Label: LANTHANUM CARBONATE tablet, chewable
-
NDC Code(s):
66993-422-47,
66993-422-85,
66993-423-53,
66993-423-85, view more66993-424-75, 66993-424-85
- Packager: Prasco Laboratories
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application Authorized Generic
Drug Label Information
Updated December 26, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LANTHANUM CARBONATE safely and effectively. See full prescribing information for LANTHANUM CARBONATE.
LANTHANUM CARBONATE chewable tablets, for oral use
Initial U.S. Approval: 2004INDICATIONS AND USAGE
- LANTHANUM CARBONATE is a phosphate binder indicated to reduce serum phosphate in patients with end-stage renal disease (ESRD). (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
- LANTHANUM CARBONATE Chewable Tablets: 500 mg, 750 mg, and 1,000 mg. (3)
CONTRAINDICATIONS
- Bowel obstruction, ileus, and fecal impaction. (4)
WARNINGS AND PRECAUTIONS
- Serious cases of gastrointestinal obstruction, ileus, subileus, gastrointestinal perforation, and fecal impaction. Risks include altered gastrointestinal anatomy, hypomotility disorders, and concomitant medications. Advise patients to chew or crush the tablet completely. (5.1)
- LANTHANUM CARBONATE has radio-opaque properties and, therefore, may give the appearance typical of an imaging agent during abdominal X-ray procedures. (5.2)
ADVERSE REACTIONS
- In controlled trials, the most common adverse reactions that were more frequent (≥5% difference vs. placebo) in LANTHANUM CARBONATE were nausea, vomiting, and abdominal pain. (6.1)
- The following adverse reactions have been identified during post-approval use of LANTHANUM CARBONATE: constipation, dyspepsia, allergic skin reactions, and tooth injury while chewing the tablet. (6.2)
To report SUSPECTED ADVERSE REACTIONS, contact Takeda Pharmaceuticals at 1-800-828-2088 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- There is a potential for LANTHANUM CARBONATE to interact with compounds that bind to cationic antacids (i.e., aluminum-, magnesium-, or calcium-based); therefore, do not take such compounds within 2 hours of dosing with LANTHANUM CARBONATE. (7.1)
- Oral quinolone antibiotics must be taken at least 1 hour before or 4 hours after LANTHANUM CARBONATE. (7.2)
- Do not take thyroid hormone replacement therapy within 2 hours of dosing with LANTHANUM CARBONATE. Monitoring of TSH levels is recommended in patients receiving both medicinal agents. (7.3)
- For oral medications where a reduction in the bioavailability of that medication would have a clinically significant effect on its safety or efficacy, consider separation of the timing of the administration of the two drugs. (7.4)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 12/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Gastrointestinal Adverse Effects
5.2 Diagnostic Tests
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Drugs Binding to Antacids
7.2 Quinolone Antibiotics
7.3 Levothyroxine
7.4 Use with Other Oral Medications
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Double-Blind Placebo-Controlled Studies
14.2 Open-Label Active-Controlled Studies
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
LANTHANUM CARBONATE is a phosphate binder indicated to reduce serum phosphate in patients with end-stage renal disease (ESRD).
Management of elevated serum phosphorus levels in patients with ESRD usually includes all of the following: reduction in dietary intake of phosphate, removal of phosphate by dialysis, and reduction of intestinal phosphate absorption with phosphate binders.
-
2 DOSAGE AND ADMINISTRATION
Divide the total daily dose of LANTHANUM CARBONATE and take with or immediately after meals. The recommended initial total daily dose of LANTHANUM CARBONATE is 1,500 mg. Titrate the dose every 2 to 3 weeks until an acceptable serum phosphate level is reached. Monitor serum phosphate levels as needed during dose titration and on a regular basis thereafter.
LANTHANUM CARBONATE has the potential to bind other orally administered drugs; consider separating the administration of other oral medications [see Drug Interactions (7)].
In clinical studies of patients with ESRD, LANTHANUM CARBONATE doses up to 4,500 mg were evaluated. Most patients required a total daily dose between 1,500 mg and 3,000 mg to reduce plasma phosphate levels to less than 6.0 mg/dL. Doses were generally titrated in increments of 750 mg/day.
Chew or crush LANTHANUM CARBONATE completely before swallowing. Do not swallow intact LANTHANUM CARBONATE Chewable Tablets.
Consider using the oral powder formulation in patients with poor dentition or who have difficulty chewing tablets.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Gastrointestinal Adverse Effects
Serious cases of gastrointestinal obstruction, ileus, subileus, gastrointestinal perforation, and fecal impaction have been reported in patients taking lanthanum, some requiring surgery or hospitalization. Consider discontinuing LANTHANUM CARBONATE in patients without another explanation for severe gastrointestinal symptoms.
Risk factors for gastrointestinal obstruction and gastrointestinal perforation identified from post-marketing reports in patients taking LANTHANUM CARBONATE Chewable Tablets include abnormal gastrointestinal anatomy (e.g., diverticular disease, peritonitis, history of gastrointestinal surgery, gastrointestinal cancer, gastrointestinal ulceration), hypomotility disorders (e.g., constipation, ileus, subileus, diabetic gastroparesis), and the use of medications known to potentiate these effects. Some cases were reported in patients with no history of gastrointestinal disease.
