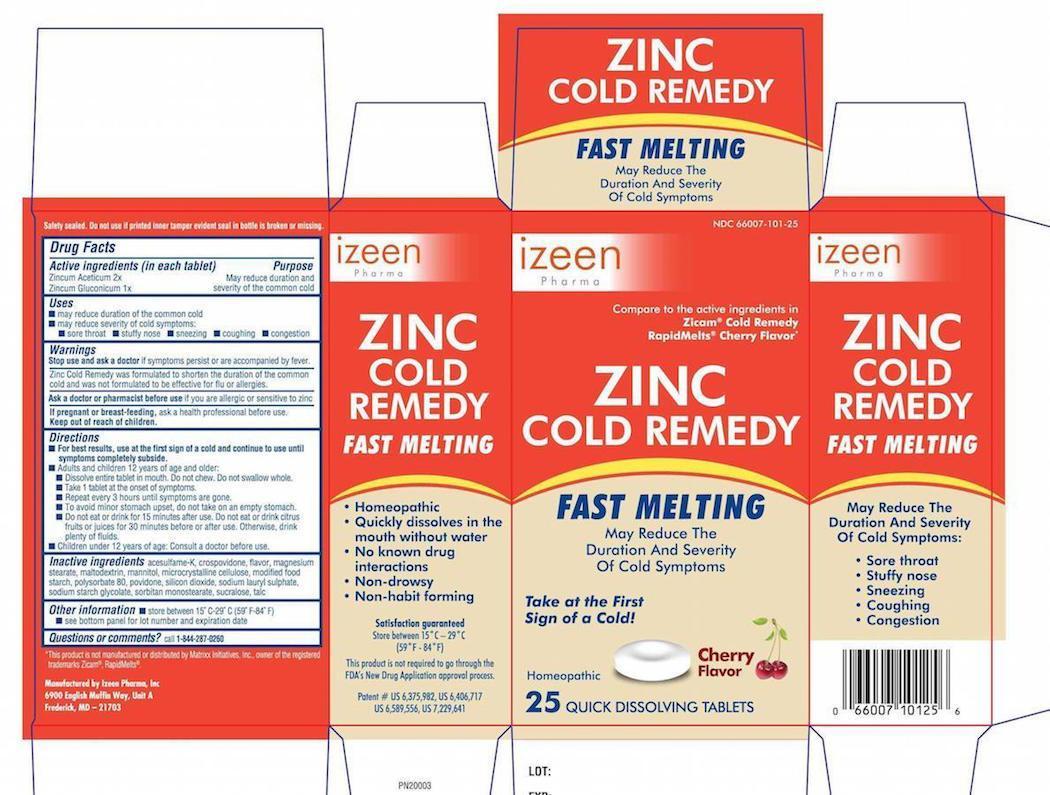
Label: ZINC COLD REMEDY FAST MELTING- zinc acetate and zinc gluconate tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 66007-101-25 - Packager: Izeen Pharma, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated February 9, 2015
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients (in each tablet)
- Purpose
- Uses
- Warnings
-
Directions
- for best results, use at the first sign of a cold and continue to use until symptoms completely subside
- adults and children 12 years of age and older:
- take 1 tablet at the onset of symptoms
- dissolve entire tablet in mouth. Do not chew. Do not swallow whole.
- repeat every 4-6 hours, not to exceed 4 tablets in 24 hours
- to avoid minor stomach upset, do not take on an empty stomach
- do not eat or drink for 15 minutes after use. Do not eat or drink citrus fruits or juices for 30 minutes before or after use. Otherwise, drink plenty of fluids.
- children under 12 years of age: ask a doctor before use
- Other information
- Inactive ingredients
- Questions/Comments
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL-25-Carton
-
INGREDIENTS AND APPEARANCE
ZINC COLD REMEDY FAST MELTING
zinc acetate and zinc gluconate tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:66007-101 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC ACETATE (UNII: FM5526K07A) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 2 [hp_X] ZINC GLUCONATE (UNII: U6WSN5SQ1Z) (ZINC CATION - UNII:13S1S8SF37) ZINC GLUCONATE 1 [hp_X] Inactive Ingredients Ingredient Name Strength SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) POLYSORBATE 80 (UNII: 6OZP39ZG8H) SODIUM LAURYL SULFATE (UNII: 368GB5141J) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) SORBITAN (UNII: 6O92ICV9RU) MODIFIED CORN STARCH (1-OCTENYL SUCCINIC ANHYDRIDE) (UNII: 461P5CJN6T) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) STEARIC ACID (UNII: 4ELV7Z65AP) SUCRALOSE (UNII: 96K6UQ3ZD4) TALC (UNII: 7SEV7J4R1U) ACESULFAME POTASSIUM (UNII: 23OV73Q5G9) Product Characteristics Color white Score no score Shape ROUND Size 15mm Flavor CHERRY Imprint Code CP115 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66007-101-25 1 in 1 CARTON 1 25 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved homeopathic 02/06/2015 Labeler - Izeen Pharma, Inc. (079300974) Establishment Name Address ID/FEI Business Operations Izeen Pharma, Inc. 079300974 MANUFACTURE(66007-101)