||
|
Cancer Study 5 Head and neck cancer (n=351) |
≥15 g/dL (M) ≥14 g/dL (F) | Not available | Locoregional progression-free survival |
Decreased 5-year locoregional progression-free survival Decreased overall survival |
|
Cancer Study 6 Head and neck cancer (n=522) | 14-15.5 g/dL | Not available | Locoregional disease control | Decreased locoregional disease control |
| No Chemotherapy or Radiotherapy | ||||
|
Cancer Study 7 Non-small cell lung cancer (n=70) | 12-14 g/dL | Not available | Quality of life | Decreased overall survival |
|
Cancer Study 8 Non-myeloid malignancy (n=989) | 12-13 g/dL |
10.6 g/dL 9.4, 11.8 g/dL | RBC transfusions |
Decreased overall survival |
Decreased overall survival:
Cancer Study 1 (the ‘BEST’ study) was previously described (see WARNINGS: Increased Mortality, Serious Cardiovascular and Thromboembolic Events). Mortality at 4 months (8.7% vs. 3.4%) was significantly higher in the Epoetin alfa arm. The most common investigator-attributed cause of death within the first 4 months was disease progression; 28 of 41 deaths in the Epoetin alfa arm and 13 of 16 deaths in the placebo arm were attributed to disease progression. Investigator assessed time to tumor progression was not different between the two groups. Survival at 12 months was significantly lower in the Epoetin alfa arm (70% vs. 76%, HR 1.37, 95% CI: 1.07, 1.75; p = 0.012).43
Cancer Study 2 was a Phase 3, double-blind, randomized (darbepoetin alfa vs. placebo) study conducted in 344 anemic patients with lymphoid malignancy receiving chemotherapy. With a median follow-up of 29 months, overall mortality rates were significantly higher among patients randomized to darbepoetin alfa as compared to placebo (HR 1.36, 95% CI: 1.02, 1.82).
Cancer Study 7 was a Phase 3, multicenter, randomized (Epoetin alfa vs. placebo), double-blind study, in which patients with advanced non-small cell lung cancer receiving only palliative radiotherapy or no active therapy were treated with Epoetin alfa to achieve and maintain hemoglobin levels between 12 and 14 g/dL. Following an interim analysis of 70 of 300 patients planned, a significant difference in survival in favor of the patients on the placebo arm of the trial was observed (median survival 63 vs. 129 days; HR 1.84; p = 0.04).
Cancer Study 8 was a Phase 3, double-blind, randomized (darbepoetin alfa vs. placebo), 16-week study in 989 anemic patients with active malignant disease, neither receiving nor planning to receive chemotherapy or radiation therapy. There was no evidence of a statistically significant reduction in proportion of patients receiving RBC transfusions. The median survival was shorter in the darbepoetin alfa treatment group (8 months) compared with the placebo group (10.8 months); HR 1.30, 95% CI: 1.07, 1.57.
Decreased progression-free survival and overall survival:
Cancer Study 3 (the ‘PREPARE’ study) was a randomized controlled study in which darbepoetin alfa was administered to prevent anemia conducted in 733 women receiving neo-adjuvant breast cancer treatment. After a median follow-up of approximately 3 years, the survival rate (86% vs. 90%, HR 1.42, 95% CI: 0.93, 2.18) and relapse-free survival rate (72% vs. 78%, HR 1.33, 95% CI: 0.99, 1.79) were lower in the darbepoetin alfa-treated arm compared to the control arm.
Cancer Study 4 (protocol GOG 191) was a randomized controlled study that enrolled 114 of a planned 460 cervical cancer patients receiving chemotherapy and radiotherapy. Patients were randomized to receive Epoetin alfa to maintain hemoglobin between 12 and 14 g/dL or to transfusion support as needed. The study was terminated prematurely due to an increase in thromboembolic events in Epoetin alfa-treated patients compared to control (19% vs. 9%). Both local recurrence (21% vs. 20%) and distant recurrence (12% vs. 7%) were more frequent in Epoetin alfa-treated patients compared to control. Progression-free survival at 3 years was lower in the Epoetin alfa-treated group compared to control (59% vs. 62%, HR 1.06, 95% CI: 0.58, 1.91). Overall survival at 3 years was lower in the Epoetin alfa-treated group compared to control (61% vs. 71%, HR 1.28, 95% CI: 0.68, 2.42).
Cancer Study 5 (the ‘ENHANCE’ study) was a randomized controlled study in 351 head and neck cancer patients where Epoetin beta or placebo was administered to achieve target hemoglobin of 14 and 15 g/dL for women and men, respectively. Locoregional progression-free survival was significantly shorter in patients receiving Epoetin beta (HR 1.62, 95% CI: 1.22, 2.14; p = 0.0008) with a median of 406 days Epoetin beta vs. 745 days placebo. Overall survival was significantly shorter in patients receiving Epoetin beta (HR 1.39, 95% CI: 1.05, 1.84; p = 0.02).38
Decreased locoregional control:
Cancer Study 6 (DAHANCA 10) was conducted in 522 patients with primary squamous cell carcinoma of the head and neck receiving radiation therapy randomized to darbepoetin alfa with radiotherapy or radiotherapy alone. An interim analysis on 484 patients demonstrated that locoregional control at 5 years was significantly shorter in patients receiving darbepoetin alfa (RR 1.44, 95% CI: 1.06, 1.96; p = 0.02). Overall survival was shorter in patients receiving darbepoetin alfa (RR 1.28, 95% CI: 0.98, 1.68; p = 0.08).
Pure Red Cell Aplasia
Cases of pure red cell aplasia (PRCA) and of severe anemia, with or without other cytopenias, associated with neutralizing antibodies to erythropoietin have been reported in patients treated with EPOGEN®. This has been reported predominantly in patients with CRF receiving EPOGEN® by subcutaneous administration. Any patient who develops a sudden loss of response to EPOGEN®, accompanied by severe anemia and low reticulocyte count, should be evaluated for the etiology of loss of effect, including the presence of neutralizing antibodies to erythropoietin (see PRECAUTIONS: Lack or Loss of Response). If anti-erythropoietin antibody-associated anemia is suspected, withhold EPOGEN® and other erythropoietic proteins. Contact Amgen (1-800-77AMGEN) to perform assays for binding and neutralizing antibodies. EPOGEN® should be permanently discontinued in patients with antibody-mediated anemia. Patients should not be switched to other erythropoietic proteins as antibodies may cross-react (see ADVERSE REACTIONS: Immunogenicity).
Albumin (Human)
EPOGEN® contains albumin, a derivative of human blood. Based on effective donor screening and product manufacturing processes, it carries an extremely remote risk for transmission of viral diseases. A theoretical risk for transmission of Creutzfeldt-Jakob disease (CJD) also is considered extremely remote. No cases of transmission of viral diseases or CJD have ever been identified for albumin.
Chronic Renal Failure Patients
Hypertension: Patients with uncontrolled hypertension should not be treated with EPOGEN®; blood pressure should be controlled adequately before initiation of therapy. Although there do not appear to be any direct pressor effects of EPOGEN®, blood pressure may rise during EPOGEN® therapy. During the early phase of treatment when the hematocrit is increasing, approximately 25% of patients on dialysis may require initiation of, or increases in, antihypertensive therapy. Hypertensive encephalopathy and seizures have been observed in patients with CRF treated with EPOGEN®.
Special care should be taken to closely monitor and aggressively control blood pressure in patients treated with EPOGEN®. Patients should be advised as to the importance of compliance with antihypertensive therapy and dietary restrictions. If blood pressure is difficult to control by initiation of appropriate measures, the hemoglobin may be reduced by decreasing or withholding the dose of EPOGEN®. A clinically significant decrease in hemoglobin may not be observed for several weeks.
It is recommended that the dose of EPOGEN® be decreased if the hemoglobin increase exceeds 1 g/dL in any 2-week period, because of the possible association of excessive rate of rise of hemoglobin with an exacerbation of hypertension. In CRF patients on hemodialysis with clinically evident ischemic heart disease or congestive heart failure, the dose of EPOGEN® should be carefully adjusted to achieve and maintain hemoglobin levels between 10-12 g/dL (see WARNINGS: Mortality, Serious Cardiovascular and Thromboembolic Events and DOSAGE AND ADMINISTRATION: Chronic Renal Failure Patients).
Seizures: Seizures have occurred in patients with CRF participating in EPOGEN® clinical trials.
In adult patients on dialysis, there was a higher incidence of seizures during the first 90 days of therapy (occurring in approximately 2.5% of patients) as compared with later timepoints.
Given the potential for an increased risk of seizures during the first 90 days of therapy, blood pressure and the presence of premonitory neurologic symptoms should be monitored closely. Patients should be cautioned to avoid potentially hazardous activities such as driving or operating heavy machinery during this period.
While the relationship between seizures and the rate of rise of hemoglobin is uncertain, it is recommended that the dose of EPOGEN® be decreased if the hemoglobin increase exceeds 1 g/dL in any 2-week period.
Thrombotic Events: During hemodialysis, patients treated with EPOGEN® may require increased anticoagulation with heparin to prevent clotting of the artificial kidney (see ADVERSE REACTIONS for more information about thrombotic events).
Other thrombotic events (eg, myocardial infarction, cerebrovascular accident, transient ischemic attack) have occurred in clinical trials at an annualized rate of less than 0.04 events per patient year of EPOGEN® therapy. These trials were conducted in adult patients with CRF (whether on dialysis or not) in whom the target hematocrit was 32% to 40%. However, the risk of thrombotic events, including vascular access thrombosis, was significantly increased in adult patients with ischemic heart disease or congestive heart failure receiving EPOGEN® therapy with the goal of reaching a normal hematocrit (42%) as compared to a target hematocrit of 30%. Patients with pre-existing cardiovascular disease should be monitored closely.
Zidovudine-treated HIV-infected Patients
In contrast to CRF patients, EPOGEN® therapy has not been linked to exacerbation of hypertension, seizures, and thrombotic events in HIV-infected patients. However, the clinical data do not rule out an increased risk for serious cardiovascular events.
