Label: TENA- dimethicone moisturizing lotion cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 59608-002-05, 59608-002-10, 59608-002-25 - Packager: SCA Personal Care Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 18, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
-
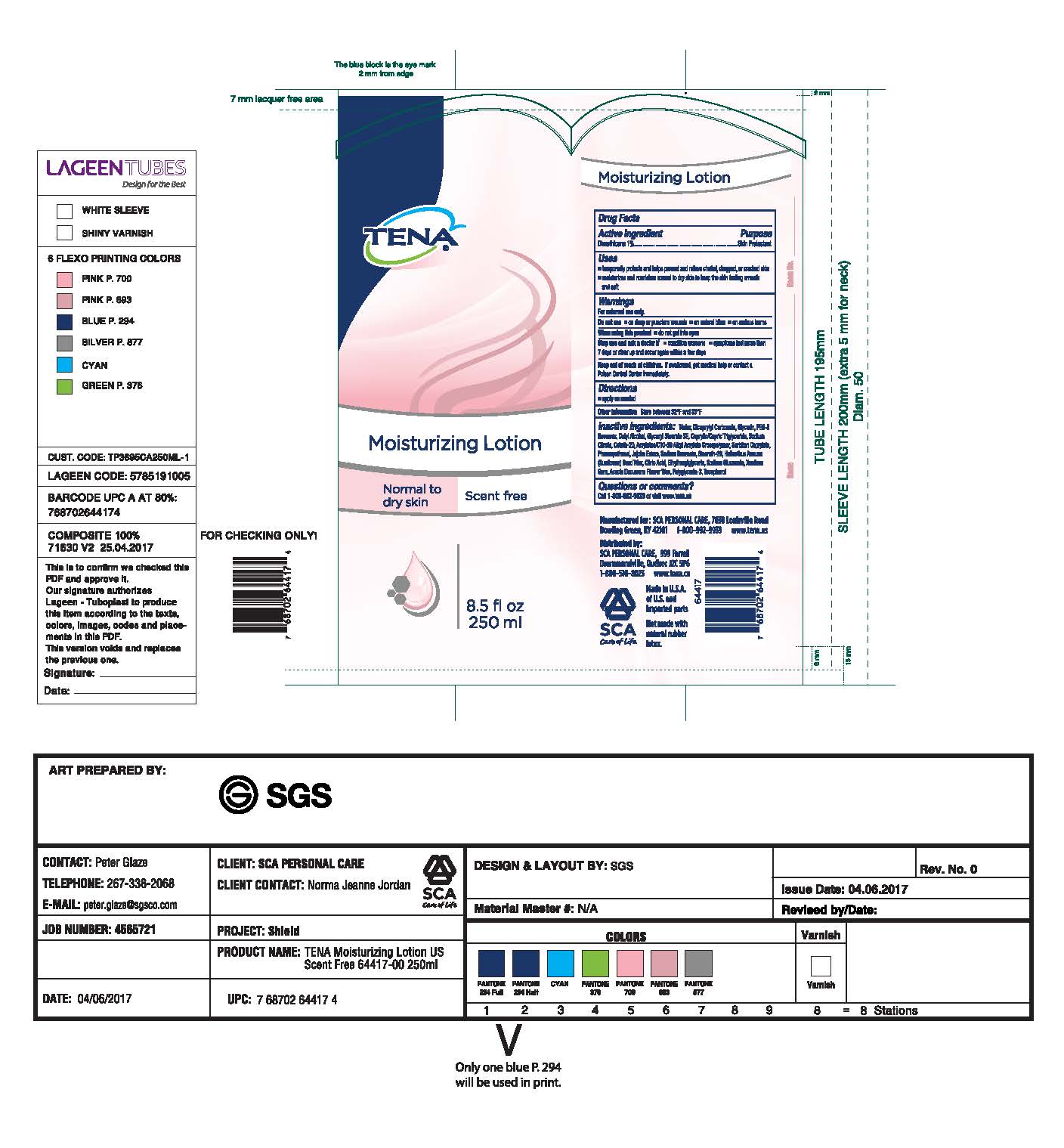
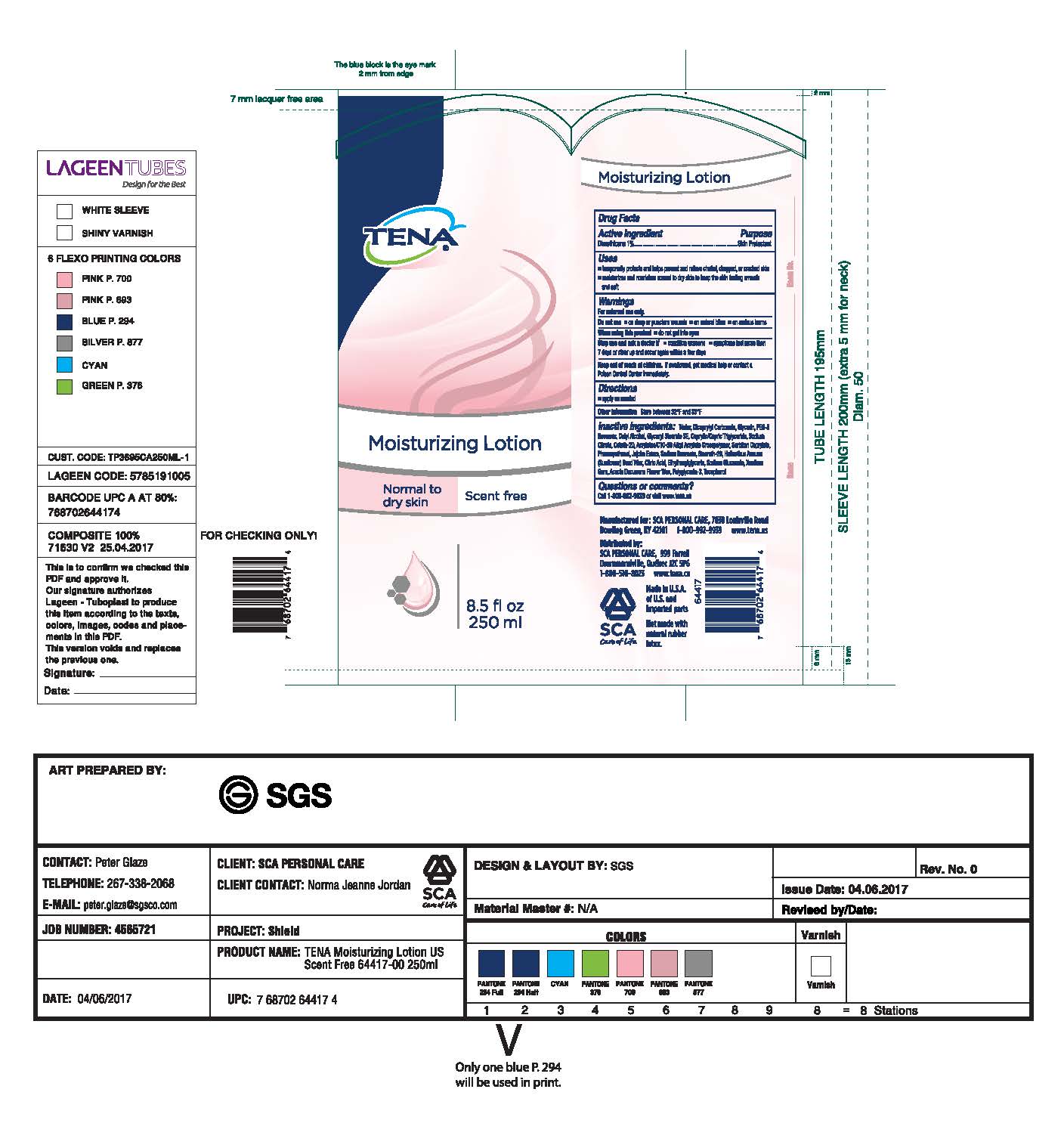
INACTIVE INGREDIENT
Inactive Ingredients: Water, Dicaprylyl Carbonate, Glycerin, PEG-8
Beeswax, Cetyl Alcohol, Glyceryl Stearate SE, Caprylic/Capric Triglyceride, Sodium
Citrate, Ceteth-20, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Sorbitan
Caprylate, Phenoxyethanol, Jojoba Esters, Sodium Benzoate, Steareth-20,
Helianthus Annus (Sunflower) Seed Wax, Citric Acid, Ethylhexylglycerin, Sodium
Gluconate, Xantham Gum, Acacia Decurrens Flower Wax, Polyglycerin-3, Tocopherol
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TENA
dimethicone moisturizing lotion creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59608-002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHICONE (UNII: 92RU3N3Y1O) (DIMETHICONE - UNII:92RU3N3Y1O) DIMETHICONE .01 