Patients with acute peptic ulcer, ulcerative colitis, Crohn's disease, or bowel obstruction were not included in LANTHANUM CARBONATE clinical studies [see Contraindications (4)].
Advise patients who are prescribed LANTHANUM CARBONATE Chewable Tablets to chew the tablet completely and not to swallow them whole. Serious gastrointestinal complications have been reported in association with unchewed or incompletely chewed tablets. [see Adverse Reactions (6.2).
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Gastrointestinal Adverse Effects [see Warnings and Precautions (5.1)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Overall, the safety profile of LANTHANUM CARBONATE has been studied in over 5,200 subjects in completed clinical trials. The most common adverse reactions for LANTHANUM CARBONATE were gastrointestinal events, such as nausea, vomiting, and abdominal pain and they generally abated over time with continued dosing.
In double-blind, placebo-controlled studies where a total of 180 and 95 patients with ESRD were randomized to LANTHANUM CARBONATE chewable tablet and placebo, respectively, for 4 to 6 weeks of treatment, the most common reactions that were more frequent (≥5% difference) in the LANTHANUM CARBONATE group were nausea, vomiting, and abdominal pain (Table 1).
Table 1. Adverse Reactions* That Were More Common on LANTHANUM CARBONATE in Placebo-Controlled, Double-Blind Studies with Treatment Periods of 4 to 6 Weeks LANTHANUM CARBONATE
%
(N=180)Placebo
%
(N=95)- *
- Expressed as the event rate for each term
Nausea 11 5 Vomiting 9 4 Abdominal pain 5 0 In an open-label, long-term 2-year extension study in 93 patients who had transitioned from other studies, resulting in a total of up to 6 years treatment, mean baseline values and changes in transaminases were similar to those observed in the earlier comparative studies, with little change during treatment.
The safety of LANTHANUM CARBONATE was studied in two long-term, open-label clinical trials, which included 1,215 patients treated with LANTHANUM CARBONATE and 944 with alternative therapy. Fourteen percent (14%) of patients treated with LANTHANUM CARBONATE discontinued treatment due to adverse events. Gastrointestinal adverse reactions, such as nausea, diarrhea, and vomiting were the most common types of event leading to discontinuation.
In pooled active comparator controlled clinical trials, hypocalcemia was noted with an incidence of approximately 5% in both lanthanum and active comparator groups. A nonclinical study and a phase 1 study have shown reduced absorption of calcium in the intestine with lanthanum carbonate treatment.
In a crossover study in 72 healthy individuals comparing LANTHANUM CARBONATE Chewable Tablets to LANTHANUM CARBONATE Oral Powder, gastrointestinal adverse reactions such as nausea, diarrhea, and vomiting were more common for the oral powder formulation (18%) than for the chewable tablets (7%).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of LANTHANUM CARBONATE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cases of constipation, intestinal perforation, intestinal obstruction, ileus, subileus, dyspepsia, allergic skin reactions, hypophosphatemia, and tooth injury while chewing the tablet have been reported.
-
7 DRUG INTERACTIONS
7.1 Drugs Binding to Antacids
There is a potential for LANTHANUM CARBONATE to interact with compounds which bind to cationic antacids (i.e., aluminum-, magnesium-, or calcium-based); therefore, do not administer such compounds within 2 hours of dosing with LANTHANUM CARBONATE. Examples of relevant classes of compounds where antacids have been demonstrated to reduce bioavailability include antibiotics (such as quinolones, ampicillin, and tetracyclines), thyroid hormones, ACE inhibitors, statin lipid regulators, and anti-malarials.
7.2 Quinolone Antibiotics
Co-administration of LANTHANUM CARBONATE with quinolone antibiotics may reduce the extent of their absorption. The bioavailability of oral ciprofloxacin was decreased by approximately 50% when taken with LANTHANUM CARBONATE in a single-dose study in healthy volunteers. Administer oral quinolone antibiotics at least 1 hour before or 4 hours after LANTHANUM CARBONATE. When oral quinolones are given for short courses, consider eliminating the doses of LANTHANUM CARBONATE that would normally be scheduled near the time of quinolone intake to improve quinolone absorption [see Clinical Pharmacology (12.3)].
7.3 Levothyroxine
The bioavailability of levothyroxine was decreased by approximately 40% when taken together with LANTHANUM CARBONATE. Administer thyroid hormone replacement therapy at least 2 hours before or 2 hours after dosing with LANTHANUM CARBONATE and monitor thyroid stimulating hormone (TSH) levels [see Clinical Pharmacology (12.3)].
