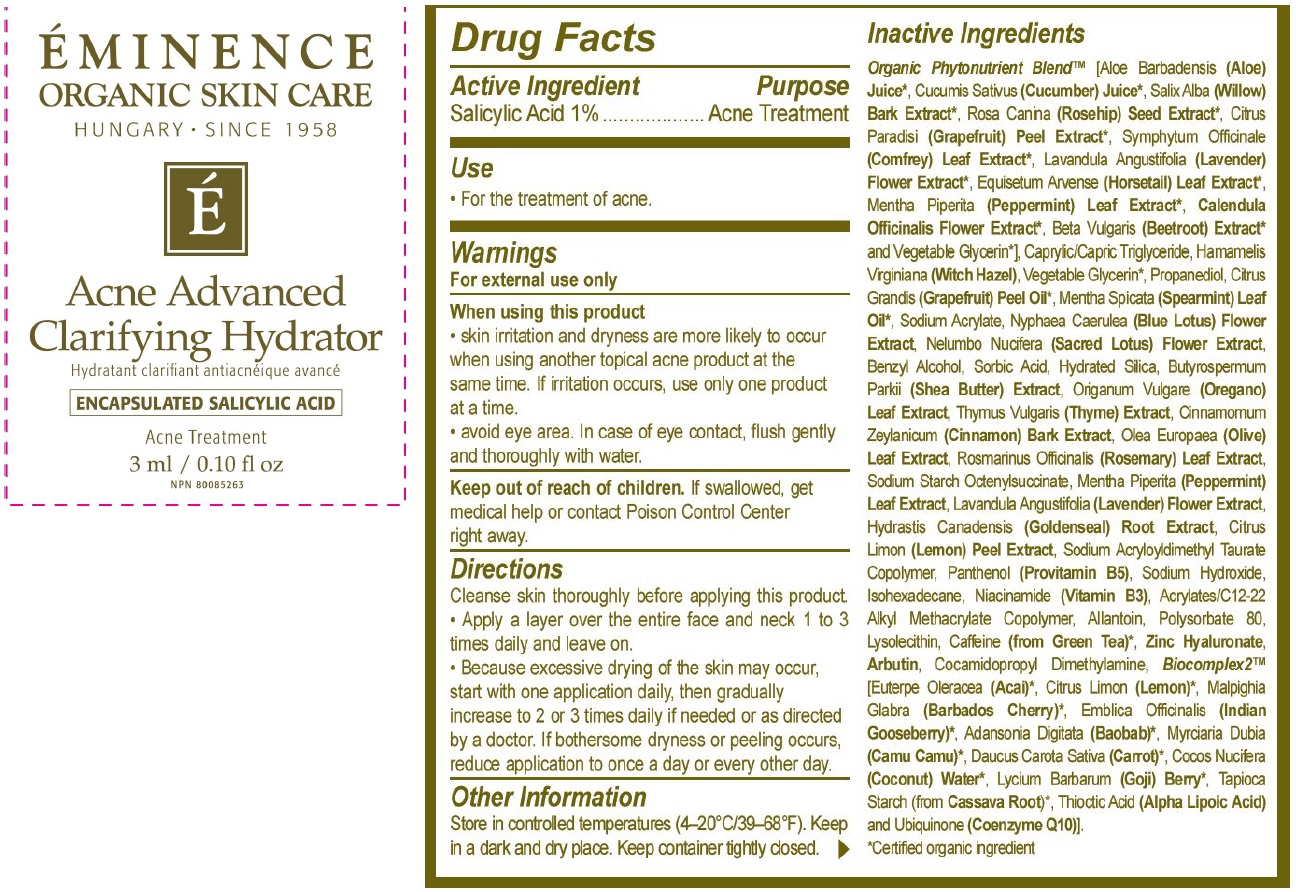
Label: ACNE ADVANCED CLARIFYING HYDRATOR- salicylic acid cream
- NDC Code(s): 15751-3030-2, 15751-3030-6, 15751-3030-9
- Packager: Eminence Organic Skin Care Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 8, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Use
- Warnings
-
Direcitons
- Cleanse skin thoroughly before applying this product
- Apply a layer over the entire face and neck 1 to 3 times daily and leave on.
- Because excessive drying of the skin may occur, start with one application daily, then gradually increase to 2 ot 3 times daily if needed or as directed by a doctor. If bothersome dryness or peeling occurs, reduce application to once a day or every other day.