PRECAUTIONS
The parenteral administration of any biologic product should be attended by appropriate precautions in case allergic or other untoward reactions occur (see CONTRAINDICATIONS). In clinical trials, while transient rashes were occasionally observed concurrently with EPOGEN® therapy, no serious allergic or anaphylactic reactions were reported (see ADVERSE REACTIONS for more information regarding allergic reactions).
The safety and efficacy of EPOGEN® therapy have not been established in patients with a known history of a seizure disorder or underlying hematologic disease (eg, sickle cell anemia, myelodysplastic syndromes, or hypercoagulable disorders).
In some female patients, menses have resumed following EPOGEN® therapy; the possibility of pregnancy should be discussed and the need for contraception evaluated.
Hematology
Exacerbation of porphyria has been observed rarely in patients with CRF treated with EPOGEN®. However, EPOGEN® has not caused increased urinary excretion of porphyrin metabolites in normal volunteers, even in the presence of a rapid erythropoietic response. Nevertheless, EPOGEN® should be used with caution in patients with known porphyria.
In preclinical studies in dogs and rats, but not in monkeys, EPOGEN® therapy was associated with subclinical bone marrow fibrosis. Bone marrow fibrosis is a known complication of CRF in humans and may be related to secondary hyperparathyroidism or unknown factors. The incidence of bone marrow fibrosis was not increased in a study of adult patients on dialysis who were treated with EPOGEN® for 12 to 19 months, compared to the incidence of bone marrow fibrosis in a matched group of patients who had not been treated with EPOGEN®.
Hemoglobin in CRF patients should be measured twice a week; zidovudine-treated HIV-infected and cancer patients should have hemoglobin measured once a week until hemoglobin has been stabilized, and measured periodically thereafter.
Lack or Loss of Response
If the patient fails to respond or to maintain a response to doses within the recommended dosing range, the following etiologies should be considered and evaluated:
- Iron deficiency: Virtually all patients will eventually require supplemental iron therapy (see IRON EVALUATION).
- Underlying infectious, inflammatory, or malignant processes.
- Occult blood loss.
- Underlying hematologic diseases (ie, thalassemia, refractory anemia, or other myelodysplastic disorders).
- Vitamin deficiencies: Folic acid or vitamin B12.
- Hemolysis.
- Aluminum intoxication.
- Osteitis fibrosa cystica.
- Pure Red Cell Aplasia (PRCA) or anti-erythropoietin antibody-associated anemia: In the absence of another etiology, the patient should be evaluated for evidence of PRCA and sera should be tested for the presence of antibodies to erythropoietin (see WARNINGS: Pure Red Cell Aplasia).
See DOSAGE AND ADMINISTRATION: Chronic Renal Failure Patients for management of patients with an insufficient hemoglobin response to EPOGEN® therapy.
Iron Evaluation
During EPOGEN® therapy, absolute or functional iron deficiency may develop. Functional iron deficiency, with normal ferritin levels but low transferrin saturation, is presumably due to the inability to mobilize iron stores rapidly enough to support increased erythropoiesis. Transferrin saturation should be at least 20% and ferritin should be at least 100 ng/mL.
Prior to and during EPOGEN® therapy, the patient’s iron status, including transferrin saturation (serum iron divided by iron binding capacity) and serum ferritin, should be evaluated. Virtually all patients will eventually require supplemental iron to increase or maintain transferrin saturation to levels which will adequately support erythropoiesis stimulated by EPOGEN®. All surgery patients being treated with EPOGEN® should receive adequate iron supplementation throughout the course of therapy in order to support erythropoiesis and avoid depletion of iron stores.
Drug Interaction
No evidence of interaction of EPOGEN® with other drugs was observed in the course of clinical trials.
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Carcinogenic potential of EPOGEN® has not been evaluated. EPOGEN® does not induce bacterial gene mutation (Ames Test), chromosomal aberrations in mammalian cells, micronuclei in mice, or gene mutation at the HGPRT locus. In female rats treated IV with EPOGEN®, there was a trend for slightly increased fetal wastage at doses of 100 and 500 Units/kg.
Pregnancy Category C
EPOGEN® has been shown to have adverse effects in rats when given in doses 5 times the human dose. There are no adequate and well-controlled studies in pregnant women. EPOGEN® should be used during pregnancy only if potential benefit justifies the potential risk to the fetus.
In studies in female rats, there were decreases in body weight gain, delays in appearance of abdominal hair, delayed eyelid opening, delayed ossification, and decreases in the number of caudal vertebrae in the F1 fetuses of the 500 Units/kg group. In female rats treated IV, there was a trend for slightly increased fetal wastage at doses of 100 and 500 Units/kg. EPOGEN® has not shown any adverse effect at doses as high as 500 Units/kg in pregnant rabbits (from day 6 to 18 of gestation).
Nursing Mothers
Postnatal observations of the live offspring (F1 generation) of female rats treated with EPOGEN® during gestation and lactation revealed no effect of EPOGEN® at doses of up to 500 Units/kg. There were, however, decreases in body weight gain, delays in appearance of abdominal hair, eyelid opening, and decreases in the number of caudal vertebrae in the F1 fetuses of the 500 Units/kg group. There were no EPOGEN®-related effects on the F2 generation fetuses.
It is not known whether EPOGEN® is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when EPOGEN® is administered to a nursing woman.
Pediatric Use
See WARNINGS: Pediatrics.
Pediatric Patients on Dialysis: EPOGEN® is indicated in infants (1 month to 2 years), children (2 years to 12 years), and adolescents (12 years to 16 years) for the treatment of anemia associated with CRF requiring dialysis. Safety and effectiveness in pediatric patients less than 1 month old have not been established (see CLINICAL EXPERIENCE: CHRONIC RENAL FAILURE, PEDIATRIC PATIENTS ON DIALYSIS). The safety data from these studies show that there is no increased risk to pediatric CRF patients on dialysis when compared to the safety profile of EPOGEN® in adult CRF patients (see ADVERSE REACTIONS and WARNINGS). Published literature27-30 provides supportive evidence of the safety and effectiveness of EPOGEN® in pediatric CRF patients on dialysis.
Pediatric Patients Not Requiring Dialysis: Published literature30,31 has reported the use of EPOGEN® in 133 pediatric patients with anemia associated with CRF not requiring dialysis, ages 3 months to 20 years‚ treated with 50 to 250 Units/kg SC or IV‚ QW to TIW. Dose-dependent increases in hemoglobin and hematocrit were observed with reductions in transfusion requirements.
Pediatric HIV-infected Patients: Published literature 32,33 has reported the use of EPOGEN® in 20 zidovudine-treated anemic HIV-infected pediatric patients ages 8 months to 17 years‚ treated with 50 to 400 Units/kg SC or IV‚ 2 to 3 times per week. Increases in hemoglobin levels and in reticulocyte counts‚ and decreases in or elimination of blood transfusions were observed.
Pediatric Cancer Patients on Chemotherapy: The safety and effectiveness of EPOGEN® were evaluated in a randomized, double-blind, placebo-controlled, multicenter study (see CLINICAL EXPERIENCE, Weekly (QW) Dosing, Pediatric Patients).
Geriatric Use
Among 1051 patients enrolled in the 5 clinical trials of EPOGEN® for reduction of allogeneic blood transfusions in patients undergoing elective surgery 745 received EPOGEN® and 306 received placebo. Of the 745 patients who received EPOGEN®, 432 (58%) were aged 65 and over, while 175 (23%) were 75 and over. No overall differences in safety or effectiveness were observed between geriatric and younger patients. The dose requirements for EPOGEN® in geriatric and younger patients within the 4 trials using the TIW schedule were similar. Insufficient numbers of patients were enrolled in the study using the weekly dosing regimen to determine whether the dosing requirements differ for this schedule.
Of the 882 patients enrolled in the 3 studies of chronic renal failure patients on dialysis, 757 received EPOGEN® and 125 received placebo. Of the 757 patients who received EPOGEN®, 361 (47%) were aged 65 and over, while 100 (13%) were 75 and over. No differences in safety or effectiveness were observed between geriatric and younger patients. Dose selection and adjustment for an elderly patient should be individualized to achieve and maintain the target hematocrit (See DOSAGE AND ADMINISTRATION).
Insufficient numbers of patients age 65 or older were enrolled in clinical studies of EPOGEN® for the treatment of anemia associated with pre-dialysis chronic renal failure, cancer chemotherapy, and Zidovudine-treatment of HIV infection to determine whether they respond differently from younger subjects.
Information for Patients
Patients should be informed of the increased risks of mortality, serious cardiovascular events, thromboembolic events, and increased risk of tumor progression or recurrence (see WARNINGS). In those situations in which the physician determines that a patient or their caregiver can safely and effectively administer EPOGEN® at home, instruction as to the proper dosage and administration should be provided. Patients should be instructed to read the EPOGEN® Medication Guide and Patient Instructions for Use and should be informed that the Medication Guide is not a disclosure of all possible side effects. Patients should be informed of the possible side effects of EPOGEN® and of the signs and symptoms of allergic drug reaction and advised of appropriate actions. If home use is prescribed for a patient, the patient should be thoroughly instructed in the importance of proper disposal and cautioned against the reuse of needles, syringes, or drug product. A puncture-resistant container should be available for the disposal of used syringes and needles, and guidance provided on disposal of the full container.
Chronic Renal Failure Patients
Patients with CRF Not Requiring Dialysis
Blood pressure and hemoglobin should be monitored no less frequently than for patients maintained on dialysis. Renal function and fluid and electrolyte balance should be closely monitored.
Hematology
Sufficient time should be allowed to determine a patient’s responsiveness to a dosage of EPOGEN® before adjusting the dose. Because of the time required for erythropoiesis and the red cell half-life, an interval of 2 to 6 weeks may occur between the time of a dose adjustment (initiation, increase, decrease, or discontinuation) and a significant change in hemoglobin.
For patients who respond to EPOGEN® with a rapid increase in hemoglobin (eg, more than 1 g/dL in any 2-week period), the dose of EPOGEN® should be reduced because of the possible association of excessive rate of rise of hemoglobin with an exacerbation of hypertension.
The elevated bleeding time characteristic of CRF decreases toward normal after correction of anemia in adult patients treated with EPOGEN®. Reduction of bleeding time also occurs after correction of anemia by transfusion.