7.4 Use with Other Oral Medications
There are no empirical data on avoiding drug interactions between LANTHANUM CARBONATE and most concomitant oral drugs. For oral medications where a reduction in the bioavailability of that medication would have a clinically significant effect on its safety or efficacy, consider separation of the timing of the administration of the two drugs. The duration of separation depends upon the absorption characteristics of the medication concomitantly administered, such as the time to reach peak systemic levels and whether the drug is an immediate-release or an extended-release product. Consider monitoring clinical responses or blood levels of concomitant medications that have a narrow therapeutic range.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data from case reports with use of LANTHANUM CARBONATE in pregnant women are insufficient to identify a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. In animal reproduction studies, oral administration of lanthanum carbonate to pregnant rats and rabbits during organogenesis at doses 3 and 2.5 times, respectively, the maximum recommended human dose (MRHD), resulted in no adverse developmental effects. In rabbits, lanthanum carbonate doses 5 times the MRHD was associated with maternal toxicity and resulted in increased post-implantation loss, reduced fetal weights, and delayed fetal ossification (see Data). Deposition of lanthanum into developing bone, including growth plate, was observed in juvenile animals in long-term animal studies with lanthanum carbonate [see Use in Specific Populations (8.4)]. Use a non-lanthanum containing phosphate binder in a pregnant woman.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defect and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In pregnant rats, oral administration of lanthanum carbonate at doses as high as 2,000 mg/kg/day during organogenesis resulted in no evidence of harm to the fetus. The MRHD for LANTHANUM CARBONATE is 5,725 mg, representing a dose of 95.4 mg/kg, or 3,530 mg/m2 for a 60-kg patient. The 2,000-mg/kg/day dose in the rat is equivalent to 12,000 mg/m2, 3 times the MRHD. In pregnant rabbits, oral administration of lanthanum carbonate at 1,500 mg/kg/day (18,000 mg/m2; 5 times the daily MRHD) during organogenesis was associated with increased postimplantation loss, reduced fetal weights, and delayed fetal ossification. No effects on the pregnant rabbits or fetuses were observed at 750 mg/kg/day (9,000 mg/m2; 2.5 times the MRHD).
In a pre- and postnatal development study in the rat, pregnant rats were dosed at up to 2,000 mg/kg/day (12,000 mg/m2/day; equivalent to 3 times the MRHD) from day 6 of pregnancy through 20 days postpartum (including lactation). At 2,000 mg/kg/day, no maternal toxicity was observed, nor were any changes seen with respect to gestational length or delivery; however, piloerection/pallor, delayed eye opening, decreased body weight, and delayed sexual development were observed in the offspring at 2,000 mg/kg/day. At 200 and 600 mg/kg/day (equivalent to 0.3 and 1 time the MRHD, respectively), slight delays in sexual development (delayed vaginal opening) were observed in the female offspring [see Nonclinical Toxicology (13.2)].
8.2 Lactation
Risk Summary
There are no data on the presence of lanthanum carbonate from LANTHANUM CARBONATE in human milk, the effects on the breastfed infant, or the effects on milk production. Deposition of lanthanum into developing bone, including growth plate, was observed in juvenile animals in long-term animal studies with lanthanum carbonate [see Use in Specific Populations (8.4)]. Use a non-lanthanum containing phosphate binder in a lactating woman.
8.4 Pediatric Use
The safety and efficacy of LANTHANUM CARBONATE in pediatric patients have not been established. While growth abnormalities were not identified in long-term animal studies, lanthanum was deposited into developing bone, including growth plate. The consequences of such deposition in developing bone in pediatric patients are unknown; therefore, the use of LANTHANUM CARBONATE in this population is not recommended.
-
10 OVERDOSAGE
The symptoms associated with overdose are adverse reactions such as headache, nausea and vomiting. In clinical trials in healthy adults, gastrointestinal (GI) symptoms were reported with daily doses up to 6,000 mg/day of lanthanum carbonate administered with food. Given the topical activity of lanthanum in the gut, and the excretion of the majority of the dose in feces, supportive therapy is recommended for overdosage. Lanthanum carbonate was not acutely toxic in animals by the oral route. No deaths and no adverse effects occurred in mice, rats, or dogs after single oral doses of 2,000 mg/kg (1.7, 3.4, and 11.3 times the MRHD, respectively, on a mg/m2 basis).
-
11 DESCRIPTION
LANTHANUM CARBONATE contains lanthanum carbonate with molecular formula La2(CO3)3 xH2O (on average x=4-5 moles of water) and molecular weight 457.8 (anhydrous mass). Lanthanum carbonate is described as white to almost-white powder. Lanthanum carbonate is practically insoluble in water and is insoluble in organic solvents; it dissolves in dilute mineral acids with effervescence.
Each LANTHANUM CARBONATE white to off-white, chewable tablet contains lanthanum carbonate hydrate equivalent to 500, 750, or 1,000 mg of elemental lanthanum and the following inactive ingredients: colloidal silicon dioxide, dextrates (hydrated), magnesium stearate.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
LANTHANUM CARBONATE is a phosphate binder that reduces absorption of phosphate by forming insoluble lanthanum phosphate complexes that pass through the GI tract unabsorbed. Both serum phosphate and calcium phosphate product are reduced as a consequence of the reduced dietary phosphate absorption.
