DUET DHA BALANCED- .beta.-carotene, ascorbic acid, cholecalciferol, .alpha.-tocopherol acetate, d-, thiamine mononitrate, riboflavin, niacinamide, pyridoxine hydrochloride, folic acid, cyanocobalamin, calcium carbonate, iron, magnesium oxide, zinc oxide, cupric oxide, iodine, omega-3 fatty acids
Eckson Labs, LLC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Duet DHA Balanced
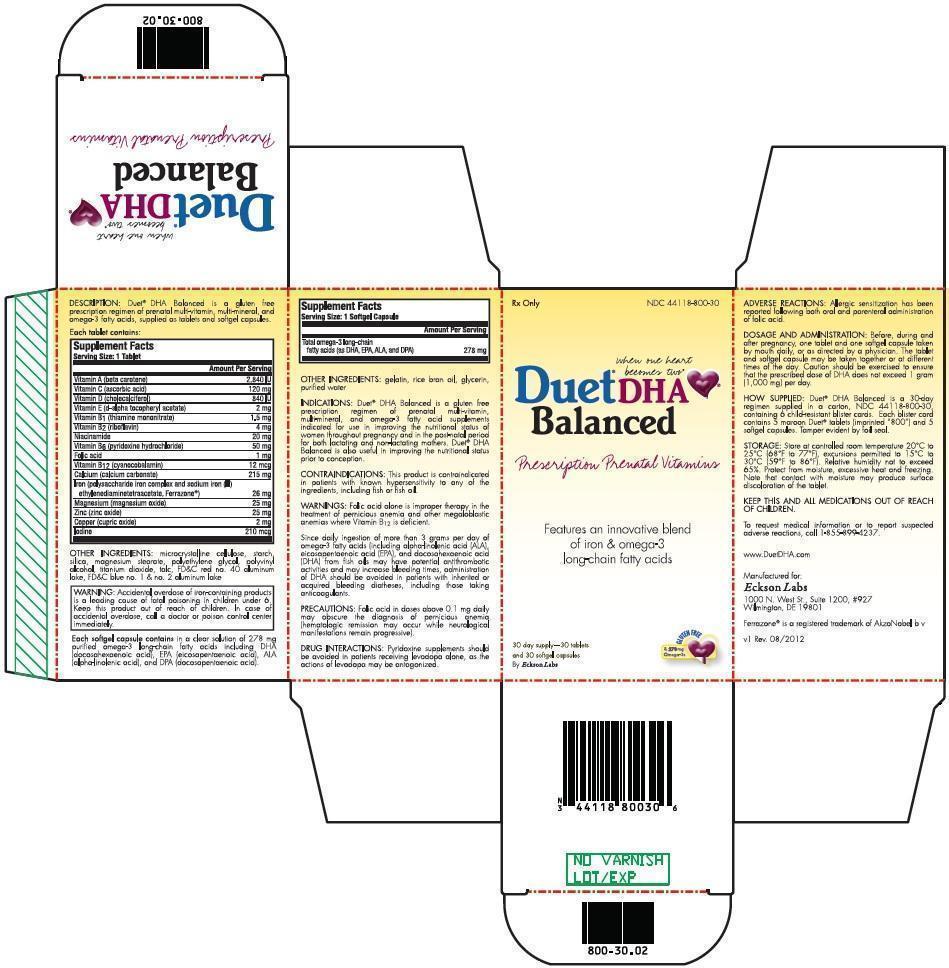
DESCRIPTION:
Duet ® DHA Balanced is a gluten free prescription regimen of prenatal multi-vitamin, multi-mineral, and omega-3 fatty acids, supplied as tablets and softgel capsules.
