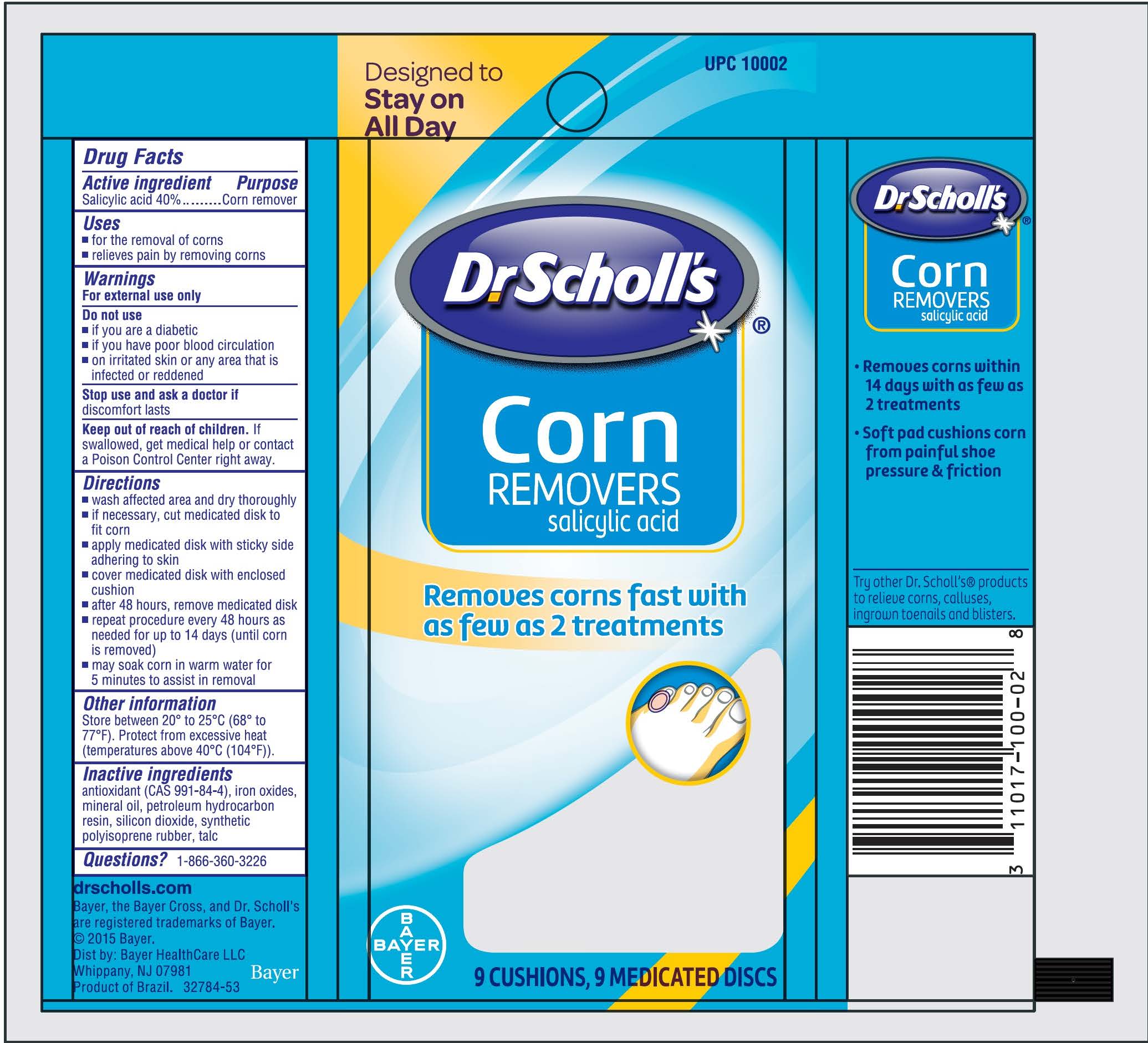
DR. SCHOLLS CORN REMOVERS- salicylic acid disc
Bayer HealthCare LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Dr Scholl's ®Corn REMOVERS
Warnings
For external use only
Directions
- wash affected area and dry thoroughly
- if necessary, cut medicated disc to fit corn
- apply medicated disc with sticky adhering to skin
- cover medicated disc with enclosed cushion
- after 48 hours , remove medicated disc
- repeat procedure every 48 hours as needed for up to 14 days (until corn is removed)
- may soak corn in warm water for 5 minutes to assist in removal
Other information
▪ Store between 20° to 25°C (68° to 77°F).
▪ Protect from excessive heat (temperatures above 40°C (104°F)).
| DR. SCHOLLS
CORN REMOVERS
salicylic acid disc |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Bayer HealthCare LLC (112117283) |
Revised: 12/2019
Document Id: 98e76167-5d16-f071-e053-2995a90a5248
Set id: eea68a23-6f04-4979-a269-b63f6a1746d9
Version: 8
Effective Time: 20191204
Bayer HealthCare LLC