12.2 Pharmacodynamics
In vitro studies have shown that lanthanum binds phosphate in the physiologically relevant pH range of 3 to 7. In simulated gastric fluid, lanthanum binds approximately 97% of the available phosphate at pH 3-5 and 67% at pH 7, when lanthanum is present in a two-fold molar excess to phosphate. Bile acids have not been shown to affect the phosphate binding affinity of lanthanum. In order to bind dietary phosphate, LANTHANUM CARBONATE must be administered with or immediately after meals.
In five phase 1 pharmacodynamic studies comparing the reduction from baseline of urinary phosphorus excretion in healthy volunteers (N=143 taking lanthanum carbonate), it was shown that the mean intestinal phosphate binding capacity of lanthanum ranged from 235 to 468 mg phosphorus/day when lanthanum was administered at a dose of 3 g per day with food. By comparison, in one study with an untreated control group (n=10) and another study with a placebo group (n=3), the corresponding mean changes from baseline were 3 mg phosphorus/day and 87 mg phosphorus/day, respectively.
In healthy subjects, LANTHANUM CARBONATE Oral Powder and LANTHANUM CARBONATE Chewable Tablets produce similar effects on urinary phosphate excretion.
12.3 Pharmacokinetics
Absorption and Distribution - Following single- or multiple-dose oral administration of LANTHANUM CARBONATE to healthy subjects, the concentration of lanthanum in plasma was very low (bioavailability <0.002%). Following oral administration in patients, the mean lanthanum Cmax was 1.0 ng/mL. During long-term administration (52 weeks) in patients with ESRD, the mean lanthanum concentration in plasma was approximately 0.6 ng/mL. There was a minimal increase in plasma lanthanum concentrations with increasing doses within the therapeutic dose range. The timing of food intake relative to lanthanum administration (during and 30 minutes after food intake) has a negligible effect on the systemic level of lanthanum.
Systemic exposure to lanthanum was approximately 30% higher following administration of LANTHANUM CARBONATE Oral Powder when compared to LANTHANUM CARBONATE Chewable Tablets. However, systemic exposure to lanthanum from both formulations in this study was within the range seen in previous pharmacokinetic studies of Chewable Tablets in healthy individuals.
In vitro, lanthanum is highly bound (>99%) to human plasma proteins, including human serum albumin, α1-acid glycoprotein, and transferrin. Binding to erythrocytes in vivo is negligible in rats.
In animal studies, lanthanum concentrations in several tissues, particularly gastrointestinal tract, mesenteric lymph nodes, bone, and liver, increased over time to levels several orders of magnitude higher than those in plasma. The level of lanthanum in the liver was higher in renally impaired rats due to higher intestinal absorption. Lanthanum was found in the lysosomes and the biliary canal consistent with transcellular transport. Steady state tissue concentrations in bone and liver were achieved in dogs between 4 and 26 weeks. Relatively high levels of lanthanum remained in these tissues for longer than 6 months after cessation of dosing in dogs. There is no evidence from animal studies that lanthanum crosses the blood-brain barrier.
In 105 bone biopsies from patients treated with LANTHANUM CARBONATE for up to 4.5 years, rising levels of lanthanum were noted over time. Estimates of elimination half-life from bone ranged from 2.0 to 3.6 years. Steady state bone concentrations were not reached during the period studied.
Metabolism and Elimination - Lanthanum is not metabolized. Lanthanum was cleared from plasma of patients undergoing dialysis with an elimination half-life of 53 hours following discontinuation of therapy.
No information is available regarding the mass balance of lanthanum in humans after oral administration. In rats and dogs, the mean recovery of lanthanum after an oral dose was about 99% and 94%, respectively, and was essentially all from feces. Biliary excretion is the predominant route of elimination for circulating lanthanum in rats. In healthy volunteers administered intravenous (IV) lanthanum as the soluble chloride salt (120 mcg), renal clearance was less than 2% of total plasma clearance.
Drug Interactions
LANTHANUM CARBONATE has a low potential for systemic drug-drug interactions because of the very low bioavailability of lanthanum and because it is not a substrate or inhibitor of major cytochrome P450 enzyme groups involved in drug metabolism (CYP1A2, CYP2C9/10, CYP2C19, CYP2D6, and CYP3A4/5).
LANTHANUM CARBONATE does not alter gastric pH; therefore, LANTHANUM CARBONATE drug interactions based on altered gastric pH are not expected.
In an in vitro investigation, lanthanum did not form insoluble complexes when mixed in simulated gastric fluid with warfarin, digoxin, furosemide, phenytoin, metoprolol, and enalapril. Clinical studies have shown that LANTHANUM CARBONATE (three doses of 1,000 mg on the day prior to exposure and one dose of 1,000 mg on the day of co-administration) administered 30 minutes earlier did not alter the pharmacokinetics of oral warfarin (10 mg), digoxin (0.5 mg), or metoprolol (100 mg). Potential pharmacodynamic interactions between lanthanum and these drugs (e.g., bleeding time or prothrombin time) were not evaluated. None of the drug interaction studies were done with the maximum recommended therapeutic dose of lanthanum carbonate. No drug interaction studies assessed the effects of drugs on phosphate binding by lanthanum carbonate.