Each tablet contains:
| Supplement Facts
Serving Size: 1 Tablet |
|
| Amount Per Serving | |
| Vitamin A (beta carotene) | 2,840 IU |
| Vitamin C (ascorbic acid) | 120 mg |
| Vitamin D (cholecalciferol) | 840 IU |
| Vitamin E (d-alpha tocopheryl acetate) | 2 mg |
| Vitamin B 1 (thiamine mononitrate) | 1.5 mg |
| Vitamin B 2 (riboflavin) | 4 mg |
| Niacinamide | 20 mg |
| Vitamin B 6 (pyridoxine hydrochloride) | 50 mg |
| Folic acid | 1 mg |
| Vitamin B 12 (cyanocobalamin) | 12 mcg |
| Calcium (calcium carbonate) | 215 mg |
| Iron (polysaccharide iron complex and sodium iron (III) ethylenediaminetetraacetate, Ferrazone ®) | 26 mg |
| Magnesium (magnesium oxide) | 25 mg |
| Zinc (zinc oxide) | 25 mg |
| Copper (cupric oxide) | 2 mg |
| Iodine | 210 mcg |
OTHER INGREDIENTS:
microcrystalline cellulose, starch, silica, magnesium stearate, polyethylene glycol, polyvinyl alcohol, titanium dioxide, talc, FD&C red no. 40 aluminum lake, FD&C blue no. 1 & no. 2 aluminum lake
| WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. Keep this product out of reach of children. In case of accidental overdose, call a doctor or poison control center immediately. |
Each softgel capsule contains
in a clear solution of 278 mg purified omega-3 long-chain fatty acids including DHA (docosahexaenoic acid), EPA (eicosapentaenoic acid), ALA (alpha-linolenic acid), and DPA (docosapentaenoic acid).
| Supplement Facts
Serving Size: 1 Softgel Capsule |
|
|
| Amount Per Serving |
| Total omega-3 long-chain fatty acids (as DHA, EPA, ALA, and DPA) | 278 mg |
OTHER INGREDIENTS:
gelatin, rice bran oil, glycerin, purified water
INDICATIONS:
Duet ® DHA Balanced is a gluten free prescription regimen of prenatal multi-vitamin, multi-mineral, and omega-3 fatty acid supplements indicated for use in improving the nutritional status of women throughout pregnancy and in the post-natal period for both lactating and non-lactating mothers. Duet ® DHA Balanced is also useful in improving the nutritional status prior to conception.
CONTRAINDICATIONS:
This product is contraindicated in patients with known hypersensitivity to any of the ingredients, including fish or fish oil.
WARNINGS:
Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where Vitamin B 12 is deficient.
Since daily ingestion of more than 3 grams per day of omega-3 fatty acids (including alpha-linolenic acid (ALA), eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA) from fish oils may have potential antithrombotic activities and may increase bleeding times, administration of DHA should be avoided in patients with inherited or acquired bleeding diatheses, including those taking anticoagulants.
PRECAUTIONS:
Folic acid in doses above 0.1 mg daily may obscure the diagnosis of pernicious anemia (hematologic remission may occur while neurological manifestations remain progressive).
DRUG INTERACTIONS:
Pyridoxine supplements should be avoided in patients receiving levodopa alone, as the actions of levodopa may be antagonized.
ADVERSE REACTIONS:
Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
DOSAGE AND ADMINISTRATION:
Before, during and after pregnancy, one tablet and one softgel capsule taken by mouth daily, or as directed by a physician. The tablet and softgel capsule may be taken together or at different times of the day. Caution should be exercised to ensure that the prescribed dose of DHA does not exceed 1 gram (1,000 mg) per day.
HOW SUPPLIED:
Duet ® DHA Balanced is a 30-day regimen supplied in a carton, NDC 44118-800-30, containing 6 child-resistant blister cards. Each blister card contains 5 maroon Duet ® tablets (imprinted “800”) and 5 softgel capsules. Tamper evident by foil seal.
STORAGE:
Store at controlled room temperature 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F). Relative humidity not to exceed 65%. Protect from moisture, excessive heat and freezing. Note that contact with moisture may produce surface discoloration of the tablet.
KEEP THIS AND ALL MEDICATIONS OUT OF REACH OF CHILDREN.
To request medical information or to report suspected adverse reactions, call 1-855-899-4237.
www.DuetDHA.com
Manufactured for:
Eckson Labs, LLC
1000 N. West St., Suite 1200, #927
Wilmington, DE 19801
Ferrazone ® is a registered trademark of AkzoNobel b v
v1 Rev. 08/2012
| DUET DHA BALANCED
.beta.-carotene, ascorbic acid, cholecalciferol, .alpha.-tocopherol acetate, d-, thiamine mononitrate, riboflavin, niacinamide, pyridoxine hydrochloride, folic acid, cyanocobalamin, calcium carbonate, iron, magnesium oxide, zinc oxide, cupric oxide, iodine, omega-3 fatty acids kit |
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Eckson Labs, LLC (078435242) |