- Other information
-
Inactive ingredients
Organic Phytonutrient Blend (Aloe Barbadensis (Aloe) Juice, Cucumis Sativus (Cucumber) Juice, Salix Alba (Willow) Bark Extract, Rosa Canina (Rosehip) Seed Extract, Citrus Paradisi (Grapefruit) Peel Extract, Symphytum Officinale (Comfrey) Leaf Extract, Lavandula Angustifolia (Lavender) Flower Extract, Equisetum Argense (Horsetail Leaf Extract, Mentha Piperita (Peppermint) Leaf Extract, Calendula Officinalis Flower Extract, Beta Vulgaris (Beetroot) Extract and Vegetable Glycerin], Caprylic/Capric Triglyceride, Hamamelis Virginiana (Witch Hazel), Vegetable Glycerin, Propanediol, Citrus Grandis (Grapefruit) Peel Oil, Mentha Spicata (Spearmint) Leaf Oil, Sodium Acrylate, Nyphaea Caerulea (Blue Lotus) Flower Extract, Nelumbo Nucifera (Sacred Lotus) Flower Extract, Origanum Vulgare (Oregano) Leaf Extract, Thymus Vulgaris (Thyme) Extract, Cinnamomum Zeylanium (Cinnamon) Bark Extract, Olea Europaea (Olive) Leaf Extract, Rosmarinus Officinalis (Rosemary) Leaf Extract, Sodium Starch Octenylsuccinate, Mentha Piperita (Peppermint) Leaf Extract, Lavandula Angustifolia (Lavender) Flower Extract, Hydrastis Canadensis (Goldenseal) Root Extract, Citrus Limon (Lemon) Peel Extract, Sodium Acrylolydimethyl Taurate Copolymer, Panthenol (Provitamin B5), Sodium Hydroxide, Isohexadecane, Niacinamide (Vitamin B3), Acrylates/C12-22 Alkyl Methacrylate Copolymer, Allantoin, Polysorbate 80, Lysolecithin, Caffeine (from Green Tea), Zinc Hyaluronate, Arbutin, Cocamidopropyl Dimethylamine, Biocomplex2 [Euterpe Oleracea (Acai), [Euterpe Oleracea (Acai), Citrus Limon (Lemon), Malpighia Glabra (Barbados Cherry), Emblica Officinalis (Indian Gooseberry), Adansonia Digitata (Baobab), Myrciaria Dubia (Camu Camu), Daucus Carota Sativa (Carrot), Cocos Nucifera (Coconut) Water, Lycium Barbarum (Goji) Berry, Tapioca Starch (from Cassave Root), Thioctic Acid (Alpha Lipoic Acid) and Ubiquinone (Coenzyme Q10)
*Certified organic ingredient
- Acne Advanced Clarifying Hydrator 1.2oz Box (15751-3030-2)
- Acne Advanced Clarifying Hydrator 1.2oz Bottle (15751-3030-2)
- Acne Advanced Clarifying Hydrator 1.9oz (15751-3030-9)
- Acne Advanced Clarifying Hydrator 0.10oz (15751-3030-6)
-
INGREDIENTS AND APPEARANCE
ACNE ADVANCED CLARIFYING HYDRATOR
salicylic acid creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:15751-3030 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALOE (UNII: V5VD430YW9) CUCUMBER (UNII: YY7C30VXJT) ROSA MOSCHATA OIL (UNII: J99W255AWF) GRAPEFRUIT (UNII: O82C39RR8C) SYMPHYTUM UPLANDICUM LEAF (UNII: D05HXK6R3G) PEPPERMINT (UNII: V95R5KMY2B) CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) BETA VULGARIS (UNII: 4G174V5051) GLYCERIN (UNII: PDC6A3C0OX) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) HAMAMELIS VIRGINIANA ROOT BARK/STEM BARK (UNII: T7S323PKJS) PROPANEDIOL (UNII: 5965N8W85T) CITRUS MAXIMA FRUIT RIND OIL (UNII: 8U3877WD44) SPEARMINT (UNII: J7I2T6IV1N) SODIUM ACRYLATE (UNII: 7C98FKB43H) OREGANO (UNII: 0E5AT8T16U) THYME (UNII: CW657OBU4N) CINNAMON (UNII: 5S29HWU6QB) OLIVE OIL (UNII: 6UYK2W1W1E) ROSEMARY (UNII: IJ67X351P9) GOLDENSEAL (UNII: ZW3Z11D0JV) LEMON (UNII: 24RS0A988O) DEXPANTHENOL (UNII: 1O6C93RI7Z) SODIUM HYDROXIDE (UNII: 55X04QC32I) ISOHEXADECANE (UNII: 918X1OUF1E) NIACIN (UNII: 2679MF687A) ALLANTOIN (UNII: 344S277G0Z) POLYSORBATE 80 (UNII: 6OZP39ZG8H) LYSOPHOSPHATIDYLCHOLINE, SOYBEAN (UNII: CQD833204Z) GREEN TEA LEAF (UNII: W2ZU1RY8B0) ARBUTIN (UNII: C5INA23HXF) COCAMIDOPROPYL DIMETHYLAMINE (UNII: L36BM7DG2T) SOYBEAN OIL (UNII: 241ATL177A) BENZYL ALCOHOL (UNII: LKG8494WBH) SUNFLOWER OIL (UNII: 3W1JG795YI) SORBIC ACID (UNII: X045WJ989B) SODIUM POLYACRYLATE (2500000 MW) (UNII: 05I15JNI2J) ACAI (UNII: 46AM2VJ0AW) CARROT (UNII: L56Z1JK48B) COCONUT (UNII: 3RT3536DHY) STARCH, TAPIOCA (UNII: 24SC3U704I) THIOCTIC ACID (UNII: 73Y7P0K73Y) UBIDECARENONE (UNII: EJ27X76M46) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:15751-3030-2 1 in 1 BOX 09/01/2018 1 35 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:15751-3030-9 55 mL in 1 BOTTLE; Type 0: Not a Combination Product 09/01/2018 3 NDC:15751-3030-6 3 mL in 1 PACKET; Type 0: Not a Combination Product 09/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 09/01/2018 Labeler - Eminence Organic Skin Care Ltd. (205753317)