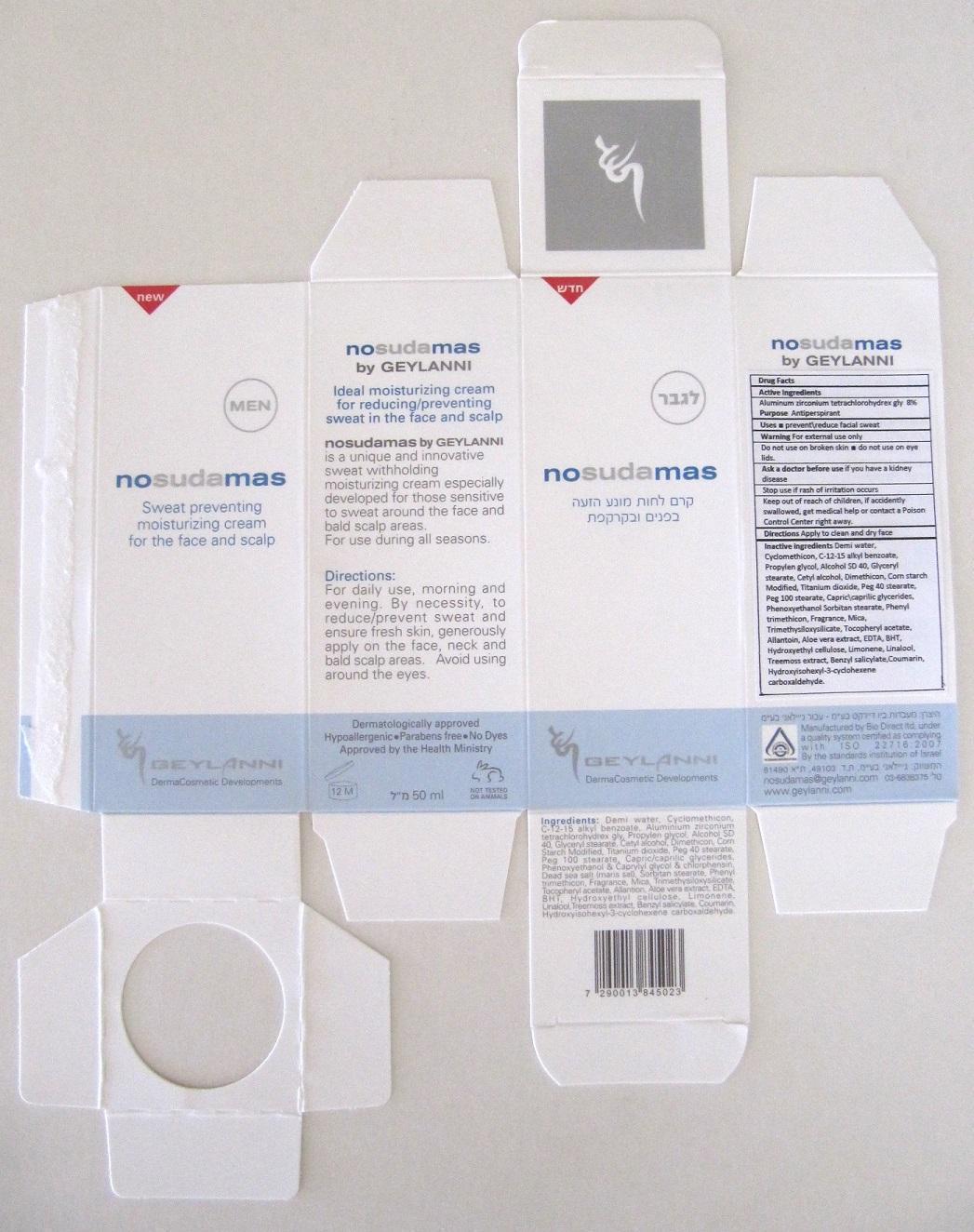
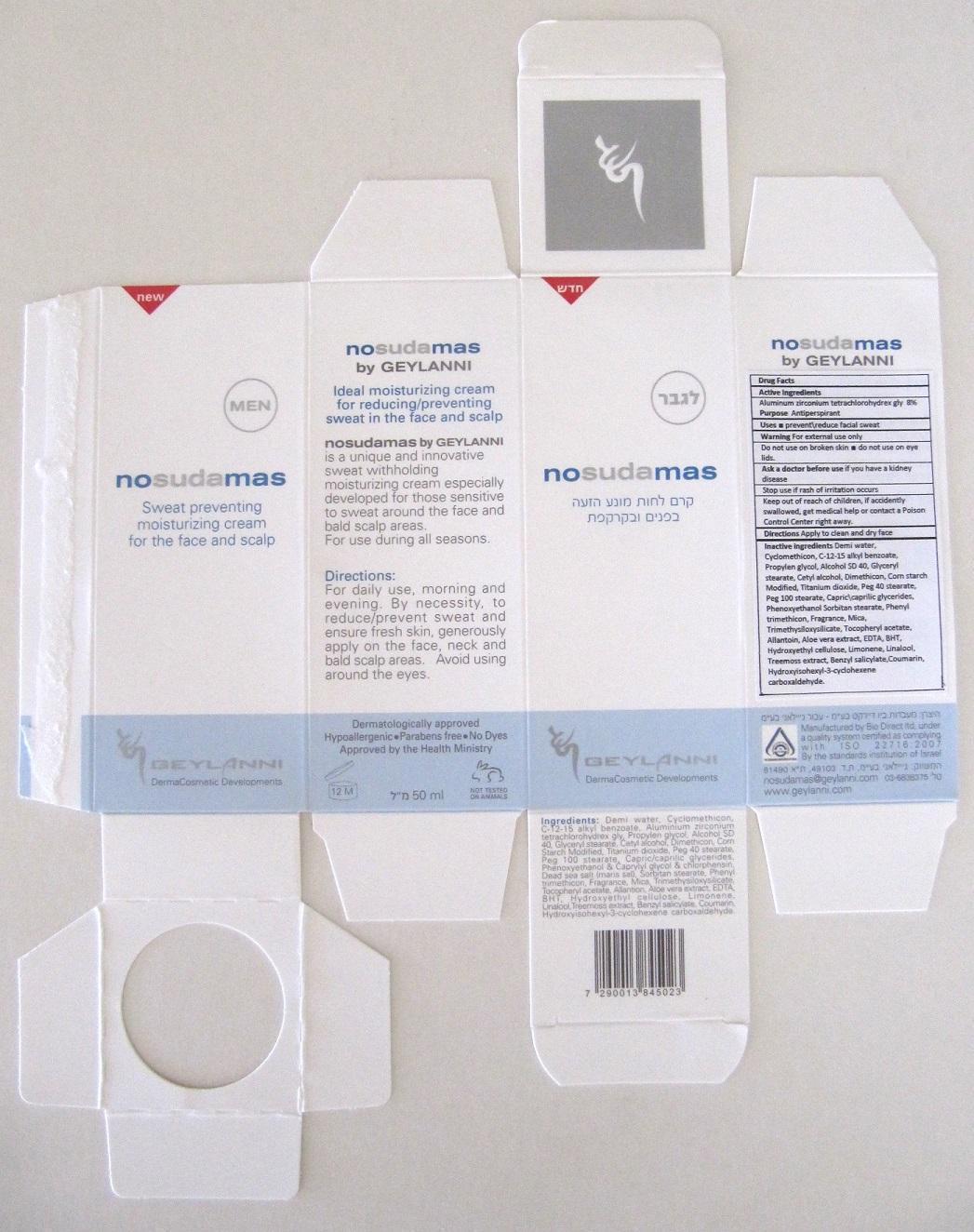
Label: NOSUDAMAS FOR MEN- aluminum zirconium tetrachlorohydrex gly cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 70064-002-01 - Packager: Geylanni International cosmetic developments LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 4, 2015
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Actives
-
Inactives
WATER
CYCLOMETHICONE
ALKYL (C12-15) BENZOATE
PROPYLENE GLYCOL
ALCOHOLCETYL ALCOHOL
GLYCERYL MONOSTEARATE
DIMETHICONE
STARCH, CORN
TITANIUM DIOXIDE
PEG-40 STEARATE
PEG-100 STEARATE
PEG-6 CAPRYLIC/CAPRIC GLYCERIDES
PHENOXYETHANOL
SORBITAN MONOSTEARATE
PHENYL TRIMETHICONE
MICA
TRIMETHYLSILOXYSILICATE (M/Q 0.6-0.8)
ALPHA-TOCOPHEROL ACETATE
ALLANTOIN
ALOE VERA LEAF
EDETIC ACID
BUTYLATED HYDROXYTOLUENE
HYDROXYETHYL CELLULOSE (100 MPA.S AT 2%)
LIMONENE, (+)-
LINALOOL, (+/-)-
PSEUDEVERNIA FURFURACEA
BENZYL SALICYLATE
COUMARINHYDROXYISOHEXYL 3-CYCLOHEXENE CARBOXALDEHYDE
- Purpose
- Usage