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) GLYCERIN (UNII: PDC6A3C0OX) YELLOW WAX (UNII: 2ZA36H0S2V) CETYL ALCOHOL (UNII: 936JST6JCN) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) SODIUM CITRATE (UNII: 1Q73Q2JULR) CETETH-20 (UNII: I835H2IHHX) CARBOMER INTERPOLYMER TYPE A (ALLYL SUCROSE CROSSLINKED) (UNII: 59TL3WG5CO) SORBITAN (UNII: 6O92ICV9RU) PHENOXYETHANOL (UNII: HIE492ZZ3T) JOJOBA OIL, RANDOMIZED (UNII: 7F0EV20QYL) SODIUM BENZOATE (UNII: OJ245FE5EU) STEARETH-20 (UNII: L0Q8IK9E08) HELIANTHUS ANNUUS SEED WAX (UNII: 42DG15CHXV) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) SODIUM GLUCONATE (UNII: R6Q3791S76) XANTHAN GUM (UNII: TTV12P4NEE) ACACIA DECURRENS FLOWER WAX (UNII: AU6XZE9IY9) POLYGLYCERIN-3 (UNII: 4A0NCJ6RD6) TOCOPHEROL (UNII: R0ZB2556P8) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59608-002-10 100 mL in 1 TUBE; Type 0: Not a Combination Product 05/28/2017 2 NDC:59608-002-25 250 mL in 1 TUBE; Type 0: Not a Combination Product 05/28/2017 3 NDC:59608-002-05 5 mL in 1 TUBE; Type 0: Not a Combination Product 05/28/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 05/28/2017 Labeler - SCA Personal Care Inc. (790100549)