Label: ONDANSETRON- ondansetron hydrochloride injection
-
Contains inactivated NDC Code(s)
NDC Code(s): 54868-5888-0 - Packager: Physicians Total Care, Inc.
- This is a repackaged label.
- Source NDC Code(s): 62778-027
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated August 15, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
The active ingredient in Ondansetron Injection, USP is the ondansetron hydrochloride (HCl), the racemic form of ondansetron and a selective blocking agent of the serotonin 5-HT3 receptor type. Chemically it is (±) 1, 2, 3, 9-tetrahydro-9-methyl-3- [(2-methyl-1H-imidazol-1-yl) methyl]-4H-carbazol-4-one, monohydrochloride dihydrate. It has the following structural formula:
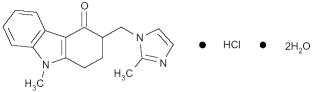
The empirical formula is C18H19N3O - HCl - 2H2O, representing a molecular weight of 365.9. Ondansetron HCl is a white to off-white powder that is soluble in water and normal saline.
Sterile Injection for Intravenous (I.V.) or Intramuscular (I.M.) Administration: Each 1 mL of aqueous solution in the 2-mL single-dose vial contains 2 mg of ondansetron as the hydrochloride dihydrate; 9.0 mg of sodium chloride, USP, and 0.5 mg of citric acid monohydrate, USP and 0.25 mg of trisodium citrate dihydrate, USP as buffers in Water for Injection, USP.
Each 1 mL of aqueous solution in the 20-mL multidose vial contains 2 mg of ondansetron as the hydrochloride dihydrate; 8.3 mg of sodium chloride, USP; 0.5 mg of citric acid monohydrate, USP and 0.25 mg of trisodium citrate dihydrate, USP buffers; and 1.2 mg of methylparaben, NF and 0.15 mg of propylparaben, NF as preservatives in Water for Injection, USP.
Ondansetron Injection, USP is a clear, colorless, nonpyrogenic, sterile solution. The pH of the injection solution is 3.3 to 4.0.
-
CLINICAL PHARMACOLOGY
Pharmacodynamics: Ondansetron is a selective 5-HT3 receptor antagonist. While ondansetron's mechanism of action has not been fully characterized, it is not a dopamine-receptor antagonist.
Serotonin receptors of the 5-HT3 type are present both peripherally on vagal nerve terminals and centrally in the chemoreceptor trigger zone of the area postrema. It is not certain whether ondansetron's antiemetic action in chemotherapy-induced nausea and vomiting is mediated centrally, peripherally, or in both sites. However, cytotoxic chemotherapy appears to be associated with release of serotonin from the enterochromaffin cells of the small intestine. In humans, urinary 5-HIAA (5-hydroxyindoleacetic acid) excretion increases after cisplatin administration in parallel with the onset of vomiting. The released serotonin may stimulate the vagal afferents through the 5-TH3 receptors and initiate the vomiting reflex.
In animals, the emetic response to cisplatin can be prevented by pretreatment with an inhibitor of serotonin synthesis, bilateral abdominal vagotomy and greater splanchnic nerve section, or pretreatment with a serotonin 5-HT3 receptor antagonist.
In normal volunteers, single I.V. doses of 0.15 mg/kg of ondansetron had no effect on esophageal motility, gastric motility, lower esophageal sphincter pressure, or small intestinal transit time. In another study in six normal male volunteers, a 16-mg dose infused over 5 minutes showed no effect of the drug on cardiac output, heart rate, stroke volume, blood pressure, or electrocardiogram (ECG).
Multiday administration of ondansetron has been shown to slow colonic transit in normal volunteers. Ondansetron has no effect on plasma prolactin concentrations.
In a gender-balanced pharmacodynamic study (n=56), ondansetron 4 mg administered intravenously or intramuscularly was dynamically similar in the prevention of nausea and vomiting using the ipecacuanha model of emesis.
Ondansetron does not alter the respiratory depressant effects produced by alfentanil or the degree of neuromuscular blockade produced by atracurium. Interactions with general or local anesthetics have not been studied.
Pharmacokinetics: Ondansetron is extensively metabolized in humans, with approximately 5% of a radiolabeled dose recovered as the parent compound from the urine. The primary metabolic pathway is hydroxylation on the indole ring followed by glucuronide or sulfate conjugation.
