SENNA- S- docusate sodium and sennosides tablet
Western Family Foods, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
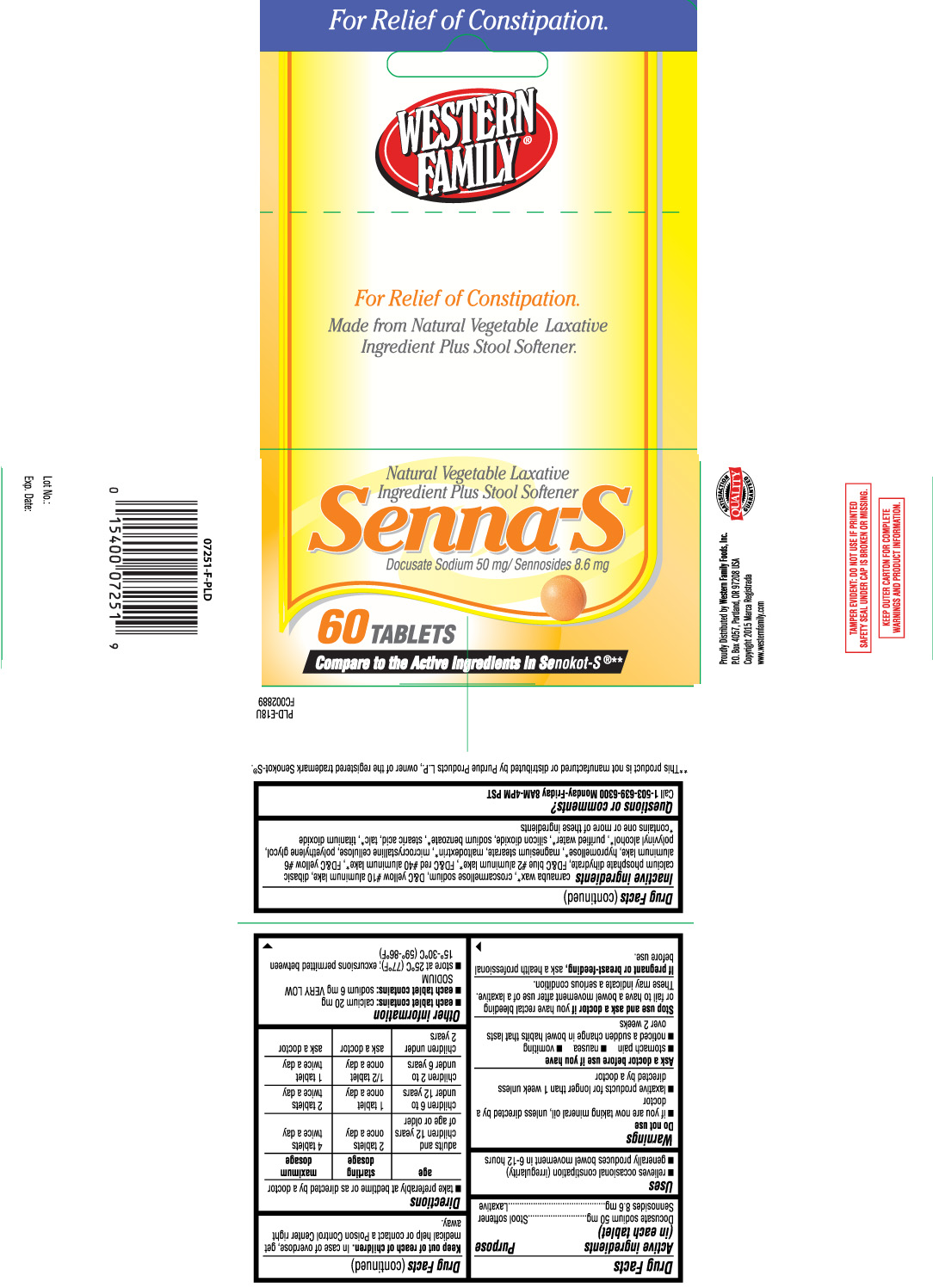
Drug Facts
Uses
- relieves occasional constipation (irregularity)
- generally produces a bowel movement in 6-12 hours
Warnings
Do not use
- if you are now taking mineral oil, unless directed by a doctor
- laxative products for longer than 1 week unless directed by a doctor
Ask a doctor before use if you have
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that lasts over 2 weeks
Directions
- take preferably at bedtime or as directed by a doctor
| age | starting dose | maximum dosage |
| adults and children 12 years of age or older | 2 tablets once a day | 4 tablets twice a day |
| children 6 to under 12 years | 1 tablet once a day | 2 tablets twice a day |
| children 2 to under 6 years | 1/2 tablet once a day | 1 tablet twice a day |
| children under 2 years | ask a doctor | ask a doctor |
Other information
- each tablet contains: calcium 20 mg
- each tablet contains: sodium 6 mg VERY LOW SODIUM
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
Inactive ingredients
carnauba wax*, croscarmellose sodium, D&C yellow #10 aluminum lake, dibasic calcium phosphate dihydrate, FD&C blue #2 aluminum lake*, FD&C red #40 aluminum lake*, FD&C yellow #6 aluminum lake, hypromellose*, magnesium stearate, maltodextrin*, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol*, purified water*, silicon dioxide, sodium benzoate*, stearic acid, talc*, titanium dioxide
*contains one or more of these ingredients
Principal Display Panel
For Relief of Constipation
Made from Natural Vegetable Laxative Ingredient Plus Stool Softener,
Senna-S
Docusate Sodium 50 mg / Sennosides 8.6 mg
TABLETS
Compare to the Active Ingredients in Senokot-S®**
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
**This product is not manufactured or distributed by Purdue Products L.P., owner of the registered trademark Senokot-S®.
Proudly Distributed by Western Family Foods, Inc.
P.O. Box 4057, Portland, OR 97208 USA
| SENNA- S
docusate sodium and sennosides tablet |
||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Western Family Foods, Inc. (192166072) |
| Registrant - P & L Development, LLC (800014821) |