Ciprofloxacin
In a randomized, two–way crossover study in healthy volunteers examining the interaction potential of a single oral dose of ciprofloxacin (750 mg) alone and with lanthanum carbonate (1 g three times a day), the maximum plasma concentration of ciprofloxacin was reduced by 56% and the area under the ciprofloxacin plasma concentration-time curve was reduced by 54%. The 24-hour urinary recovery of ciprofloxacin was reduced 52% by LANTHANUM CARBONATE [see Drug Interactions (7.2)].
Levothyroxine
In a single-dose crossover study of levothyroxine (1 mg) with or without simultaneous administration of a single dose of LANTHANUM CARBONATE (500 mg) in six euthyroid normal healthy volunteers, the area under the serum T4 concentration-time curve was decreased by 40% [see Drug Interactions (7.3)].
Fat-Soluble Vitamins
LANTHANUM CARBONATE appears not to affect the availability of fat-soluble vitamins (A, D, E, and K) or other nutrients [see Clinical Studies (14.2)].
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Oral administration of lanthanum carbonate to rats for up to 104 weeks, at doses up to 1,500 mg of the salt per kg/day (2.5 times the MRHD of 5,725 mg, on a mg/m2 basis, assuming a 60-kg patient) revealed no evidence of carcinogenic potential. In the mouse, oral administration of lanthanum carbonate for up to 99 weeks, at a dose of 1,500 mg/kg/day (1.3 times the MRHD) was associated with an increased incidence of glandular stomach adenomas in male mice.
Lanthanum carbonate tested negative for mutagenic activity in an in vitro Ames assay using Salmonella typhimurium and Escherichia coli strains and in vitro HGPRT gene mutation and chromosomal aberration assays in Chinese hamster ovary cells. Lanthanum carbonate also tested negative in an oral mouse micronucleus assay at doses up to 2,000 mg/kg (1.7 times the MRHD), and in micronucleus and unscheduled DNA synthesis assays in rats given IV lanthanum chloride at doses up to 0.1 mg/kg, a dose that produced plasma lanthanum concentrations greater than 2,000 times the peak human plasma concentration.
Lanthanum carbonate, at doses up to 2,000 mg/kg/day (3.4 times the MRHD), did not affect fertility or mating performance of male or female rats.
13.2 Animal Toxicology and/or Pharmacology
In pregnant rats, oral administration of lanthanum carbonate at doses as high as 2,000 mg/kg/day (3.4 times the MRHD) resulted in no evidence of harm to the fetus. In pregnant rabbits, oral administration of lanthanum carbonate at 1,500 mg/kg/day (5 times the MRHD) was associated with a reduction in maternal body weight gain and food consumption, increased post-implantation loss, reduced fetal weights, and delayed fetal ossification. No effects on pregnant rabbits or fetuses were observed at 750 mg/kg/day (2.5 times the MRHD). Lanthanum carbonate administered to rats from implantation through lactation at 2,000 mg/kg/day (3.4 times the MRHD) caused delayed eye opening, reduction in body weight gain, and delayed sexual development (preputial separation and vaginal opening) of the offspring. At 200 and 600 mg/kg/day (equivalent to 0.3 and 1 time the MRHD, respectively), slight delays in vaginal opening were observed in the female offspring.
-
14 CLINICAL STUDIES
The effectiveness of LANTHANUM CARBONATE in reducing serum phosphorus in patients with ESRD was demonstrated in one short-term, placebo-controlled, double-blind dose-ranging study; two placebo-controlled, randomized withdrawal studies; and two long-term, active-controlled, open-label studies in patients undergoing either hemodialysis or peritoneal dialysis.
14.1 Double-Blind Placebo-Controlled Studies
One-hundred and forty-four patients with chronic renal failure undergoing hemodialysis and with elevated phosphate levels were randomized to double-blind treatment at a fixed dose of lanthanum carbonate of 225 mg (n=27), 675 mg (n=29), 1,350 mg (n=30), or 2,250 mg (n=26) or placebo (n=32) in divided doses with meals. Fifty-five percent of subjects were male, 71% black, 25% white, and 4% of other races. The mean age was 56 years and the duration of dialysis ranged from 0.5 to 15.3 years. Steady-state effects were achieved after two weeks. The effect after six weeks of treatment is shown in Figure 1.