Laboratory Monitoring
The hemoglobin should be determined twice a week until it has stabilized in the suggested hemoglobin range and the maintenance dose has been established. After any dose adjustment, the hemoglobin should also be determined twice weekly for at least 2 to 6 weeks until it has been determined that the hemoglobin has stabilized in response to the dose change. The hemoglobin should then be monitored at regular intervals.
A complete blood count with differential and platelet count should be performed regularly. During clinical trials, modest increases were seen in platelets and white blood cell counts. While these changes were statistically significant, they were not clinically significant and the values remained within normal ranges.
In patients with CRF, serum chemistry values (including blood urea nitrogen [BUN], uric acid, creatinine, phosphorus, and potassium) should be monitored regularly. During clinical trials in adult patients on dialysis, modest increases were seen in BUN, creatinine, phosphorus, and potassium. In some adult patients with CRF not on dialysis treated with EPOGEN®, modest increases in serum uric acid and phosphorus were observed. While changes were statistically significant, the values remained within the ranges normally seen in patients with CRF.
Diet
The importance of compliance with dietary and dialysis prescriptions should be reinforced. In particular, hyperkalemia is not uncommon in patients with CRF. In US studies in patients on dialysis, hyperkalemia has occurred at an annualized rate of approximately 0.11 episodes per patient-year of EPOGEN® therapy, often in association with poor compliance to medication, diet, and/or dialysis.
Dialysis Management
Therapy with EPOGEN® results in an increase in hematocrit and a decrease in plasma volume which could affect dialysis efficiency. In studies to date, the resulting increase in hematocrit did not appear to adversely affect dialyzer function8,9 or the efficiency of high flux hemodialysis.10 During hemodialysis, patients treated with EPOGEN® may require increased anticoagulation with heparin to prevent clotting of the artificial kidney.
Patients who are marginally dialyzed may require adjustments in their dialysis prescription. As with all patients on dialysis, the serum chemistry values (including BUN, creatinine, phosphorus, and potassium) in patients treated with EPOGEN® should be monitored regularly to assure the adequacy of the dialysis prescription.
Renal Function
In adult patients with CRF not on dialysis, renal function and fluid and electrolyte balance should be closely monitored. In patients with CRF not on dialysis, placebo-controlled studies of progression of renal dysfunction over periods of greater than 1 year have not been completed. In shorter term trials in adult patients with CRF not on dialysis, changes in creatinine and creatinine clearance were not significantly different in patients treated with EPOGEN® compared with placebo-treated patients. Analysis of the slope of 1/serum creatinine versus time plots in these patients indicates no significant change in the slope after the initiation of EPOGEN® therapy.
Zidovudine-treated HIV-infected Patients
Hypertension
Exacerbation of hypertension has not been observed in zidovudine-treated HIV-infected patients treated with EPOGEN®. However, EPOGEN® should be withheld in these patients if pre-existing hypertension is uncontrolled, and should not be started until blood pressure is controlled. In double-blind studies, a single seizure has been experienced by a patient treated with EPOGEN®.23
Cancer Patients on Chemotherapy
Hypertension
Hypertension, associated with a significant increase in hemoglobin, has been noted rarely in patients treated with EPOGEN®. Nevertheless, blood pressure in patients treated with EPOGEN® should be monitored carefully, particularly in patients with an underlying history of hypertension or cardiovascular disease.
Seizures
In double-blind, placebo-controlled trials, 3.2% (n = 2/63) of patients treated with EPOGEN® TIW and 2.9% (n = 2/68) of placebo-treated patients had seizures. Seizures in 1.6% (n = 1/63) of patients treated with EPOGEN® TIW occurred in the context of a significant increase in blood pressure and hematocrit from baseline values. However, both patients treated with EPOGEN® also had underlying CNS pathology which may have been related to seizure activity.
In a placebo-controlled, double-blind trial utilizing weekly dosing with EPOGEN®, 1.2% (n = 2/168) of safety-evaluable patients treated with EPOGEN® and 1% (n = 1/165) of placebo-treated patients had seizures. Seizures in the patients treated with weekly EPOGEN® occurred in the context of a significant increase in hemoglobin from baseline values however significant increases in blood pressure were not seen. These patients may have had other CNS pathology.
Thrombotic Events
In double-blind, placebo-controlled trials, 3.2% (n = 2/63) of patients treated with EPOGEN® TIW and 11.8% (n = 8/68) of placebo-treated patients had thrombotic events (eg, pulmonary embolism, cerebrovascular accident), (see WARNINGS: Increased Mortality, Serious Cardiovascular and Thromboembolic Events).
In a placebo-controlled, double-blind trial utilizing weekly dosing with EPOGEN®, 6.0% (n = 10/168) of safety-evaluable patients treated with EPOGEN® and 3.6% (n = 6/165) (p = 0.444) of placebo-treated patients had clinically significant thrombotic events (deep vein thrombosis requiring anticoagulant therapy, embolic event including pulmonary embolism, myocardial infarction, cerebral ischemia, left ventricular failure and thrombotic microangiopathy). A definitive relationship between the rate of hemoglobin increase and the occurrence of clinically significant thrombotic events could not be evaluated due to the limited schedule of hemoglobin measurements in this study.
The safety and efficacy of EPOGEN®were evaluated in a randomized, double-blind, placebo-controlled, multicenter study that enrolled 222 anemic patients ages 5 to 18 receiving treatment for a variety of childhood malignancies. Due to the study design (small sample size and the heterogeneity of the underlying malignancies and of anti-neoplastic treatments employed), a determination of the effect of EPOGEN® on the incidence of thrombotic events could not be performed. In the EPOGEN® arm, the overall incidence of thrombotic events was 10.8% and the incidence of serious or life-threatening events was 7.2%.
Surgery Patients
Hypertension
Blood pressure may rise in the perioperative period in patients being treated with EPOGEN®. Therefore, blood pressure should be monitored carefully.
ADVERSE REACTIONS
Immunogenicity
As with all therapeutic proteins, there is the potential for immunogenicity. Neutralizing antibodies to erythropoietin, in association with PRCA or severe anemia (with or without other cytopenias), have been reported in patients receiving EPOGEN® (see WARNINGS: Pure Red Cell Aplasia) during post-marketing experience.
There has been no systematic assessment of immune responses, i.e., the incidence of either binding or neutralizing antibodies to EPOGEN®, in controlled clinical trials.
Where reported, the incidence of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies across products within this class (erythropoietic proteins) may be misleading.
Chronic Renal Failure Patients
In double-blind, placebo-controlled studies involving over 300 patients with CRF, the events reported in greater than 5% of patients treated with EPOGEN® during the blinded phase were:
| Event | Patients Treated With EPOGEN® | Placebo-treated Patients |
| (n = 200) | (n = 135) | |
| Hypertension | 24% | 19% |
| Headache | 16% | 12% |
| Arthralgias | 11% | 6% |
| Nausea | 11% | 9% |
| Edema | 9% | 10% |
| Fatigue | 9% | 14% |
| Diarrhea | 9% | 6% |
| Vomiting | 8% | 5% |
| Chest Pain | 7% | 9% |
| Skin Reaction | 7% | 12% |
| (Administration Site) | ||
| Asthenia | 7% | 12% |
| Dizziness | 7% | 13% |
| Clotted Access | 7% | 2% |
Significant adverse events of concern in patients with CRF treated in double-blind, placebo-controlled trials occurred in the following percent of patients during the blinded phase of the studies:
| Seizure | 1.1% | 1.1% |
| CVA/TIA | 0.4% | 0.6% |
| MI | 0.4% | 1.1% |
| Death | 0% | 1.7% |
In the US EPOGEN® studies in adult patients on dialysis (over 567 patients), the incidence (number of events per patient-year) of the most frequently reported adverse events were: hypertension (0.75), headache (0.40), tachycardia (0.31), nausea/vomiting (0.26), clotted vascular access (0.25), shortness of breath (0.14), hyperkalemia (0.11), and diarrhea (0.11). Other reported events occurred at a rate of less than 0.10 events per patient per year.
Events reported to have occurred within several hours of administration of EPOGEN® were rare, mild, and transient, and included injection site stinging in dialysis patients and flu-like symptoms such as arthralgias and myalgias.
In all studies analyzed to date, EPOGEN® administration was generally well-tolerated, irrespective of the route of administration.
Pediatric CRF Patients: In pediatric patients with CRF on dialysis, the pattern of most adverse events was similar to that found in adults. Additional adverse events reported during the double-blind phase in >10% of pediatric patients in either treatment group were: abdominal pain, dialysis access complications including access infections and peritonitis in those receiving peritoneal dialysis, fever, upper respiratory infection, cough, pharyngitis, and constipation. The rates are similar between the treatment groups for each event.
Hypertension: Increases in blood pressure have been reported in clinical trials, often during the first 90 days of therapy. On occasion, hypertensive encephalopathy and seizures have been observed in patients with CRF treated with EPOGEN®. When data from all patients in the US phase 3 multicenter trial were analyzed, there was an apparent trend of more reports of hypertensive adverse events in patients on dialysis with a faster rate of rise of hematocrit (greater than 4 hematocrit points in any 2-week period). However, in a double-blind, placebo-controlled trial, hypertensive adverse events were not reported at an increased rate in the group treated with EPOGEN® (150 Units/kg TIW) relative to the placebo group.
Seizures: There have been 47 seizures in 1010 patients on dialysis treated with EPOGEN® in clinical trials, with an exposure of 986 patient-years for a rate of approximately 0.048 events per patient-year. However, there appeared to be a higher rate of seizures during the first 90 days of therapy (occurring in approximately 2.5% of patients) when compared to subsequent 90-day periods. The baseline incidence of seizures in the untreated dialysis population is difficult to determine; it appears to be in the range of 5% to 10% per patient-year.34-36
Thrombotic Events: In clinical trials where the maintenance hematocrit was 35 ± 3% on EPOGEN®, clotting of the vascular access (A-V shunt) has occurred at an annualized rate of about 0.25 events per patient-year, and other thrombotic events (eg, myocardial infarction, cerebral vascular accident, transient ischemic attack, and pulmonary embolism) occurred at a rate of 0.04 events per patient-year. In a separate study of 1111 untreated dialysis patients, clotting of the vascular access occurred at a rate of 0.50 events per patient-year. However, in CRF patients on hemodialysis who also had clinically evident ischemic heart disease or congestive heart failure, the risk of A-V shunt thrombosis was higher (39% vs 29%, p < 0.001), and myocardial infarctions, vascular ischemic events, and venous thrombosis were increased, in patients targeted to a hematocrit of 42 ± 3% compared to those maintained at 30 ± 3% (see WARNINGS).