Although some nonconjugated metabolites have pharmacologic activity, these are not found in plasma at concentrations likely to significantly contribute to the biological activity of ondansetron.
In vitro metabolism studies have shown that ondansetron is a substrate for human hepatic cytochrome P-450 enzymes, including CYP1A2, CYP2D6, and CYP3A4. In terms of overall ondansetron turnover, CYP3A4 played the predominant role. Because of the multiplicity of metabolic enzymes capable of metabolizing ondansetron, it is likely that inhibition or loss of one enzyme (e.g., CYP2D6 genetic deficiency) will be compensated by others and may result in little change in overall rates of ondansetron elimination. Ondansetron elimination may be affected by cytochrome P-450 inducers. In a pharmacokinetic study of 16 epileptic patients maintained chronically on CYP3A4 inducers, carbamazepine or phenytoin, reduction in AUC, Cmax and T1/2 of ondansetron was observed1. This resulted in a significant increase in clearance. However, on the basis of available data, no dosage adjustment for ondansetron is recommended (see PRECAUTIONS: Drug Interactions).
In humans, carmustine, etoposide, and cisplatin do not affect the pharmacokinetics of ondansetron.
In normal adult volunteers, the following mean pharmacokinetic data have been determined following a single 0.15 mg/kg I.V. dose.
Table 1. Pharmacokinetics in Normal Adult Volunteers Age-Group
nPeak Plasma Concentration (ng/mL) Mean Elimination Halt-life (h) Plasma Clearance (L/h/kg) 19-40 11 102 3.5 0.381 61-74 12 106 4.7 0.319 >75 11 170 5.5 0.262 A reduction in clearance and increase in elimination half-life are seen in patients over 75 years of age. In clinical trials with cancer patients, safety and efficacy were similar in patients over 65 years of age and those under 65 years of age; there was an insufficient number of patients over 75 years of age to permit conclusions in that age-group. No dosagee adjustment is recommended in the elderly.
In patients with mild-to-moderate hepatic impairment, clearance is reduced 2-fold and mean half-life is increased to 11.6 hours compared to 5.7 hours in normals. In patients with severe hepatic impairment (Child-Pugh2 score of 10 or greater), clearance is reduced 2-fold to 3-fold and apparent volume of distribution is increased with a resultant increase in half-life to 20 hours. In patients with severe hepatic impairment, a total daily dose of 8 mg should not be exceeded.
Due to the very small contribution (5%) of renal clearance to the overall clearance, renal impairment was not expected to significantly influence the total clearance of ondansetron. However ondansetron mean plasma clearance was reduced by about 41% in patients with severe renal impairment (creatinine clearance <30 mL/min). This reduction in clearance is variable and was not consistent with an increase in half-life. No reduction in dose or dosing frequency in these patients is warranted.
In adult cancer patients, the mean elimination half-life was 4.0 hours, and there was no difference in the multidose pharmacokinetics over a 4-day period. In a study of 21 pediatric cancer patients (4 to 18 years of age) who received three I.V. doses of 0.15 mg/kg of ondansetron at 4-hours intervals, patients older than 15 years of age exhibited ondansetron pharmacokinetic parameters similar to those of adults. Patients 4 to 12 years of age generally showed higher clearance and somewhat larger volume of distribution than adults. Most pediatric patients younger than 15 years of age with cancer had a shorter (2.4 hours) ondansetron plasma half-life than patients older than 15 years of age. It is not known whether these differences in ondansetron plasma half-life may result in differences in efficacy between adults and some young pediatric patients (see CLINICAL TRIALS: Pediatric Studies).
Pharmacokinetic samples were collected from 74 cancer patients 6 to 48 months of age, who received a dose of 0.15 mg/kg of I.V. ondansetron every 4 hours for 3 doses during a safety and efficacy trial. These data were combined with sequential pharmacokinetics data from 41 surgery patients 1 month to 24 months of age, who received a single dose of 0.1 mg/kg of I.V. ondansetron prior to surgery with general anesthesia, and a population pharmacokinetic analysis was performed on the combined data set. The results of this analysis are included in Table 2 and are compared to the pharmacokinetic results in cancer patients 4 to 18 years of age.