Figure 1. Difference in Phosphate Reduction in the LANTHANUM CARBONATE and Placebo Group in a 6-Week, Dose-Ranging, Double-Blind Study in Patients with ESRD (with 95% Confidence Intervals)
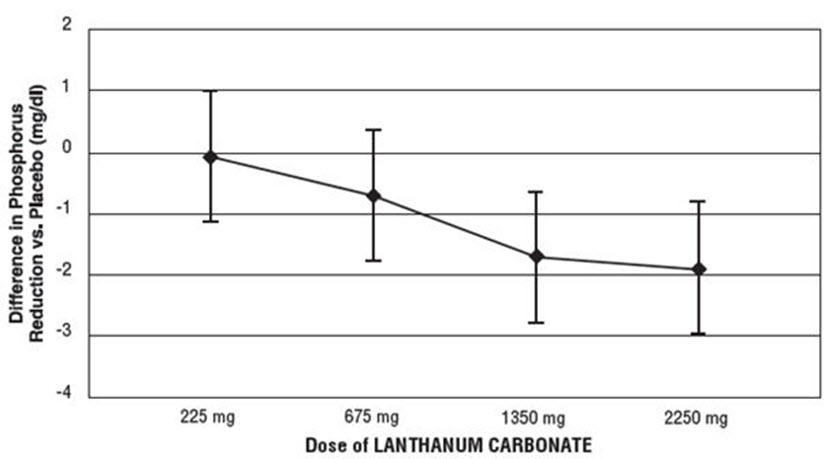
One-hundred and eighty-five patients with ESRD undergoing either hemodialysis (n=146) or peritoneal dialysis (n=39) were enrolled in two placebo-controlled, randomized withdrawal studies. Sixty-four percent of subjects were male, 28% black, 62% white, and 10% of other races. The mean age was 58.4 years and the duration of dialysis ranged from 0.2 to 21.4 years. After titration of lanthanum carbonate to achieve a phosphate level between 4.0 and 5.6 mg/dL in one study (doses up to 2,250 mg/day) or ≤5.9 mg/dL in the second study (doses up to 3,000 mg/day) and maintenance through 6 weeks, patients were randomized to lanthanum or placebo. During the placebo-controlled, randomized withdrawal phase (four weeks), the phosphorus concentration rose in the placebo group by 1.7 mg/dL in one study and 1.9 mg/dL in the other study relative to patients who remained on lanthanum carbonate therapy.
14.2 Open-Label Active-Controlled Studies
Two long-term open-label studies were conducted, involving a total of 2,028 patients with ESRD undergoing hemodialysis. Patients were randomized to receive LANTHANUM CARBONATE or alternative phosphate binders for up to six months in one study and two years in the other. The daily LANTHANUM CARBONATE doses, divided and taken with meals, ranged from 375 mg to 3,000 mg. Doses were titrated to reduce serum phosphate levels to a target level. The daily doses of the alternative therapy were based on current prescribing information or those commonly utilized. Both treatment groups had similar reductions in serum phosphate of about 1.8 mg/dL. Maintenance of reduction was observed for up to three years in patients treated with LANTHANUM CARBONATE in long-term, open-label extensions.
No effects of LANTHANUM CARBONATE on serum levels of 25-dihydroxy vitamin D3, vitamin A, vitamin B12, vitamin E, and vitamin K were observed in patients who were monitored for 6 months.
Paired bone biopsies (at baseline and at one or two years) in 69 patients randomized to either LANTHANUM CARBONATE or calcium carbonate in one study and 99 patients randomized to either LANTHANUM CARBONATE or alternative therapy in a second study showed no differences in the development of mineralization defects between the groups.
Vital status was known for over 2,000 patients, 97% of those participating in the clinical program during and after receiving treatment. The adjusted yearly mortality rate (rate/years of observation) for patients treated with LANTHANUM CARBONATE or alternative therapy was 6.6%.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
LANTHANUM CARBONATE is supplied as a chewable tablet in three dosage strengths for oral administration: 500-mg tablets, 750-mg tablets, and 1,000-mg tablets. Each chewable tablet is white to off-white round, flat with a beveled edge, and debossed on one side with 'S405' above the dosage strength corresponding to the content of elemental lanthanum.
500-mg Patient Pack (2 bottles of 45 tablets, NDC 66993-422-47, per each patient pack) NDC 66993-422-85.
750-mg Patient Pack (6 bottles of 15 tablets, NDC 66993-423-53, per each patient pack) NDC 66993-423-85.
1,000-mg Patient Pack (9 bottles of 10 tablets, NDC 66993-424-75, per each patient pack) NDC 66993-424-85.
-
17 PATIENT COUNSELING INFORMATION
- Advise the patient to read the FDA-approved patient labeling (Medication Guide).
- Advise patients to take LANTHANUM CARBONATE with or immediately after meals [see Dosage and Administration (2)].
- Instruct patients on concomitant medications that should be dosed apart from LANTHANUM CARBONATE [see Drug Interactions (7)].
- Instruct patients who are prescribed LANTHANUM CARBONATE Chewable Tablets to chew or crush tablets completely before swallowing. Emphasize that LANTHANUM CARBONATE Chewable Tablets should not be swallowed intact. Consider crushing LANTHANUM CARBONATE Chewable Tablets completely or prescribing the oral powder formulation for patients with poor dentition or who have difficulty chewing tablets [see Dosage and Administration (2)].
- Advise patients who are taking an oral medication where a reduction in the bioavailability of that medication would have a clinically significant effect on its safety or efficacy to separate the dosing of LANTHANUM CARBONATE from the dosing of the affected drug by several hours [see Drug Interactions (7)].