- Ask Doctor Before Use
- Stop Using
- Directions for Use
- Keep Out of Reach of Children
- Warning
- Carton Label
-
INGREDIENTS AND APPEARANCE
NOSUDAMAS FOR MEN
aluminum zirconium tetrachlorohydrex gly creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70064-002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Aluminum Zirconium Tetrachlorohydrex GLY (UNII: 8O386558JE) (ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY - UNII:8O386558JE) Aluminum Zirconium Tetrachlorohydrex GLY 80 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CYCLOMETHICONE (UNII: NMQ347994Z) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) ALCOHOL (UNII: 3K9958V90M) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) CETYL ALCOHOL (UNII: 936JST6JCN) DIMETHICONE (UNII: 92RU3N3Y1O) STARCH, CORN (UNII: O8232NY3SJ) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) PEG-40 STEARATE (UNII: ECU18C66Q7) PEG-100 STEARATE (UNII: YD01N1999R) PEG-6 CAPRYLIC/CAPRIC GLYCERIDES (UNII: GO50W2HWO8) PHENOXYETHANOL (UNII: HIE492ZZ3T) SORBITAN MONOSTEARATE (UNII: NVZ4I0H58X) PHENYL TRIMETHICONE (UNII: DR0K5NOJ4R) MICA (UNII: V8A1AW0880) TRIMETHYLSILOXYSILICATE (M/Q 0.6-0.8) (UNII: 5041RX63GN) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) ALLANTOIN (UNII: 344S277G0Z) ALOE VERA LEAF (UNII: ZY81Z83H0X) EDETIC ACID (UNII: 9G34HU7RV0) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) HYDROXYETHYL CELLULOSE (100 MPA.S AT 2%) (UNII: R33S7TK2EP) LIMONENE, (+)- (UNII: GFD7C86Q1W) LINALOOL, (+/-)- (UNII: D81QY6I88E) PSEUDEVERNIA FURFURACEA (UNII: 49ZMN09Q0K) BENZYL SALICYLATE (UNII: WAO5MNK9TU) COUMARIN (UNII: A4VZ22K1WT) HYDROXYISOHEXYL 3-CYCLOHEXENE CARBOXALDEHYDE (UNII: QUE43B9Z2Q) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70064-002-01 50 mL in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part350 09/03/2015 Labeler - Geylanni International cosmetic developments LTD (533631474) Establishment Name Address ID/FEI Business Operations Bio Direct Ltd. 532814811 manufacture(70064-002)