In patients treated with commercial EPOGEN®, there have been rare reports of serious or unusual thromboembolic events including migratory thrombophlebitis, microvascular thrombosis, pulmonary embolus, and thrombosis of the retinal artery, and temporal and renal veins. A causal relationship has not been established.
Allergic Reactions: There have been no reports of serious allergic reactions or anaphylaxis associated with EPOGEN® administration during clinical trials. Skin rashes and urticaria have been observed rarely and when reported have generally been mild and transient in nature.
There have been rare reports of potentially serious allergic reactions including urticaria with associated respiratory symptoms or circumoral edema, or urticaria alone. Most reactions occurred in situations where a causal relationship could not be established. Symptoms recurred with rechallenge in a few instances, suggesting that allergic reactivity may occasionally be associated with EPOGEN® therapy. If an anaphylactoid reaction occurs, EPOGEN® should be immediately discontinued and appropriate therapy initiated.
Zidovudine-treated HIV-infected Patients
In double-blind, placebo-controlled studies of 3 months duration involving approximately 300 zidovudine-treated HIV-infected patients, adverse events with an incidence of ≥ 10% in either patients treated with EPOGEN® or placebo-treated patients were:
| Event | Patients Treated With EPOGEN® (n = 144) | Placebo-treated Patients (n = 153) |
| Pyrexia | 38% | 29% |
| Fatigue | 25% | 31% |
| Headache | 19% | 14% |
| Cough | 18% | 14% |
| Diarrhea | 16% | 18% |
| Rash | 16% | 8% |
| Congestion, Respiratory | 15% | 10% |
| Nausea | 15% | 12% |
| Shortness of Breath | 14% | 13% |
| Asthenia | 11% | 14% |
| Skin Reaction, Medication Site | 10% | 7% |
| Dizziness | 9% | 10% |
In the 297 patients studied, EPOGEN® was not associated with significant increases in opportunistic infections or mortality.23 In 71 patients from this group treated with EPOGEN® at 150 Units/kg TIW, serum p24 antigen levels did not appear to increase.25 Preliminary data showed no enhancement of HIV replication in infected cell lines in vitro.23
Peripheral white blood cell and platelet counts are unchanged following EPOGEN® therapy.
Allergic Reactions: Two zidovudine-treated HIV-infected patients had urticarial reactions within 48 hours of their first exposure to study medication. One patient was treated with EPOGEN® and one was treated with placebo (EPOGEN® vehicle alone). Both patients had positive immediate skin tests against their study medication with a negative saline control. The basis for this apparent pre-existing hypersensitivity to components of the EPOGEN® formulation is unknown, but may be related to HIV-induced immunosuppression or prior exposure to blood products.
Seizures: In double-blind and open-label trials of EPOGEN® in zidovudine-treated HIV-infected patients, 10 patients have experienced seizures.23 In general, these seizures appear to be related to underlying pathology such as meningitis or cerebral neoplasms, not EPOGEN® therapy.
Cancer Patients on Chemotherapy
In double-blind, placebo-controlled studies of up to 3 months duration involving 131 cancer patients, adverse events with an incidence > 10% in either patients treated with EPOGEN® or placebo-treated patients were as indicated below:
| Event | Patients Treated With EPOGEN® (n=63) | Placebo-treated Patients (n = 68) |
|
||
| Pyrexia | 29% | 19% |
| Diarrhea | 21%* | 7% |
| Nausea | 17%* | 32% |
| Vomiting | 17% | 15% |
| Edema | 17%* | 1% |
| Asthenia | 13% | 16% |
| Fatigue | 13% | 15% |
| Shortness of Breath | 13% | 9% |
| Parasthesia | 11% | 6% |
| Upper Respiratory Infection | 11% | 4% |
| Dizziness | 5% | 12% |
| Trunk Pain | 3%* | 16% |
Although some statistically significant differences between patients being treated with EPOGEN® and placebo-treated patients were noted, the overall safety profile of EPOGEN® appeared to be consistent with the disease process of advanced cancer. During double-blind and subsequent open-label therapy in which patients (n = 72 for total exposure to EPOGEN®) were treated for up to 32 weeks with doses as high as 927 Units/kg, the adverse experience profile of EPOGEN® was consistent with the progression of advanced cancer.
Three hundred thirty-three (333) cancer patients enrolled in a placebo-controlled double-blind trial utilizing Weekly dosing with EPOGEN® for up to 4 months were evaluable for adverse events. The incidence of adverse events was similar in both the treatment and placebo arms.
Surgery Patients
Adverse events with an incidence of ≥ 10% are shown in the following table:
| Event | Patients Treated With EPOGEN® 300 U/kg (n = 112)* | Patients Treated With EPOGEN® 100 U/kg (n = 101)* | Placebo-treated Patients (n = 103) * | Patients Treated With EPOGEN® 600 U/kg (n = 73)† | Patients Treated With EPOGEN® 300 U/kg (n = 72)† |
| Pyrexia | 51% | 50% | 60% | 47% | 42% |
| Nausea | 48% | 43% | 45% | 45% | 58% |
| Constipation | 43% | 42% | 43% | 51% | 53% |
| Skin Reaction, Medication Site | 25% | 19% | 22% | 26% | 29% |
| Vomiting | 22% | 12% | 14% | 21% | 29% |
| Skin Pain | 18% | 18% | 17% | 5% | 4% |
| Pruritus | 16% | 16% | 14% | 14% | 22% |
| Insomnia | 13% | 16% | 13% | 21% | 18% |
| Headache | 13% | 11% | 9% | 10% | 19% |
| Dizziness | 12% | 9% | 12% | 11% | 21% |
| Urinary Tract Infection | 12% | 3% | 11% | 11% | 8% |
| Hypertension | 10% | 11% | 10% | 5% | 10% |
| Diarrhea | 10% | 7% | 12% | 10% | 6% |
| Deep Venous Thrombosis | 10% | 3% | 5% | 0%‡ | 0%‡ |
| Dyspepsia | 9% | 11% | 6% | 7% | 8% |
| Anxiety | 7% | 2% | 11% | 11% | 4% |
| Edema | 6% | 11% | 8% | 11% | 7% |
Thrombotic/Vascular Events: In three double-blind, placebo-controlled orthopedic surgery studies, the rate of deep venous thrombosis (DVT) was similar among Epoetin alfa and placebo-treated patients in the recommended population of patients with a pretreatment hemoglobin of > 10 g/dL to ≤ 13 g/dL.17,19,26 However, in 2 of 3 orthopedic surgery studies the overall rate (all pretreatment hemoglobin groups combined) of DVTs detected by postoperative ultrasonography and/or surveillance venography was higher in the group treated with Epoetin alfa than in the placebo-treated group (11% vs. 6%). This finding was attributable to the difference in DVT rates observed in the subgroup of patients with pretreatment hemoglobin > 13 g/dL.
In the orthopedic surgery study of patients with pretreatment hemoglobin of > 10 g/dL to ≤ 13 g/dL which compared two dosing regimens (600 Units/kg weekly x 4 and 300 Units/kg daily x 15), 4 subjects in the 600 Units/kg weekly EPOGEN® group (5%) and no subjects in the 300 Units/kg daily group had a thrombotic vascular event during the study period.18
In a study examining the use of Epoetin alfa in 182 patients scheduled for coronary artery bypass graft surgery, 23% of patients treated with Epoetin alfa and 29% treated with placebo experienced thrombotic/vascular events. There were 4 deaths among the Epoetin alfa-treated patients that were associated with a thrombotic/vascular event (see WARNINGS).
OVERDOSAGE
The expected manifestations of EPOGEN® overdosage include signs and symptoms associated with an excessive and/or rapid increase in hemoglobin concentration, including any of the cardiovascular events described in WARNINGS and listed in ADVERSE REACTIONS. Patients receiving an overdosage of EPOGEN® should be monitored closely for cardiovascular events and hematologic abnormalities. Polycythemia should be managed acutely with phlebotomy as clinically indicated. Following resolution of the effects due to EPOGEN® overdosage, reintroduction of EPOGEN® therapy should be accompanied by close monitoring for evidence of rapid increases in hemoglobin concentration (>1 gm/dL per 14 days). In patients with an excessive hematopoietic response, reduce the EPOGEN® dose in accordance with the recommendations described in DOSAGE AND ADMINISTRATION.
DOSAGE AND ADMINISTRATION
IMPORTANT: See BOXED WARNINGS and WARNINGS: Increased Mortality, Serious Cardiovascular and Thromboembolic Events.
Chronic Renal Failure Patients
The recommended range for the starting dose of EPOGEN® is 50 to 100 Units/kg TIW for adult patients. The recommended starting dose for pediatric CRF patients on dialysis is 50 Units/kg TIW. Individualize dosing to achieve and maintain hemoglobin levels between 10-12 g/dL. The dose of EPOGEN® should be reduced as the hemoglobin approaches 12 g/dL or increases by more than 1 g/dL in any 2-week period. If hemoglobin excursions outside the recommended range occur, the EPOGEN® dose should be adjusted as described below.
EPOGEN® may be given either as an IV or SC injection. In patients on hemodialysis, the IV route is recommended (see WARNINGS: Pure Red Cell Aplasia). While the administration of EPOGEN® is independent of the dialysis procedure, EPOGEN® may be administered into the venous line at the end of the dialysis procedure to obviate the need for additional venous access. In adult patients with CRF not on dialysis, EPOGEN® may be given either as an IV or SC injection.
Patients who have been judged competent by their physicians to self-administer EPOGEN® without medical or other supervision may give themselves either an IV or SC injection. The table below provides general therapeutic guidelines for patients with CRF:
Individually titrate to achieve and maintain hemoglobin levels between 10 to 12 g/dL.
| Starting Dose: | |
| Adults | 50 to 100 Units/kg TIW; IV or SC |
| Pediatric Patients | 50 Units/kg TIW; IV or SC |
| Increase Dose by 25% If: | 1. Hemoglobin is < 10 g/dL and has not increased by 1 g/dL after 4 weeks of therapy or 2. Hemoglobin decreases below 10 g/dL |
| Reduce Dose by 25% When: | 1. Hemoglobin approaches 12 g/dL or, 2. Hemoglobin increases > 1 g/dL in any 2-week period |
During therapy, hematological parameters should be monitored regularly. Doses must be individualized to ensure that hemoglobin is maintained at an appropriate level for each patient.