- Advise patients to notify their physician that they are taking LANTHANUM CARBONATE prior to an abdominal X-ray or if they have a history of gastrointestinal disease [see Warnings and Precautions (5.1, 5.2)].
- SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDELANTHANUM CARBONATE(LAN-tha-num KAR-bo-nate)
Read this Medication Guide before you start taking LANTHANUM CARBONATE and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
What is the most important information I should know about LANTHANUM CARBONATE?
LANTHANUM CARBONATE may cause a bowel blockage, a hole in the bowel, or severe constipation, which can be serious, and sometimes lead to surgery or treatment in a hospital.
- You may have a higher risk of bowel blockage, a hole in the bowel, or severe constipation if you take LANTHANUM CARBONATE and have:
- a history of surgery, ulcers or cancer in the stomach or bowel
- a history of bowel blockage, or problems resulting in a decreased movement of food through your stomach and bowel (e.g., feeling full quickly after eating or constipation)
- an infection or inflammation of the stomach/bowel (peritonitis)
Do not swallow LANTHANUM CARBONATE Chewable Tablets whole. Chew tablets completely before swallowing. If you cannot chew tablets completely, you may crush the tablets thoroughly before swallowing or discuss the oral powder formulation with your healthcare provider.
What is LANTHANUM CARBONATE?
LANTHANUM CARBONATE is a prescription medicine used in people with end-stage renal disease (ESRD) to lower the amount of phosphate in the blood.
Who should not take LANTHANUM CARBONATE?
Do not take LANTHANUM CARBONATE if you:
- have blocked bowels
- have severe constipation
LANTHANUM CARBONATE has not been studied in children and adolescents under 18 years of age.
What should I tell my healthcare provider before taking LANTHANUM CARBONATE?
LANTHANUM CARBONATE may not be right for you. Before starting LANTHANUM CARBONATE, tell your healthcare provider if you:
- have a history of surgery, ulcers or cancer in the stomach or bowel
- have a history of a bowel blockage, constipation, or problems resulting in a decreased movement of food through your stomach and bowel especially if you also have diabetes
- have ulcerative colitis, Crohn's disease or an infection or inflammation of the stomach/bowel (peritonitis)
- plan to have an X-ray of your stomach (abdomen)
- have any other medical conditions
- are pregnant, plan to become pregnant, or plan to breastfeed. Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
Especially tell your healthcare provider if you take:
- antacids
- antibiotics
- thyroid medicine
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine.
How should I take LANTHANUM CARBONATE?
- Take LANTHANUM CARBONATE exactly as prescribed by your healthcare provider
- Your healthcare provider will tell you how much LANTHANUM CARBONATE to take
- Your healthcare provider may change your dose if needed
- Chewable Tablets - Do not swallow tablets whole. Chew tablets completely before swallowing. If you cannot chew tablets completely, or if you have tooth disease, you may crush the tablets thoroughly before swallowing or discuss the oral powder formulation with your healthcare provider.
- Take LANTHANUM CARBONATE with or right after meals
- If you take an antacid medicine, take the antacid 2 hours before or 2 hours after you take LANTHANUM CARBONATE
- If you take medicine for your thyroid (levothyroxine), take the thyroid medicine 2 hours before or 2 hours after you take LANTHANUM CARBONATE
- If you take an antibiotic medicine, take the antibiotic 1 hour before or 4 hours after you take LANTHANUM CARBONATE
What are possible or reasonably likely side effects of LANTHANUM CARBONATE?
See "What is the most important information I should know about LANTHANUM CARBONATE?"
The most common side effects of LANTHANUM CARBONATE include:
- nausea
- vomiting
- diarrhea
- stomach pain
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the side effects of LANTHANUM CARBONATE. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store LANTHANUM CARBONATE?
- Store LANTHANUM CARBONATE between 59°F to 86°F (15°C to 30°C).
Keep LANTHANUM CARBONATE and all medicines out of the reach of children.
General information about LANTHANUM CARBONATE
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use LANTHANUM CARBONATE for a condition for which it was not prescribed. Do not give LANTHANUM CARBONATE to other people, even if they have the same condition. It may harm them.
This Medication Guide summarizes the most important information about LANTHANUM CARBONATE. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about LANTHANUM CARBONATE that is written for healthcare professionals.
For more information call 1-800-828-2088.
What are the ingredients in LANTHANUM CARBONATE?
Active ingredient: lanthanum carbonate
Inactive ingredients: colloidal silicon dioxide NF, dextrates (hydrated) NF, and magnesium stearate NF.
This Medication Guide has been approved by the US Food and Drug Administration.
Distributed by:
Prasco Laboratories
Mason, OH 45040 USARev [8/2020]
- You may have a higher risk of bowel blockage, a hole in the bowel, or severe constipation if you take LANTHANUM CARBONATE and have:
-
PRINCIPAL DISPLAY PANEL - 500 mg Tablet Bottle Package
NDC 66993-422-85
PRASCO
Lanthanum Carbonate
chewable tablets500 mg
Do not swallow tablets whole.
Chew or crush tablets completely before
swallowing.
