Label: MAXIMUM STRENGTH ACNE- benzoyl peroxide 10% gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 36800-878-01 - Packager: TopCare
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 25, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
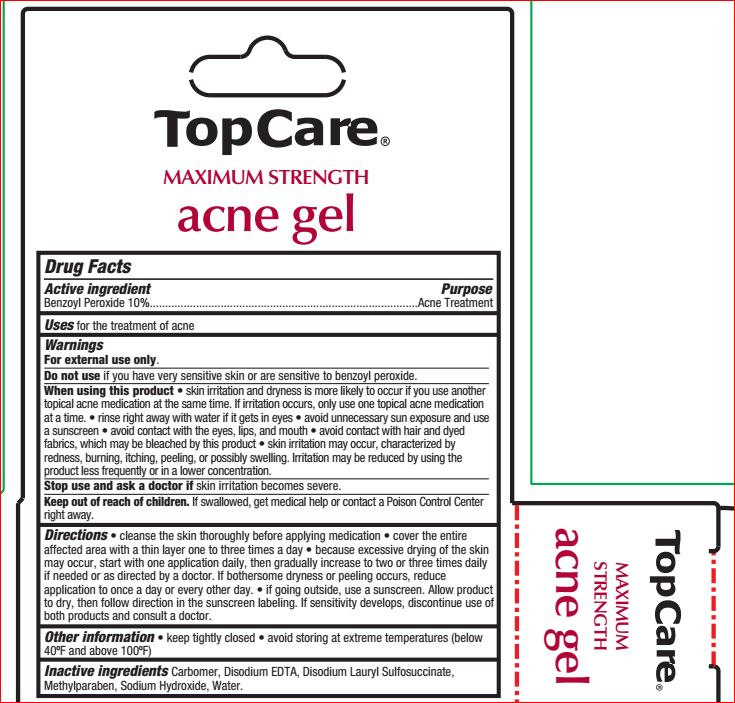
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
For external use only.
Do not use if you have very sensitive skin or are sensitive to benzoyl peroxide.
When using this product • skin irritation and dryness is more likely to occur if you use another
topical acne medication at the same time. If irritation occurs, only use one topical acne medication
at a time. • rinse right away with water if it gets in eyes • avoid unnecessary sun exposure and use
a sunscreen • avoid contact with the eyes, lips, and mouth • avoid contact with hair and dyed
fabrics, which may be bleached by this product • skin irritation may occur, characterized by
redness, burning, itching, peeling, or possibly swelling. Irritation may be reduced by using the
product less frequently or in a lower concentration.
Stop use and ask a doctor if skin irritation becomes severe.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center
right away - KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions • cleanse the skin thoroughly before applying medication • cover the entire
affected area with a thin layer one to three times a day • because excessive drying of the skin
may occur, start with one application daily, then gradually increase to two or three times daily
if needed or as directed by a doctor. If bothersome dryness or peeling occurs, reduce
application to once a day or every other day. • if going outside, use a sunscreen. Allow product
to dry, then follow direction in the sunscreen labeling. If sensitivity develops, discontinue use of
both products and consult a doctor. - INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MAXIMUM STRENGTH ACNE
benzoyl peroxide 10% gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:36800-878 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Benzoyl Peroxide (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) Benzoyl Peroxide 10 g in 100 g Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Disodium Lauryl Sulfosuccinate (UNII: P160Q81342) CARBOMER HOMOPOLYMER TYPE C (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 4Q93RCW27E) Methylparaben (UNII: A2I8C7HI9T) EDETATE DISODIUM (UNII: 7FLD91C86K) Sodium Hydroxide (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:36800-878-01 28 g in 1 BOTTLE; Type 0: Not a Combination Product 11/01/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333D 11/01/2011 Labeler - TopCare (006935977) Registrant - Product Quest Mfg (927768135) Establishment Name Address ID/FEI Business Operations Product Quest Mfg 927768135 manufacture(36800-878) , label(36800-878)