Table 2. Pharmacokinetics in Pediatric Cancer Patients 1 Month to 18 Years of Age Subjects and Age-Group N CL (L/h/kg) Vdss
(L/kg)T1/2
(h)
Geometic Mean Mean Pediatriac Cancer Patients
4 to 18 years of ageN = 21 0.599 1.9 2.8 Population PK Patients
1 month to 48 months of ageN = 115 0.582 3.65 4.9 * Population PK (Pharmacokinetic) Patients: 64% cancer patients and 36% surgery patients
Based on the population pharmacokinetic analysis, cancer patients 6 to 48 months of age who receive a dose of 0.15 mg/kg of I.V. ondansetron every 4 hours for 3 doses would be expected to achieve a systemic exposure (AUC) consistent with the exposure achieved in previous pediatric studies in cancer patients (4 to 18 years of age) at similar doses.
Pharmacokinetic information for pediatric cancer patients 6 months to 48 months of age is approved for GlaxoSmithKline Corporation's ondansetron injection. However, due to GlaxoSmithKline's marketing exclusivity rights, this drug product is not labeled for use in this subpopulations of pediatric patients.
In a study of 21 pediatric patients (age 3 to 12 years) who were undergoing surgery requiring anesthesia for a duration of 45 minutes to 2 hours, a single I.V. dose of ondansetron, 2 mg (3 to 7 years) or 4 mg (8 to 12 years), was administered immediately prior to anesthesia induction. Mean weight-normalized clearance and volume of distribution values in these pediatric surgical patients were similar to those previously reported for young adults. Mean terminal half-life was slightly reduced in pediatric patients (range 2.5 to 3 hours) in comparison with adults (range, 3 to 3.5 hours).
In a study of 51 pediatric patients (1 month to 24 months of age) who were undergoing surgery requiring general anesthesia, a single I.V. dose of ondansetron, 0.1 or 0.2 mg/kg, was administered prior to surgery. As shown in Table 3, the 41 patients with pharmacokinetic data were divided into 2 groups, patients 1 month to 4 months of age and patients 5 to 24 months of age, and are compared to pediatric patients 3 to 12 years of age.
Table 3. Pharmacokinetics in Pediatric Surgery Patients 1 Month to 12 Years of Age Subjects and Age-Group N CL (L/h/kg) Vdss
(L/kg)T1/2
(h)
Geometic Mean Mean Pediatriac Surgery Patients
3 to 12 years of ageN = 21 0.439 1.65 2.9 Pediatiiac Surgery Patients
5 to 24 months of ageN = 22 0.581 2.3 2.9 Pediatriac Surgery Patients
1 month to 4 months of ageN = 19 0.401 3.5 6.7 In general, surgical and cancer pediatric patients younger than 18 years tend to have a higher ondansetron clearance compared to adults leading to a shorter half-life in most pediatric patients. In patients 1 month to 4 months of age, a longer half-life was observed due to the higher volume of distribution in this age group.
Pharmacokinetic information for pediatric surgical patients 1 month to 24 months of age is approved for GlaxoSmithKline Corporation's ondansetron injection. However, due to GlaxoSmithKline's marketing exclusivity rights, this drug product is not labeled for use in this subpopulations of pediatric patients.
In normal volunteers (19 to 39 years old, n=23) the peak plasma concentration was 264 ng/mL following a single 32 mg dose administered as a 15-minute I.V. infusion. The mean elimination half-life was 4.1 hours. Systemic exposure to 32 mg of ondansetron was not proportional to dose as measured by comparing dose-normalized AUC values to an 8 mg dose. This is consistent with a small decrease in systemic clearance with increasing plasma concentrations.
A study was performed in normal volunteers (n=56) to evaluate the pharmacokinetics of a single 4-mg dose administered as a 5-minute infusion compared to a single intramuscular injection. Systemic exposure as measured by mean AUC was equivalent, with values of 156 [95%CI 136, 180] and 161 [95% CI 137, 190] ngh/mL for I.V. and I.M. groups, respectively. Mean peak plasma concentrations were 42.9 [95% CI 33.8, 54.4] ng/mL at 10 minutes after I.V. infusion and 31.9[95% CI 26.3,38.6] ng/mL at 41 minutes after I.M. injection. The mean elimination half-life was not affected by route of administration.