For patients whose hemoglobin does not attain a level within the range of 10 to 12 g/dL despite the use of appropriate EPOGEN® dose titrations over a 12-week period:
- do not administer higher EPOGEN® doses and use the lowest dose that will maintain a hemoglobin level sufficient to avoid the need for recurrent RBC transfusions,
- evaluate and treat for other causes of anemia (see PRECAUTIONS: Lack or Loss of Response), and
- thereafter, hemoglobin should continue to be monitored and if responsiveness improves, EPOGEN® dose adjustments should be made as described above; discontinue EPOGEN® if responsiveness does not improve and the patient needs recurrent RBC transfusions.
Pretherapy Iron Evaluation: Prior to and during EPOGEN® therapy, the patient’s iron stores, including transferrin saturation (serum iron divided by iron binding capacity) and serum ferritin, should be evaluated. Transferrin saturation should be at least 20%, and ferritin should be at least 100 ng/mL. Virtually all patients will eventually require supplemental iron to increase or maintain transferrin saturation to levels that will adequately support erythropoiesis stimulated by EPOGEN®.
Dose Adjustment: The dose should be adjusted for each patient to achieve and maintain hemoglobin levels between 10 to 12 g/dL.
Increases in dose should not be made more frequently than once a month. If the hemoglobin is increasing and approaching 12 g/dL, the dose should be reduced by approximately 25%. If the hemoglobin continues to increase, dose should be temporarily withheld until the hemoglobin begins to decrease, at which point therapy should be reinitiated at a dose approximately 25% below the previous dose. If the hemoglobin increases by more than 1 g/dL in a 2-week period, the dose should be decreased by approximately 25%.
If the increase in the hemoglobin is less than 1 g/dL over 4 weeks and iron stores are adequate (see PRECAUTIONS: Laboratory Monitoring), the dose of EPOGEN® may be increased by approximately 25% of the previous dose. Further increases may be made at 4-week intervals until the specified hemoglobin is obtained.
Maintenance Dose: The maintenance dose must be individualized for each patient on dialysis. In the US phase 3 multicenter trial in patients on hemodialysis, the median maintenance dose was 75 Units/kg TIW, with a range from 12.5 to 525 Units/kg TIW. Almost 10% of the patients required a dose of 25 Units/kg, or less, and approximately 10% of the patients required more than 200 Units/kg TIW to maintain their hematocrit in the suggested target range. In pediatric hemodialysis and peritoneal dialysis patients, the median maintenance dose was 167 Units/kg/week (49 to 447 Units/kg per week) and 76 Units/kg per week (24 to 323 Units/kg/week) administered in divided doses (TIW or BIW), respectively to achieve the target range of 30% to 36%.
If the transferrin saturation is greater than 20%, the dose of EPOGEN® may be increased. Such dose increases should not be made more frequently than once a month, unless clinically indicated, as the response time of the hemoglobin to a dose increase can be 2 to 6 weeks. Hemoglobin should be measured twice weekly for 2 to 6 weeks following dose increases. In adult patients with CRF not on dialysis, the dose should also be individualized to maintain hemoglobin levels between 10 to 12 g/dL. EPOGEN® doses of 75 to 150 Units/kg/week have been shown to maintain hematocrits of 36% to 38% for up to 6 months.
Lack or Loss of Response: If a patient fails to respond or maintain a response, an evaluation for causative factors should be undertaken (see WARNINGS: Pure Red Cell Aplasia, PRECAUTIONS: Lack or Loss of Response, and PRECAUTIONS: Iron Evaluation). If the transferrin saturation is less than 20%, supplemental iron should be administered.
Zidovudine-treated HIV-infected Patients
Prior to beginning EPOGEN®, it is recommended that the endogenous serum erythropoietin level be determined (prior to transfusion). Available evidence suggests that patients receiving zidovudine with endogenous serum erythropoietin levels > 500 mUnits/mL are unlikely to respond to therapy with EPOGEN®.
In zidovudine-treated HIV-infected patients the dosage of EPOGEN® should be titrated for each patient to achieve and maintain the lowest hemoglobin level sufficient to avoid the need for blood transfusion and not to exceed the upper safety limit of 12 g/dL.
Starting Dose: For adult patients with serum erythropoietin levels ≤ 500 mUnits/mL who are receiving a dose of zidovudine ≤ 4200 mg/week, the recommended starting dose of EPOGEN® is 100 Units/kg as an IV or SC injection TIW for 8 weeks. For pediatric patients, see PRECAUTIONS: PEDIATRIC USE.
Increase Dose: During the dose adjustment phase of therapy, the hemoglobin should be monitored weekly. If the response is not satisfactory in terms of reducing transfusion requirements or increasing hemoglobin after 8 weeks of therapy, the dose of EPOGEN® can be increased by 50 to 100 Units/kg TIW. Response should be evaluated every 4 to 8 weeks thereafter and the dose adjusted accordingly by 50 to 100 Units/kg increments TIW. If patients have not responded satisfactorily to an EPOGEN® dose of 300 Units/kg TIW, it is unlikely that they will respond to higher doses of EPOGEN®.
Maintenance Dose: After attainment of the desired response (ie, reduced transfusion requirements or increased hemoglobin), the dose of EPOGEN® should be titrated to maintain the response based on factors such as variations in zidovudine dose and the presence of intercurrent infectious or inflammatory episodes. If the hemoglobin exceeds the upper safety limit of 12 g/dL, the dose should be discontinued until the hemoglobin drops below 11 g/dL. The dose should be reduced by 25% when treatment is resumed and then titrated to maintain the desired hemoglobin.
Cancer Patients on Chemotherapy
Although no specific serum erythropoietin level has been established which predicts which patients would be unlikely to respond to EPOGEN® therapy, treatment of patients with grossly elevated serum erythropoietin levels (eg, > 200 mUnits/mL) is not recommended. Therapy should not be initiated at hemoglobin levels ≥ 10 g/dL. The hemoglobin should be monitored on a weekly basis in patients receiving EPOGEN® therapy until hemoglobin becomes stable. The dose of EPOGEN® should be titrated for each patient to achieve and maintain the lowest hemoglobin level sufficient to avoid the need for blood transfusion (see recommended Dose Modifications, below).
Recommended Dose: The initial recommended dose of EPOGEN® in adults is 150 Units/kg SC TIW or 40,000 Units SC Weekly. The initial recommended dose of EPOGEN® in pediatric patients is 600 Units/kg IV weekly. Discontinue EPOGEN® following the completion of a chemotherapy course (see BOXED WARNINGS: Cancer).
Dose Modification
| Starting Dose: | |
| Adults | 150 Units/kg SC TIW |
| Reduce Dose by 25% when: |
Hemoglobin reaches a level needed to avoid transfusion or increases > 1 g/dL in any 2-week period. |
| Withhold Dose if: | Hemoglobin exceeds a level needed to avoid transfusion. Restart at 25% below the previous dose when the hemoglobin approaches a level where transfusions may be required. |
| Increase Dose to 300 Units/kg TIW if: | Response is not satisfactory (no reduction in transfusion requirements or rise in hemoglobin) after 4 weeks to achieve and maintain the lowest hemoglobin level sufficient to avoid the need for RBC transfusion. |
| Discontinue: | If after 8 weeks of therapy there is no response as measured by hemoglobin levels or if transfusions are still required. |
| Starting Dose: | |
| Adults | 40,000 Units SC |
| Pediatrics | 600 Units/kg IV (maximum 40,000 Units) |
| Reduce Dose by 25% when: | Hemoglobin reaches a level needed to avoid transfusion or increases > 1 g/dL in any 2-weeks. |
| Withhold Dose if: | Hemoglobin exceeds a level needed to avoid transfusion and restart at 25% below the previous dose when the hemoglobin approaches a level where transfusions may be required. |
| Increase Dose if: For Adults: 60,000 Units SC Weekly For Pediatrics: 900 Units/kg IV (maximum 60,000 Units) if: | Response is not satisfactory (no increase in hemoglobin by ≥ 1g/dL after 4 weeks of therapy, in the absence of a RBC transfusion) to achieve and maintain the lowest hemoglobin level sufficient to avoid the need for RBC transfusion. |
| Discontinue: | If after 8 weeks of therapy there is no response as measured by hemoglobin levels or if transfusions are still required. |
Surgery Patients
Prior to initiating treatment with EPOGEN® a hemoglobin should be obtained to establish that it is > 10 to ≤ 13 g/dL.17 The recommended dose of EPOGEN® is 300 Units/kg/day subcutaneously for 10 days before surgery, on the day of surgery, and for 4 days after surgery.
An alternate dose schedule is 600 Units/kg EPOGEN® subcutaneously in once weekly doses (21, 14, and 7 days before surgery) plus a fourth dose on the day of surgery.18
All patients should receive adequate iron supplementation. Iron supplementation should be initiated no later than the beginning of treatment with EPOGEN® and should continue throughout the course of therapy. Deep venous thrombosis prophylaxis should be strongly considered (see BOXED WARNINGS).
PREPARATION AND ADMINISTRATION OF EPOGEN®
- Do not shake. It is not necessary to shake EPOGEN®. Prolonged vigorous shaking may denature any glycoprotein, rendering it biologically inactive.
- Protect the solution from light. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Do not use any vials exhibiting particulate matter or discoloration.
- Using aseptic techniques, attach a sterile needle to a sterile syringe. Remove the flip top from the vial containing EPOGEN®, and wipe the septum with a disinfectant. Insert the needle into the vial, and withdraw into the syringe an appropriate volume of solution.
-
Single-dose: 1 mL vial contains no preservative. Use one dose per vial; do not re-enter the vial. Discard unused portions.
Multidose: 1 mL and 2 mL vials contain preservative. Store at 2° to 8° C after initial entry and between doses. Discard 21 days after initial entry.
