Label: CHILDRENS LORATADINE SUGAR FREE- loratadine solution
- NDC Code(s): 63941-131-08
- Packager: Valu Merchandisers Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated December 15, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each 5 mL teaspoonful)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product do not take more than directed. Taking more than directed may cause drowsiness.
-
Directions
- use only with enclosed dosing cup
adults and children 6 years and over 2 teaspoonfuls (tsp) daily; do not take more than 2 teaspoonfuls (tsp) in 24 hours children 2 to under 6 years of age 1 teaspoonful (tsp) daily; do not take more than 1 teaspoonful (tsp) in 24 hours children under 2 years of age ask a doctor consumers with liver or kidney disease ask a doctor - Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
-
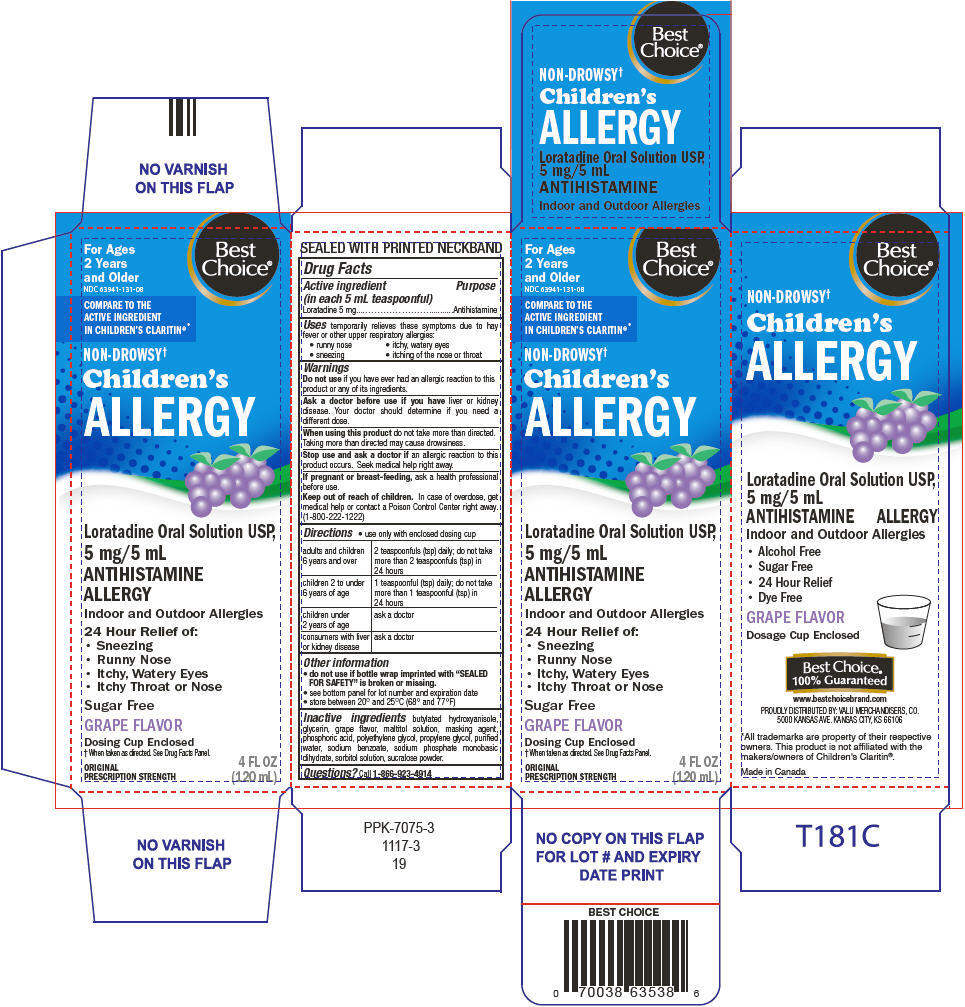
PRINCIPAL DISPLAY PANEL - 120 mL Bottle Carton
For Ages
2 Years
and OlderNDC 63941-131-08
Best
Choice®COMPARE TO THE
ACTIVE INGREDIENT
IN CHILDREN'S CLARITIN®*NON-DROWSY†
Children's
ALLERGYLoratadine Oral Solution USP,
5 mg/5 mL
ANTIHISTAMINE
ALLERGYIndoor and Outdoor Allergies
24 Hour Relief of:
- Sneezing
- Runny Nose
- Itchy, Watery Eyes
- Itchy Throat or Nose
Sugar Free
GRAPE FLAVOR
Dosing Cup Enclosed
† When taken as directed. See Drug Facts Panel.
ORIGINAL
PRESCRIPTION STRENGTH4 FL OZ
(120 mL)
-
INGREDIENTS AND APPEARANCE
CHILDRENS LORATADINE SUGAR FREE
loratadine solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63941-131 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Loratadine (UNII: 7AJO3BO7QN) (Loratadine - UNII:7AJO3BO7QN) Loratadine 5 mg in 5 mL Inactive Ingredients Ingredient Name Strength butylated hydroxyanisole (UNII: REK4960K2U) glycerin (UNII: PDC6A3C0OX) SORBITOL (UNII: 506T60A25R) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) phosphoric acid (UNII: E4GA8884NN) propylene glycol (UNII: 6DC9Q167V3) water (UNII: 059QF0KO0R) sodium benzoate (UNII: OJ245FE5EU) SODIUM PHOSPHATE, MONOBASIC, DIHYDRATE (UNII: 5QWK665956) Product Characteristics Color YELLOW (colorless to slightly yellow) Score Shape Size Flavor GRAPE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63941-131-08 1 in 1 CARTON 11/15/2017 1 120 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076805 03/17/2017 Labeler - Valu Merchandisers Company (868703513)