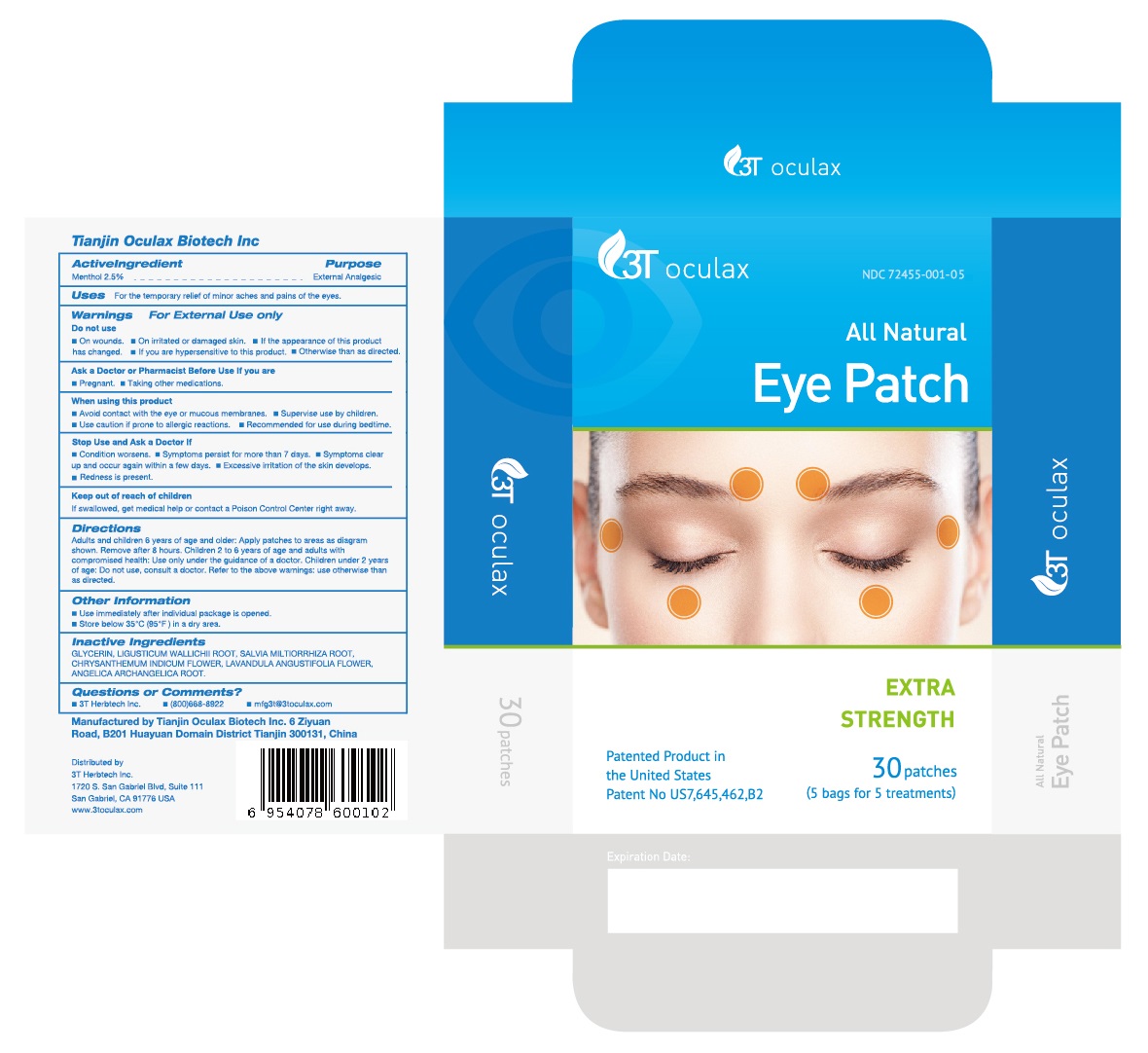
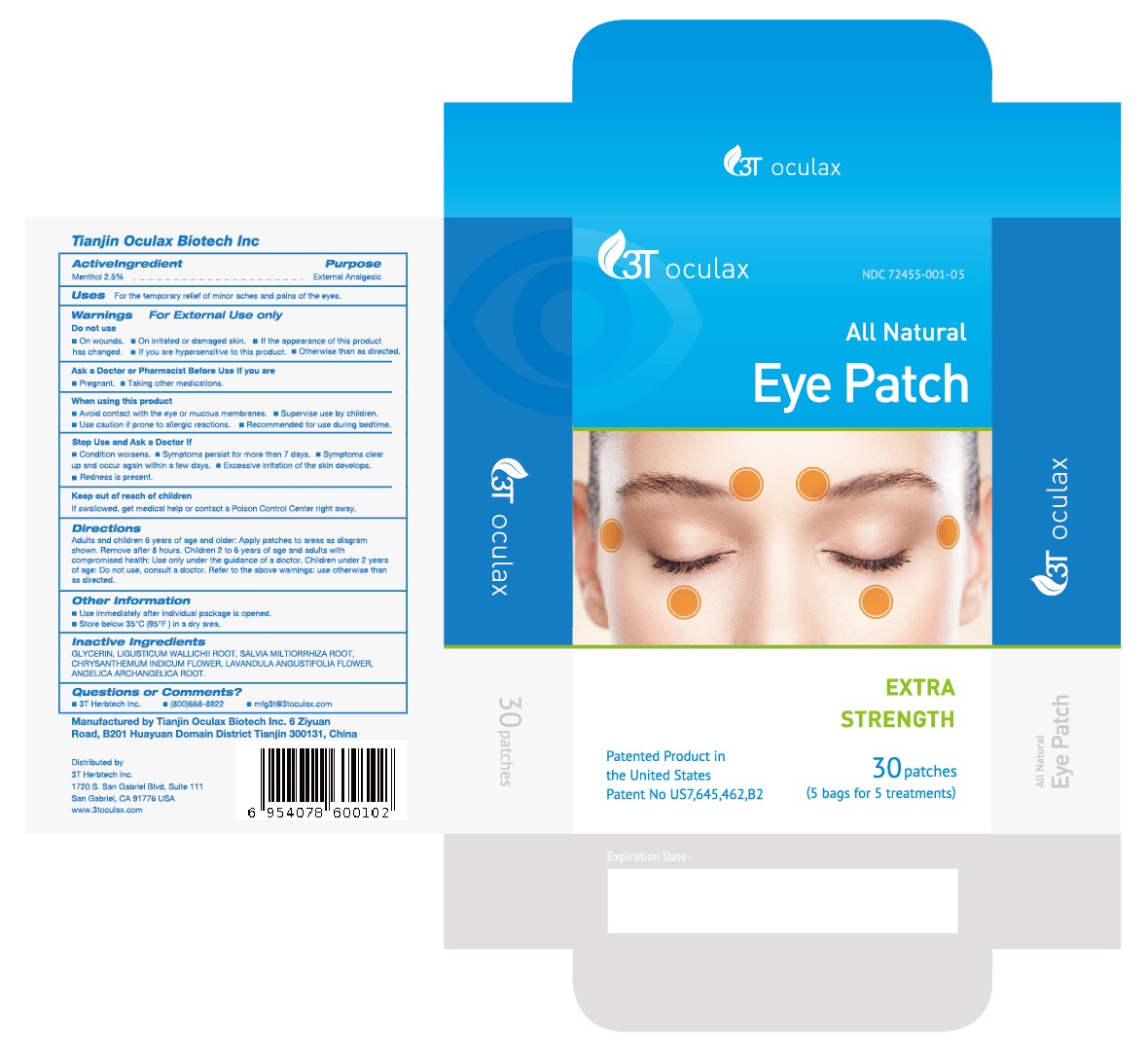
Label: ALL NATURAL EYE PATCH- menthol patch
- NDC Code(s): 72455-001-05, 72455-001-10, 72455-001-20
- Packager: Tianjin Oculax Biotech Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 26, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- USES
- Warnings
- Do not use
- Ask a Doctor or Pharmacist Before Use If you are
- When using this product
- Stop Use and Ask a Doctor If
- Keep out of reach of children.
-
Directions
Adults and children 6 years of age and older: Apply patches to areas as diagram shown. Remove after 8 hours. Children 2 to 6 years of age and adults with compromised health: Use only under the guidance of a doctor. Children under 2 years of age: Do not use, consult a doctor. Refer to the above warnings: use otherwise than as directed.
- Other Information
- Inactive Ingredients
- Questions or Comments?
- Manufactured byTianjin Oculax Biotech Inc 6 Ziyuan Road, B201 Huayuan Domain DistrictTianjin 300131, China
- Drug Facts
-
INGREDIENTS AND APPEARANCE
ALL NATURAL EYE PATCH
menthol patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72455-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 2.5 mg in 100 mg Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) LIGUSTICUM WALLICHII ROOT (UNII: R81AD159QS) SALVIA MILTIORRHIZA ROOT (UNII: 1693AM5SBN) CHRYSANTHEMUM INDICUM FLOWER (UNII: I6OER6U04L) LAVANDULA ANGUSTIFOLIA FLOWER (UNII: 19AH1RAF4M) ANGELICA ARCHANGELICA ROOT (UNII: DTN01M69SN) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72455-001-20 120 in 1 BOX 07/24/2018 1 NDC:72455-001-10 60 in 1 BOX 1 NDC:72455-001-05 30 in 1 BOX 1 24 mg in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 07/24/2018 Labeler - Tianjin Oculax Biotech Inc (542949666) Registrant - Tianjin Oculax Biotech Inc (542949666) Establishment Name Address ID/FEI Business Operations Tianjin Oculax Biotech Inc 542949666 manufacture(72455-001)