- Do not dilute or administer in conjunction with other drug solutions. However, at the time of SC administration, preservative-free EPOGEN® from single-use vials may be admixed in a syringe with bacteriostatic 0.9% sodium chloride injection, USP, with benzyl alcohol 0.9% (bacteriostatic saline) at a 1:1 ratio using aseptic technique. The benzyl alcohol in the bacteriostatic saline acts as a local anesthetic which may ameliorate SC injection site discomfort. Admixing is not necessary when using the multidose vials of EPOGEN® containing benzyl alcohol.
HOW SUPPLIED
EPOGEN®, containing Epoetin alfa, is available in the following packages:
1 mL Single-dose, Preservative-free Solution
2000 Units/mL (NDC 55513-126-10)
3000 Units/mL (NDC 55513-267-10)
4000 Units/mL (NDC 55513-148-10)
10,000 Units/mL (NDC 55513-144-10)
40,000 Units/mL (NDC 55513-823-10)
Supplied in dispensing packs containing 10 single-dose vials.
2 mL Multidose, Preserved Solution
10,000 Units/mL (NDC 55513-283-10)
1 mL Multidose, Preserved Solution
20,000 Units/mL (NDC 55513-478-10)
Supplied in dispensing packs containing 10 multidose vials.
STORAGE
Store at 2° to 8° C (36° to 46° F). Do not freeze or shake. Protect from light.
REFERENCES
- Egrie JC, Strickland TW, Lane J, et al. Characterization and Biological Effects of Recombinant Human Erythropoietin. Immunobiol. 1986;72:213-224.
- Graber SE, Krantz SB. Erythropoietin and the Control of Red Cell Production. Ann Rev Med. 1978;29:51-66.
- Eschbach JW, Adamson JW. Anemia of End-Stage Renal Disease (ESRD). Kidney Intl. 1985;28:1-5.
- Eschbach JW, Egrie JC, Downing MR, et al. Correction of the Anemia of End-Stage Renal Disease with Recombinant Human Erythropoietin. NEJM. 1987;316:73-78.
- Eschbach JW, Abdulhadi MH, Browne JK, et al. Recombinant Human Erythropoietin in Anemic Patients with End-Stage Renal Disease. Ann Intern Med. 1989;111:992-1000.
- Eschbach JW, Egrie JC, Downing MR, et al. The Use of Recombinant Human Erythropoietin (r-HuEPO): Effect in End-Stage Renal Disease (ESRD). In: Friedman, Beyer, DeSanto, Giordano, eds. Prevention of Chronic Uremia. Philadelphia, PA: Field and Wood Inc; 1989:148-155.
- Egrie JC, Eschbach JW, McGuire T, Adamson JW. Pharmacokinetics of Recombinant Human Erythropoietin (rHuEPO) Administered to Hemodialysis (HD) Patients. Kidney Intl. 1988;33:262.
- Paganini E, Garcia J, Ellis P, et al. Clinical Sequelae of Correction of Anemia with Recombinant Human Erythropoietin (r-HuEPO); Urea Kinetics, Dialyzer Function and Reuse. Am J Kid Dis. 1988;11:16.
- Delano BG, Lundin AP, Golansky R, et al. Dialyzer Urea and Creatinine Clearances Not Significantly Changed in rHuEPO Treated Maintenance Hemodialysis (MD) Patients. Kidney Intl. 1988;33:219.
- Stivelman J, Van Wyck D, Ogden D. Use of Recombinant Erythropoietin (r-HuEPO) with High Flux Dialysis (HFD) Does Not Worsen Azotemia or Shorten Access Survival. Kidney Intl. 1988;33:239.
- Lim VS, DeGowin RL, Zavala D, et al. Recombinant Human Erythropoietin Treatment in Pre-Dialysis Patients: A Double-Blind Placebo Controlled Trial. Ann Int Med.
1989;110:108-114. - Stone WJ, Graber SE, Krantz SB, et al. Treatment of the Anemia of Pre-Dialysis Patients with Recombinant Human Erythropoietin: A Randomized, Placebo-Controlled Trial. Am J Med Sci. 1988;296:171-179.
- Braun A, Ding R, Seidel C, Fies T, Kurtz A, Scharer K. Pharmacokinetics of recombinant human erythropoietin applied subcutaneously to children with chronic renal failure. Pediatr Nephrol. 1993;7:61-64.
- Geva P, Sherwood JB. Pharmacokinetics of recombinant human erythropoietin (rHuEPO) in pediatric patients on chronic cycling peritoneal dialysis (CCPD). Blood. 1991;78 (Suppl 1):91a.
- Jabs K, Grant JR, Harmon W, et al. Pharmacokinetics of Epoetin alfa (rHuEPO) in pediatric hemodialysis (HD) patients. J Am Soc Nephrol. 1991;2:380.
- Kling PJ, Widness JA, Guillery EN, Veng-Pedersen P, Peters C, DeAlarcon PA. Pharmacokinetics and pharmacodynamics of erythropoietin during therapy in an infant with renal failure. J Pediatr. 1992;121:822-825.
- deAndrade JR and Jove M. Baseline Hemoglobin as a Predictor of Risk of Transfusion and Response to Epoetin alfa in Orthoperdic Surgery Patients. Am. J. of Orthoped. 1996;25 (8):533-542.
- Goldberg MA and McCutchen JW. A Safety and Efficacy Comparison Study of Two Dosing Regimens of Epoetin alfa in Patients Undergoing Major Orthopedic Surgery. Am. J. of Orthoped. 1996;25 (8):544-552.
- Faris PM and Ritter MA. The Effects of Recombinant Human Erythropoietin on Perioperative Transfusion Requirements in Patients Having a Major Orthopedic Operation. J. Bone and Joint surgery. 1996;78-A:62-72.
- Amgen Inc., data on file.
- Eschbach JW, Kelly MR, Haley NR, et al. Treatment of the Anemia of Progressive Renal Failure with Recombinant Human Erythropoietin. NEJM. 1989;321:158-163.
- The US Recombinant Human Erythropoietin Predialysis Study Group. Double-Blind, Placebo-Controlled Study of the Therapeutic Use of Recombinant Human Erythropoietin for Anemia Associated with Chronic Renal Failure in Predialysis Patients. Am J Kid Dis. 1991;18:50-59.
- Ortho Biologics, Inc., data on file.
- Danna RP, Rudnick SA, Abels RI. Erythropoietin Therapy for the Anemia Associated with AIDS and AIDS Therapy and Cancer. In: MB Garnick, ed. Erythropoietin in Clinical Applications - An International Perspective. New York, NY: Marcel Dekker; 1990:301-324.
- Fischl M, Galpin JE, Levine JD, et al. Recombinant Human Erythropoietin for Patients with AIDS Treated with Zidovudine. NEJM. 1990;322:1488-1493.
- Laupacis A. Effectiveness of Perioperative Recombinant Human Eyrthropoietin in Elective Hip Replacement. Lancet. 1993;341:1228-1232.
- Campos A, Garin EH. Therapy of renal anemia in children and adolescents with recombinant human erythropoietin (rHuEPO). Clin Pediatr (Phila). 1992;31:94-99.
- Montini G, Zacchello G, Baraldi E, et al. Benefits and risks of anemia correction with recombinant human erythropoietin in children maintained by hemodialysis. J Pediatr. 1990;117:556-560.
- Offner G, Hoyer PF, Latta K, Winkler L, Brodehl J, Scigalla P. One year's experience with recombinant erythropoietin in children undergoing continuous ambulatory or cycling peritoneal dialysis. Pediatr Nephrol. 1990;4:498-500.
- Muller-Wiefel DE, Scigalla P. Specific problems of renal anemia in childhood. Contrib Nephrol. 1988;66:71-84.
- Scharer K, Klare B, Dressel P, Gretz N. Treatment of renal anemia by subcutaneous erythropoietin in children with preterminal chronic renal failure. Acta Paediatr. 1993;82:953-958.
- Mueller BU, Jacobsen RN, Jarosinski P, et al. Erythropoietin for zidovudine-associated anemia in children with HIV infection. Pediatr AIDS and HIV Infect: Fetus to Adolesc. 1994;5:169-173.
- Zuccotti GV, Plebani A, Biasucci G, et al. Granulocyte-colony stimulating factor and erythropoietin therapy in children with human immunodeficiency virus infection. J Int Med Res. 1996;24:115-121.
- Raskin NH, Fishman RA. Neurologic Disorders in Renal Failure (First of Two Parts). NEJM. 1976;294:143-148.
- Raskin NH and Fishman RA. Neurologic Disorders in Renal Failure (Second of Two Parts). NEJM. 1976;294:204-210.
- Messing RO, Simon RP. Seizures as a Manifestation of Systemic Disease. Neurologic Clinics. 1986;4:563-584.
- Besarab A, Bolton WK, Browne JK, et al. The effects of normal as compared with low hematocrit values in patients with cardiac disease who are receiving hemodialysis and epoetin. NEJM. 1998;339:584-90.
- Henke, M, Laszig, R, Rübe, C., et al. Erythropoietin to treat head and neck cancer patients with anaemia undergoing radiotherapy: randomized, double-blind, placebo-controlled trial. The Lancet. 2003; 362:1255-1260.
- Widness JA, Veng-Pedersen P, Peters C, Pereira LM, Schmidt RL, Lowe SL. Erythropoietin Pharmacokinetics in Premature Infants: Developmental, Nonlinearity, and Treatment Effects. JAppl Physiol. 1996;80 (1):140-148.
- Singh AK, Szczech L, Tang KL, et al. Correction of Anemia with Epoetin Alfa in Chronic Kidney Disease, N Engl j Med. 2006; 355:2085-98.
- Bohlius J, Wilson J, Seidenfeld J, et at. Recombinant HumanErythropoietins and Cancer Patients: Updated Meta-Analysis of 57 Studies Including 9353 Patients. J Natl Cancer Inst. 2006; 98:708-14.
- D’Ambra MN, Gray RJ, Hillman R, et al. Effect of Recombinant Human Erythropoietin on Transfusion Risk in Coronary Bypass Patients. Ann Thorac Surg. 1997; 64: 1686-93.