Take with or immediately after meals.2 bottles/45 chewable tablets each
Rx only
Active Ingredient: Each
tablet contains 500 mg
of lanthanum as lanthanum
carbonate hydrateInactive Ingredients:
Colloidal silicon dioxide,
dextrates, and magnesium
stearate.See package insert for full
prescribing information.
Store at 25°C (77°F)
excursions permitted to
15-30°C (59-86°F)
[see USP Controlled Room
Temperature]
-
PRINCIPAL DISPLAY PANEL - 750 mg Tablet Bottle Package
NDC 66993-423-85
PRASCO
Lanthanum Carbonate
chewable tablets750 mg
Do not swallow tablets whole.
Chew or crush tablets completely before
swallowing.
Take with or immediately after meals.6 bottles/15 chewable tablets each
Rx onlyActive Ingredient: Each
tablet contains 750 mg
of lanthanum as lanthanum
carbonate hydrateInactive Ingredients:
Colloidal silicon dioxide,
dextrates, and magnesium
stearate.See package insert for full
prescribing information.
Store at 25°C (77°F)
excursions permitted to
15-30°C (59-86°F)
[see USP Controlled Room
Temperature]
-
PRINCIPAL DISPLAY PANEL - 1000 mg Tablet Bottle Package
NDC 66993-424-85
PRASCO
Lanthanum Carbonate
chewable tablets1000 mg
Do not swallow tablets whole.
Chew or crush tablets completely before
swallowing.
Take with or immediately after meals.9 bottles/10 chewable tablets each
Rx onlyActive Ingredient: Each
tablet contains 1000 mg
of lanthanum as lanthanum
carbonate hydrateInactive Ingredients:
Colloidal silicon dioxide,
dextrates, and magnesium
stearate.See package insert for full
prescribing information.
Store at 25° C (77°F)
excursions permitted to
15-30°C (59-86°F)
[see USP Controlled Room
Temperature]
-
INGREDIENTS AND APPEARANCE
LANTHANUM CARBONATE
lanthanum carbonate tablet, chewableProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:66993-422 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LANTHANUM CARBONATE (UNII: 490D9F069T) (LANTHANUM CATION (3+) - UNII:O7FU5X12W5) LANTHANUM CATION (3+) 500 mg Inactive Ingredients Ingredient Name Strength DEXTRATES (UNII: G263MI44RU) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color WHITE Score no score Shape ROUND Size 18mm Flavor Imprint Code S405;500 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66993-422-85 2 in 1 PACKAGE 08/30/2017 1 NDC:66993-422-47 45 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA authorized generic NDA021468 08/30/2017 LANTHANUM CARBONATE
lanthanum carbonate tablet, chewableProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:66993-423 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LANTHANUM CARBONATE (UNII: 490D9F069T) (LANTHANUM CATION (3+) - UNII:O7FU5X12W5) LANTHANUM CATION (3+) 750 mg Inactive Ingredients Ingredient Name Strength DEXTRATES (UNII: G263MI44RU) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color WHITE Score no score Shape ROUND Size 20mm Flavor Imprint Code S405;750 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66993-423-85 6 in 1 PACKAGE 08/30/2017 1 NDC:66993-423-53 15 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA authorized generic NDA021468 08/30/2017 LANTHANUM CARBONATE
lanthanum carbonate tablet, chewableProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:66993-424 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LANTHANUM CARBONATE (UNII: 490D9F069T) (LANTHANUM CATION (3+) - UNII:O7FU5X12W5) LANTHANUM CATION (3+) 1000 mg Inactive Ingredients Ingredient Name Strength DEXTRATES (UNII: G263MI44RU) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color WHITE Score no score Shape ROUND Size 22mm Flavor Imprint Code S405;1000 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66993-424-85 9 in 1 PACKAGE 08/30/2017 1 NDC:66993-424-75 10 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA authorized generic NDA021468 08/30/2017 Labeler - Prasco Laboratories (065969375) Establishment Name Address ID/FEI Business Operations Patheon Manufacturing Services LLC 079415560 ANALYSIS(66993-422, 66993-423, 66993-424) , LABEL(66993-422, 66993-423, 66993-424) , MANUFACTURE(66993-422, 66993-423, 66993-424) , PACK(66993-422, 66993-423, 66993-424) Establishment Name Address ID/FEI Business Operations Element Materials Technology Pharma US LLC 081315325 ANALYSIS(66993-422, 66993-423, 66993-424) Establishment Name Address ID/FEI Business Operations Reading Scientific Services Ltd (RSSL) 293610085 ANALYSIS(66993-422, 66993-423, 66993-424) Establishment Name Address ID/FEI Business Operations Triclinic Labs 017662237 ANALYSIS(66993-422, 66993-423, 66993-424) Establishment Name Address ID/FEI Business Operations Microbac Laboratories, Inc. 178463126 ANALYSIS(66993-422, 66993-423, 66993-424) Establishment Name Address ID/FEI Business Operations Reckitt Benckiser Healthcare International Ltd. 230780363 MANUFACTURE(66993-422, 66993-423, 66993-424)



