HYLATOPICPLUS EMOLLIENT- dressing, wound, drug
aerosol, foam
Onset Dermatologics LLC
----------
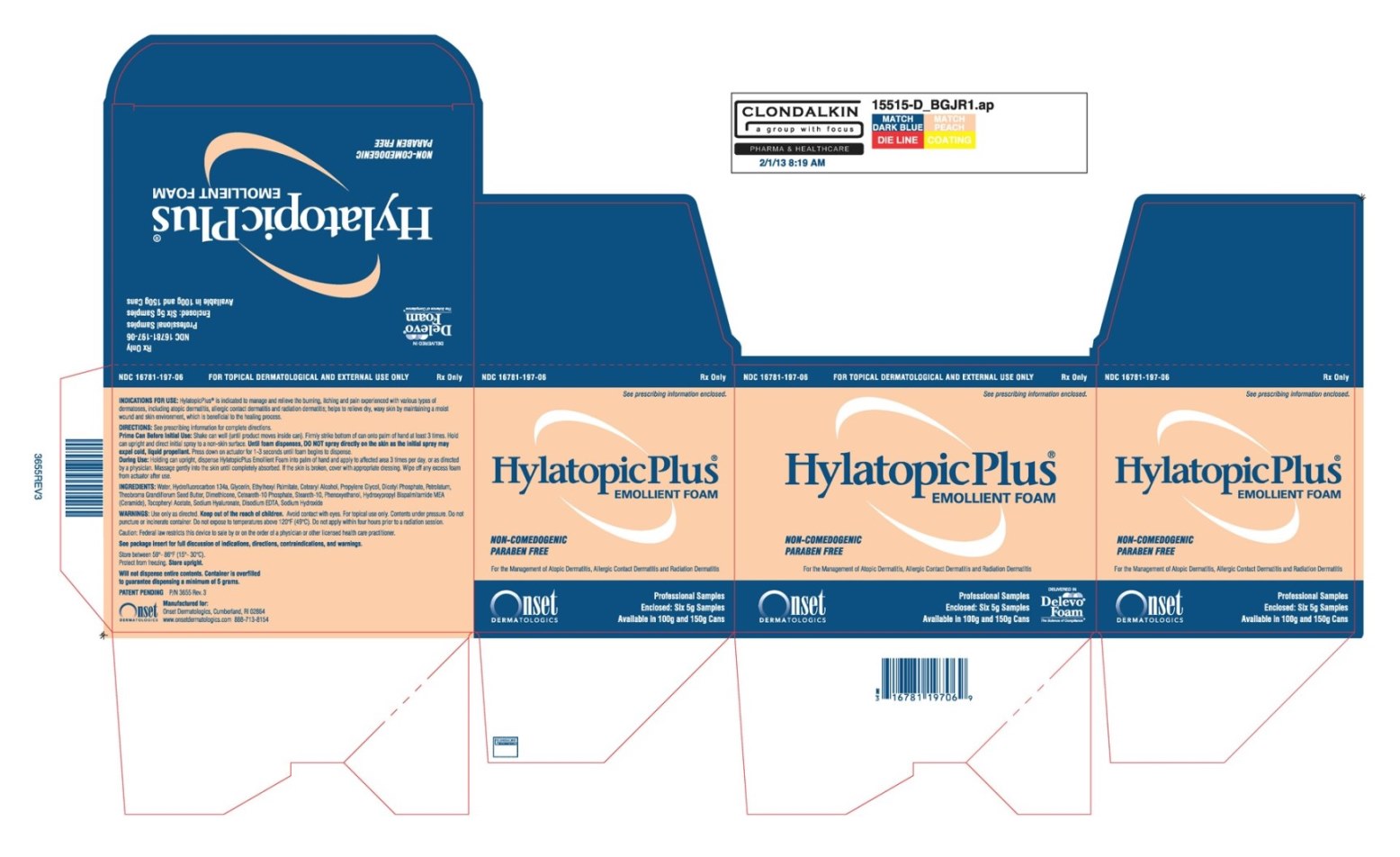
HylatopicPlus Emollient Foam
INDICATIONS FOR USE
Under the supervision of a healthcare professional, HylatopicPlus® Emollient Foam is indicated to manage and relieve the burning, itching and pain experienced with various types of dermatoses, including atopic dermatitis, allergic contact dermatitis and radiation dermatitis. HylatopicPlus® Emollient Foam also helps to relieve dry, waxy skin by maintaining a moist wound & skin environment, which is beneficial to the healing process.
CONTRAINDICATIONS
HylatopicPlus® is contraindicated in persons with a known hypersensitivity to any of the components of the formulation.
WARNINGS
Use only as directed. Keep out of the reach of children. Avoid contact with eyes. For topical use only. Not for ophthalmic use. Contents under pressure. Do not puncture or incinerate container. Do not expose to temperatures above 120ºF (49ºC). Do not apply within four hours prior to a radiation session.
PRECAUTIONS AND OBSERVATIONS
- HylatopicPlus® Emollient Foam does not contain a sunscreen and should not be used prior to extended exposure to the sun.
- If clinical signs of infection are present, appropriate treatment should be initiated; use of HylatopicPlus Emollient Foam may be continued during the anti-infective therapy.
- If the condition does not improve within 10-14 days, consult a physician.
- HylatopicPlus® Emollient Foam may dissolve fuchsin when this dye is used to define the margins of the radiation fields to be treated.
INSTRUCTIONS FOR USE
Important: Prime Can Before Initial Use.
To Prime Can: Shake can vigorously (until product moves inside can). Firmly strike bottom of can onto palm of other hand or a hard surface at least 3 times. Hold can upright and direct initial spray to a non-skin surface. Until foam dispenses, DO NOT spray directly on the skin as the initial spray may expel cold liquid propellant. Press down on actuator for 1 to 3 seconds until foam begins to dispense. If foam does not dispense within 3 seconds, prime can again.
During Use: Holding can upright, dispense HylatopicPlus® Emollient Foam into palm of hand and apply to affected area 3 times per day, or as directed by a physician. Massage gently into the skin until completely absorbed. If the skin is broken, cover with appropriate dressing. Wipe off any excess foam from actuator after use.
INGREDIENTS
Water, Hydrofluorocarbon 134a , Glycerin, Ethylhexyl Palmitate, Cetearyl Alcohol, Propylene Glycol, Dicetyl Phosphate, Petrolatum, Theobroma Grandiflorum Seed Butter, , Dimethicone, Ceteareth-10 Phosphate, Steareth-10, Phenoxyethanol, Hydroxypropyl Bispalmitamide MEA (Ceramide), Tocopheryl Acetate, , Sodium Hyaluronate, Disodium EDTA, Sodium Hydroxide.
HOW SUPPLIED
HylatopicPlus® Emollient Foam is available in 5g (NDC 16781-197-06) professional sample aluminum cans and 100g (NDC 16781-197-96) and 150g (NDC 16781-197-97) commercial aluminum cans.
Will not dispense entire contents. Container is overfilled to guarantee dispensing at least the listed amount.
Caution: Federal law restricts this device to sale by or on the order of a physician or other licensed heath care practitioner.
Store between 59º and 86ºF (15ºC-30ºC).
Protect from freezing.
Store upright.
Manufactured for:

Onset Dermatologics
Cumberland, RI 02864
(888) 713-8154
www.onsetdermatologics.com
Patent Pending
P/N: 2618-Rev.3

| HYLATOPICPLUS EMOLLIENT
dressing, wound, drug aerosol, foam |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Onset Dermatologics LLC (793223707) |