Plasma protein binding of ondansetron as measured in vitro was 70% to 76%, with binding constant over the pharmacologic concentration range (10 to 500 ng/mL). Circulating drug also distributes into erythrocytes.
A positive lymphoblast transformation test to ondansetron has been reported, which suggests immunologic sensitivity to ondansetron.
-
CLINICAL TRIALS
Chemotherapy-Induced Nausea and Vomiting:
Adults Studies: In a double-blind study of three different dosing regimens of Ondanselron Injection, USP, 0.015 mg/kg, 0.15 mg/kg, and 0.30 mg/kg, each given three times during the course of cancer chemotherapy, the 0.15-mg/kg dosing regimen was more effective than the 0.015-mg/kg dosing regimen. The 0.30-mg/kg dosing regimen was not shown to be more effective than the 0.15-mg/kg dosing regimen.
Cisplatin-Based Chemotherapy: In a double-blind study in 28 patients, Ondansetron Injection, USP (three 0.15 mg/kg doses) was significantly more effective than placebo in preventing nausea and vomiting induced by cisplatin-based chemotherapy. Treatment response was as shown in Table 4.
Table 4. Prevention of Chemotherapy-Induced Nausea and Vomiting in Single-Day Cisplatin Therapy* in Adults
ONDANSETRON INJECTION, USP Placebo P Value† Number of patients 14 14
Treatment response:
0 Emetic episodes 2 (14%) 0 (0%)
1-2 Emetic episodes 8 (57%) 0 (0%)
3-5 Emetic episodes 2 (14%) 1 (7%)
More than 5 emetic episodes/rescued 2 (14%) 13 (93%) 0.001 Median number of emetic episodes 1.5 Undefined‡
Median time to first emetic episode (h) 11.6 2.8 0.001 Median nausea scores (0-100)§ 3 59 0.034 Global satisfaction with control of nausea and vomiting (0-100)II 96 10.5 0.009 * Chemotherapy was high dose (100 and 120mg/m2; Ondansetron Injection, USP n=6, placebo n=5) or moderate dose (50 and 80 mg/m2; Ondansetron Injection, USP n=8, placebo n=9). Other chemotherapeutic agents included fluorouracil, doxorubicin, and cyclophosphamide. There was no difference between treatments in the types of chemotherapy that would account for differences in response.
† Efficacy based on "all patients treated" analysis.
‡ Median undefined since at least 50% of the patients were rescued or had more than five emetic episodes. § Visual analog scale assessment of nausea: 0 = no nausea, 100 = nausea as bad as it can be.II Visual analog scale assessment of satisfaction: 0 = not at all satisfied, 100 = totally satisfied.
Ondanselron was compared with metoclopramide in a single-blind trial in 307 patients receiving cisplatin ≥100 mg/m2 with or without other chemotherapeutic agents. Patients received the first dose of ondansetron or metoclopramide 30 minutes before cisplatin. Two additional ondansetron doses were administered 4 and 8 hours later, or five additional metoclopramide doses were administered 2, 4, 7, 10, and 13 hours later. Cisplatin was administered over a period of 3 hours or less. Episodes of vomiting and retching were tabulated over the period of 24 hours after cisplatin. The results of this study are summarized in Table 5.
Table 5. Prevention of Vomiting Induced by Cisplatin (≥100 mg/m2) Single-Day Therapy* in Adults
ONDANSETRON INJECTION, USP Metoclopramide P Value Dose 0.15 mg/kg x 3 2 mg/kg x 6
Number of palienls in efficacy population 136 138
Treatment response:
0 Emelic episodes 54 (40%) 41 (30%)
1-2 Emelic episodes 34 (25%) 30 (22%)
3-5 Emelic episodes 19 (14%) 18 (13%)
More than 5 emetic episodes/rescued 29 (21%) 49 (36%)
Comparison of treatments with respect to
0 Emetic episodes 54/136 41/138 0.083 More than 5 emetic episodes/rescued 29/136 49/138 0.009 Median number of emetic episodes 1 2 0.005 Median time to first emetic episode (h) 20.5 4.3 <0.001 Global satisfaction with control of nausea and vomiting (0-100)† 85 63 0.001 Acute dystonic reactions 0 8 0.005 Akathisia 0 10 0.002 * In addition to cisplatin, 68% of patients received other chemotherapeutic agents, including cyclophosphamide, etoposide, and fluorouracil. There was no difference between treatments in the types of chemotherapy that would account for differences in response.