- Leyland-Jones B, Semiglazov V, Pawlicki M, et al. Maintaining Normal Hemoglobin Levels With Epoetin Alfa in Mainly Nonanemic Patients With Metastatic Breast Cancer Receiving First-Line Chemotherapy: A Survival Study. JCO. 2005; 23(25): 1-13.
This product’s label may have been revised after this insert was used in production. For further product information and the current package insert, please visit www.amgen.com or call our medical information department toll-free at 1-800-77AMGEN (1-800-772-6436).
Manufactured by:
Amgen Manufacturing, Limited, a subsidiary of Amgen Inc.
One Amgen Center Drive
Thousand Oaks, CA 91320-1799
©1989-2008 Amgen Inc. All rights reserved.
1xxxxxx – v20
Revised: 08/2008
MEDICATION GUIDE
Epogen® (Ee-po-jen)
(epoetin alfa)
Read this Medication Guide before you start Epogen, each time you refill your prescription, and if you are told by your healthcare provider that there is new information about Epogen. This Medication Guide was revised August 2008. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or your treatment. Talk with your healthcare provider regularly about the use of Epogen and ask if there is new information about Epogen.
What is the most important information I should know about Epogen?
Using Epogen can lead to death or other serious side effects.
Patients with cancer:
Your tumor may grow faster and you may die sooner when Epogen is used experimentally to try to raise your hemoglobin beyond the amount needed to avoid red blood cell transfusion or given to patients who are not getting strong doses of chemotherapy. It is not known whether these risks exist when Epogen is given according to the FDA-approved directions for use.
You should discuss with your doctor:
- Why Epogen treatment is being prescribed.
- What are the chances you will get red blood cell transfusions if you do not take Epogen.
- What are the chances you will get red blood cell transfusions even if you take Epogen.
- How taking Epogen may affect the success of your cancer treatment.
If you decide to take Epogen, your healthcare provider should prescribe the smallest dose of Epogen to lower the chance of getting red blood cell transfusions.
- After you have finished your chemotherapy course, Epogen treatment should be stopped.
- Epogen does not improve the symptoms of anemia (lower than normal number of red blood cells), quality of life, fatigue, or well-being for patients with cancer.
All patients, including patients with cancer or chronic kidney failure:
- You may get serious heart problems such as heart attack, stroke, heart failure, and may die sooner if you are treated with Epogen to a hemoglobin level above 12 g/dL.
- You may get blood clots at any time while taking Epogen. If you are receiving Epogen and you are going to have surgery, talk to your healthcare provider about whether or not you need to take a blood thinner to lessen the chance of blood clots during or following surgery. Clots can form in blood vessels (veins), especially in your leg (deep venous thrombosis or DVT). Pieces of a blood clot may travel to the lungs and block the blood circulation in the lungs (pulmonary embolus).
Call your healthcare provider or get medical help right away if you have any of these symptoms of blood clots:
- Chest pain
- Trouble breathing or shortness of breath
- Pain in your legs, with or without swelling
- A cool or pale arm or leg
- Sudden confusion, trouble speaking, or trouble understanding others’ speech
- Sudden numbness or weakness in your face, arm, or leg, especially on one side of your body
- Sudden trouble seeing
- Sudden trouble walking, dizziness, loss of balance or coordination
- Loss of consciousness (fainting)
- Hemodialysis vascular access stops working. If you are a patient with chronic kidney failure and have a hemodialysis vascular access, blood clots may form in this access.
Also see “What are the possible side effects of Epogen?" below.
What is Epogen?
Epogen is a man-made form of the protein human erythropoietin that is given to patients to lessen the need for red blood cell transfusions. Epogen stimulates your bone marrow to make more red blood cells. Having more red blood cells raises your hemoglobin level. If your hemoglobin level stays too high or if your hemoglobin goes up too quickly, this may lead to serious health problems which may result in death. These serious health problems may happen even if you take Epogen and do not have an increase in your hemoglobin level.
Epogen may be used to treat a lower than normal number of red blood cells (anemia) if it is caused by:
- Chronic kidney failure (you may or may not be on dialysis)
- Chemotherapy that is used for at least two months to treat some types of cancer
- A medicine called zidovudine (AZT) used to treat HIV infection
Epogen may also be used if you are scheduled for certain surgeries with a lot of blood loss to reduce the chance you will need red blood cell transfusions.
Who should not take Epogen?
Do not take Epogen if you:
- Have high blood pressure that is not controlled (uncontrolled hypertension).
- Have allergies to any of the ingredients in Epogen. See the end of this Medication Guide for a complete list of ingredients in Epogen.
What should I tell my healthcare provider before taking Epogen?
Epogen may not be right for you. Tell your healthcare provider about all your health conditions, including if you:
- Have heart disease.
- Have high blood pressure.
- Have had a seizure (convulsion) or stroke.
- Are pregnant or planning to become pregnant. It is not known if Epogen may harm your unborn baby.
- Are breast-feeding or planning to breast-feed. It is not known if Epogen passes into breast milk.
- Women who do not have regular monthly menstrual periods may begin to have monthly periods while taking Epogen. Talk with your doctor about the possibility of pregnancy while taking Epogen.
Tell your healthcare provider about all the medicines you take, including prescription and nonprescription medicines, vitamins, and herbal supplements.
Know the medicines you take. Keep a list of your medicines with you and show it to your healthcare provider when you get a new medicine.
How should I take Epogen?
- Continue to follow your healthcare provider’s instructions for diet, dialysis, and medicines, including medicines for high blood pressure, while taking Epogen.
- Have your blood pressure checked as instructed by your healthcare provider.
- If you or your caregiver has been trained to give Epogen shots (injections) at home:
- Be sure that you read, understand, and follow the “Patient Instructions for Use” that come with Epogen.
- Take Epogen exactly as your healthcare provider tells you to. Do not change the dose of Epogen unless told to do so by your healthcare provider.
- Your healthcare provider will show you how much Epogen to use, how to inject it, how often it should be injected, and how to safely throw away the used vial, syringes, and needles.
- If you miss a dose of Epogen, call your healthcare provider right away and ask what to do.
- If you take more than the prescribed amount of Epogen, call your healthcare provider right away.
What are the possible side effects of Epogen?
Epogen may cause serious side effects. See “What is the most important information I should know about Epogen?”
Other side effects of Epogen, which may also be serious, include:
- High blood pressure in patients with chronic kidney failure. Your blood pressure may go up or be difficult to control with blood pressure medicine while taking Epogen. This can happen even if you have never had high blood pressure before. Your healthcare provider should check your blood pressure often. If your blood pressure does go up, your healthcare provider may prescribe new or more blood pressure medicine.
- Seizures. If you have any seizures while taking Epogen, get medical help right away and tell your healthcare provider.
- Antibodies to Epogen. Your body may make antibodies to Epogen. These antibodies can block or lessen your body’s ability to make red blood cells and cause you to have severe anemia. Call your healthcare provider if you have unusual tiredness, lack of energy, dizziness, or fainting. You may need to stop taking Epogen.
- Serious allergic reactions. Serious allergic reactions can cause a rash over your whole body, shortness of breath, wheezing, dizziness and fainting because of a drop in blood pressure, swelling around your mouth or eyes, fast pulse, or sweating. If you have a serious allergic reaction, stop using Epogen and call your healthcare provider or get medical help right away.
- Dangers of giving Epogen to premature babies. Epogen from multidose vials that contain benzyl alcohol should not be given to premature babies because it can cause death and brain damage.
Common side effects of Epogen include:
- Rash
- Swelling in your legs and arms
- Injection site reaction, including irritation and pain
These are not all of the possible side effects of Epogen. Your healthcare provider can give you a more complete list. Tell your healthcare provider about any side effects that bother you or that do not go away.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store Epogen?
- Store Epogen in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Do not freeze. Do not use a vial of Epogen that has been frozen.
- Keep away from direct light.
- Do not shake Epogen.
- Throw away multidose vials of Epogen after 21 days from the first day that you put a needle into the vial.
- Single use vials of Epogen should be used only one time. Throw the vial away after use even if there is medicine left in the vial.
Keep Epogen and all medicines out of the reach of children.
General information about Epogen
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Use Epogen only for the condition for which it has been prescribed. Do not give Epogen to other people even if they have the same symptoms that you have. It may harm them.
This Medication Guide summarizes the most important information about Epogen. If you would like more information about Epogen, talk to your healthcare provider. You can ask your healthcare provider or pharmacist for information about Epogen that is written for healthcare professionals. For more information, go to the following website: www.epogen.com or call 1-800-77-AMGEN.
What are the ingredients in Epogen?
Active Ingredient: epoetin alfa
Inactive Ingredients: All formulations include albumin (human), sodium citrate, sodium chloride, and citric acid in water for injection. Multi-use vials contain benzyl alcohol. Certain formulations also contain sodium phosphate monobasic monohydrate and sodium phosphate dibasic anhydrate.
Revised: 08/2008
This Medication Guide has been approved by the U.S. Food and Drug Administration.
[Amgen Logo]
Manufactured by:
Amgen Manufacturing, Limited, a subsidiary of Amgen Inc.
One Amgen Center Drive
Thousand Oaks, CA 91320-1799
© 1989-2008 Amgen Inc. All rights reserved.
1xxxxxx- v1
Patient Instructions for Use
Epogen® (Ee-po-jen)
(epoetin alfa)
Use these instructions if you or your caregiver has been trained to give Epogen injections at home. Do not give yourself the injection unless you have received training from your healthcare provider. If you are not sure about giving the injection or you have questions, ask your healthcare provider for help.
Before reading these instructions for use, read the Medication Guide that comes with Epogen for the most important information you need to know.
When you receive your Epogen vial and syringes make sure that:
- The name Epogen appears on the carton and vial label.
- The expiration date on the vial label has not passed. Do not use a vial of Epogen after the expiration date on the label.
- The dose strength of the Epogen vial (number of Units per mL on the vial label) is the same as your healthcare provider prescribed.
- You understand what the dose strength of Epogen means. Epogen vials come in several dose strengths. For example, the dose strength may be described as 10,000 Units/mL on the vial label. This strength means that 10,000 Units of medicine are contained in each 1 mL (milliliter) of liquid. Your healthcare provider may also refer to a mL as a “cc.” One mL is the same as one “cc.”
- The Epogen liquid in the vial is clear and colorless. Do not use Epogen if the liquid in the vial looks discolored or cloudy, or if the liquid has lumps, flakes, or particles.
- The Epogen vial has a color cap on the top of the vial. Do not use a vial of Epogen if the color cap on the top of the vial has been removed or is missing.
- Use only the type of disposable syringe and needle that your healthcare provider has prescribed.
- Do not shake Epogen. If shaking has occurred, the solution in the vial may look foamy and should not be used.
- Do not freeze Epogen. Do not use a vial of Epogen that has been frozen.
- Keep Epogen away from light.
How should I prepare for an injection of Epogen?
- Always keep an extra syringe and needle on hand.
- Follow your healthcare provider’s instructions on how to measure your dose of Epogen. This dose will be measured in Units per mL or cc (1 mL is the same as 1 cc). Use a syringe that is marked in tenths of mL (for example, 0.2 mL or 0.2 cc). Using the wrong syringe can lead to a mistake in your dose and you could inject too much or too little Epogen.
Only use disposable syringes and needles. Use the syringes and needles only one time and thenthrow them away as instructed by your healthcare provider.
What do I need to know about the different types of Epogen vials?
Epogen comes in two different types of vials.
- Single-Use Vials
- Multidose-Use Vials
Single-Use Vials
If you have been prescribed Epogen vials for single use:
- Single-Use vials of Epogen come in several different strengths (Units per mL). For example, the vial label may say the strength is 2,000 Units/mL, which means the vial contains 2,000 Units of medicine in 1 mL. Injecting 1 mL of this strength means you will receive 2,000 Units of Epogen.
- Double-check to be sure you are using a vial that contains the correct strength of Epogen.
- The Single-Use Vials cannot be used more than one time and any unused medicine in the vial should be thrown away as directed by your healthcare provider.
Multidose-Use Vials
If you have been prescribed Epogen vials for use multiple times:
- Multidose vials of Epogen may be used to inject more than one dose of Epogen, as prescribed by your healthcare provider.
- Multidose vials of Epogen come in 1 mL and 2 mL vials.
- 1 mL vials contain 20,000 Units of Epogen in 1 mL of liquid (this means each vial contains a total of 20,000 Units of Epogen).
- 2 mL vials contain 10,000 Units of Epogen in each 1 mL of liquid, and since the vials contain a total of 2 mL of liquid, each vial contains a total of 20,000 Units of Epogen.
- After removing a dose from the vial, store the vial in the refrigerator (but not the freezer). Do not store the vial for more than 21 days.
- If the vial is stored for more than 21 days, or when a vial no longer contains a full dose of medicine, throw the vial away as directed by your healthcare provider.
Important: Follow these instructions exactly to help avoid infections.
Preparing the dose:
- Remove the vial of Epogen from the refrigerator. During this time, protect the solution from light.
- Do not use a Single-Use vial of Epogen more than one time.
- Do not shake Epogen.
- Gather the other supplies you will need for your injection (vial, syringe, alcohol wipes, cotton ball, and a puncture-proof container for throwing away the syringe and needle). See Figure 1.
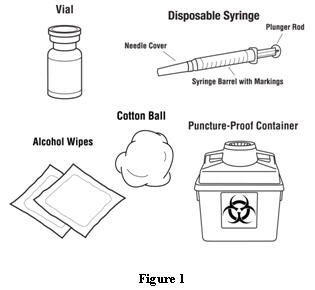
- Check the date on the Epogen vial to be sure that the drug has not expired.
- Wash your hands well with soap and water before preparing the medicine. See Figure 2.
- Flip off the protective color cap on the top of the vial. Do not remove the grey rubber stopper. Wipe the top of the grey rubber stopper with an alcohol wipe. See Figures 3 and 4.
- Check the package containing the syringe. If the package has been opened or damaged, do not use that syringe. Throw away the syringe in the puncture-proof disposable container. If the syringe package is undamaged, open the package and remove the syringe.
- Using a syringe and needle that has been recommended by your healthcare provider, carefully remove the needle cover. See Figure 5. Then draw air into the syringe by pulling back on the plunger. The amount of air drawn into the syringe should be equal to the amount (mL or cc) of the Epogen dose prescribed by your healthcare provider. See Figure 6.
- With the vial on a flat work surface, insert the needle straight down through the grey rubber stopper of the Epogen vial. See Figure 7.
- Push the plunger of the syringe down to inject the air from the syringe into the vial of Epogen. The air injected into the vial will allow Epogen to be easily withdrawn into the syringe. See Figure 7.
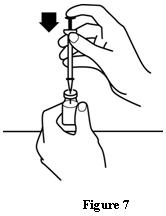
- Keep the needle inside the vial. Turn the vial and syringe upside down. Be sure the tip of the needle is in the Epogen liquid. Keep the vial upside down. Slowly pull back on the plunger to fill the syringe with Epogen liquid to the number (mL or cc) that matches the dose your healthcare provider prescribed. See Figure 8.
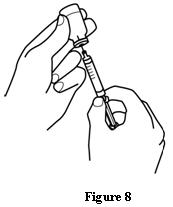
- Keep the needle in the vial. Check for air bubbles in the syringe. A small amount of air is harmless. Too large an air bubble will give you the wrong Epogen dose. To remove air bubbles, gently tap the syringe with your fingers until the air bubbles rise to the top of the syringe. Slowly push the plunger up to force the air bubbles out of the syringe. Keep the tip of the needle in the Epogen liquid. Pull the plunger back to the number on the syringe that matches your dose. Check again for air bubbles. If there are still air bubbles, repeat the steps above to remove them. See Figures 9 and 10.
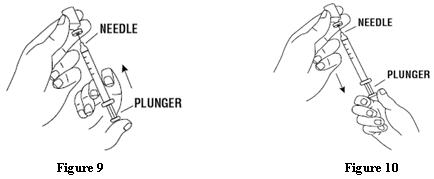
- Double-check that you have the correct dose in the syringe. Lay the vial down on its side with the needle still in it until after you have selected and prepared your site for injection.
Selecting and preparing the injection site:
Epogen can be injected into your body using two different ways (routes) as described below. Follow your healthcare provider’s instructions about how you should inject Epogen. In patients on hemodialysis, the intravenous (IV) route is recommended.
1. Subcutaneous Route:
- Epogen can be injected directly into a layer of fat under your skin. This is called a subcutaneous injection. When giving subcutaneous injections, follow your healthcare provider’s instructions about changing the site for each injection. You may wish to write down the site where you have injected.
- Do not inject Epogen into an area that is tender, red, bruised, hard, or has scars or stretch marks. Recommended sites for injection are presented in Figure 11 below, including:
- The outer area of the upper arms
- The abdomen (except for the 2-inch area around the navel)
- The front of the middle thighs
- The upper outer area of the buttocks
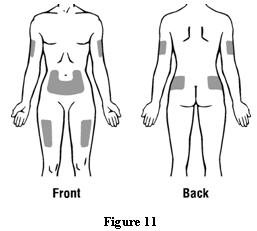
- Clean the skin with an alcohol wipe where the injection is to be made. Be careful not to touch the skin that has been wiped clean. See Figure 12.
- Double-check that the correct amount of Epogen is in the syringe.
- Remove the prepared syringe and needle from the vial of Epogen and hold it in the hand that you will use to inject the medicine.
- Use the other hand to pinch a fold of skin at the cleaned injection site. Do not touch the cleaned area of skin. See Figure 13.
- Hold the syringe like you would hold a pencil. Use a quick “dart-like” motion to insert the needle either straight up and down (90-degree angle) or at a slight angle (45 degrees) into the skin. Let go of the skin and pull the plunger back slightly. If blood comes into the syringe, do not inject Epogen since the needle may have entered a blood vessel; instead, withdraw the syringe, discard it in the puncture-proof container. Prepare a new syringe of Epogen using the instructions above. Clean a new area of skin. In this new area of clean skin, again insert a new needle (as you did before), and again pull the plunger back slightly. If blood does not enter the syringe, inject the Epogen by pushing the plunger all the way down. See Figure 14.
- Pull the needle out of the skin and press a cotton ball or gauze over the injection site and hold it there for several seconds. Do not recap the needle.
- Dispose of the used syringe and needle as described below. Do not reuse syringes and needles.
2. Intravenous Route:
- Epogen can be injected in your vein through a special access port put in by your healthcare provider. This type of Epogen injection is called an intravenous (IV) injection. This route is usually for hemodialysis patients.
- If you have a dialysis vascular access, make sure it is working by checking it as your healthcare provider has shown you. Be sure to let your healthcare provider know right away if you are having any problems, or if you have any questions.
- Wipe off the venous port of the hemodialysis tubing with an alcohol wipe. See Figure 15.
- Insert the needle of the syringe into the cleaned venous port and push the plunger all the way down to inject all the Epogen. See Figure 16.
- Remove the syringe from the venous port. Do not recap the needle.
- Dispose of the used syringe and needle as described below.
How should I dispose of syringes and needles?
Do not reuse disposable syringes and needles. Throw away syringes and needles as instructed by your healthcare provider by following these steps:
- Do not throw the needle, syringe, or disposable container in the household trash or recycle.
- Do not put the needle cover back on the needle.
- Place all used needles and syringes in a puncture-proof disposable container with a lid. Do not use glass or clear plastic containers, or any container that will be recycled or returned to a store.
- Keep the container out of the reach of children.
- When the container is full, tape around the cap or lid to make sure the cap or lid does not come off. Throw away the puncture-proof disposable container as instructed by your healthcare provider. There may be special state and local laws for disposing of used needles and syringes. Do not throw the disposable container in the household trash. Do not recycle.
[Amgen Logo]
Manufactured by:
Amgen Manufacturing, Limited, a subsidiary of Amgen Inc.
One Amgen Center Drive
Thousand Oaks, California 91320-1799
© 1989-2008 Amgen Inc. All rights reserved.
1xxxxxx - v1
Revised: 08/2008
| EPOGEN
epoetin alfa solution |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| EPOGEN
epoetin alfa solution |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| EPOGEN
epoetin alfa solution |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| EPOGEN
epoetin alfa solution |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| EPOGEN
epoetin alfa solution |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| EPOGEN
epoetin alfa solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| EPOGEN
epoetin alfa solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
Revised: 08/2008Amgen