† Visual analog scale assessment: 0 = not at all satisfied, 100 = totally satisfied.
In a stratified, randomized, double-blind, parallel-group, multicenter study, a single 32 mg dose of ondansetron was compared with three 0.15-mg/kg doses in patients receiving cisplatin doses of either 50 to 70 mg/m2 or >100 mg/m2. Patients received the first ondansetron dose 30 minutes before cisplatin. Two additional ondansetron doses were administered 4 and 8 hours later to the group receiving three 0.15-mg/kg doses. In both strata, significantly fewer patients on the single 32-mg dose than those receiving the three-dose regimen failed.
Table 6. Prevention of Chemotherapy-Induced Nausea and Vomiting in Single-Dose Therapy in Adults
0.15 mg/kg x 3 Ondansetron Dose 32 mg x 1 P Value High-dose cisplatin (≥ 100 mg/m2)
Number of patients 100 102
Treatment response:
0 Emetic episodes 41 (41%) 49 (48%) 0.315 1-2 Emetic episodes 19 (19%) 25 (25%)
3-5 Emetic episodes 4 (4%) 8 (8%)
More than 5 emetic episodes/rescued 36 (36%) 20 (20%) 0.009 Median time of first emetic episode (h) 21.7 23 0.173 Median nausea scores (0-100)* 28 13 0.004 Median-dose cisplatin (50-70 mg/m2)
Number of patients 101 93
Treatment response:
0 Emetic episodes 62 (61%) 68 (73%) 0.083 1-2 Emetic episodes 11 (11%) 14 (15%)
3-5 Emetic episodes 6 (6%) 3 (3%)
More than 5 emetic episodes/rescued 22 (22%) 8 (9%) 0.011 Median time to first emetic episode (h) Undefined† Undefined
Median nausea scores (0-100)* 9 3 0.131 * Visual analog scale assessment: 0 = no nausea; 100 = nausea as bad as it can be.
† Median undefined since at least 50% of patients did not have any emetic episodes.
Cyclophosphamide-Based Chemotherapy: In a double-blind, placebo-controlled study of Ondansetron Injection, USP (three 0.15-mg/kg doses) in 20 patients receiving cyclophosphamide (500 to 600 mg/m2) chemotherapy, Ondansetron Injection, USP was significantly more effective than placebo in preventing nausea and vomiting. The results are summarized in Table 7.
Table 7. Prevention of Chemotherapy-Induced Nausea and Vomiting in Single-Day Cyclophosphamide Therapy*in Adults
ONDANSETRON INJECTION. USP Placebo P Value † Number of patients 10 10
Treatment response:
0 Emetic episodes 7 (70%) 0 (0%) 0.001 1-2 Emetic episodes 0 (0%) 2 (20%)
3-5 Emetic episodes 2 (20%) 4 (40%)
More than 5 emetic episodes/rescued 1 (10%) 4 (40%) 0.131 Median number of emetic episodes 0 4 0.008 Median time to first emetic episode (h) Undefined‡ 8.79
Median nausea scores (0-100)§ 0 60 0.001 Global satisfaction with control of nausea and vomiting (0-100)II 100 52 0.008 * Chemotherapy consisted of cyclophosphamide in all patients, plus other agents, including fluorouracil, doxorubicin, methotrexate and vincristine. There was no difference between treatments in the type of chemotherapy that would account for differences in response.
† Efficacy based on "all patients treated" analysis.
‡ Median undefined since at least 50% of the patients did not have any emetic episodes.
§ Visual analog scale assessment of nausea: 0 = no nausea, 100 = nausea as bad as it can be. II Visual analog scale assessment of satisfaction: 0 = not at all satisfied, 100 = totally satisfied.
Re-treatment: In uncontrolled trials, 127 patients receiving cisplatin (median dose, 100 mg/m2) and ondansetron who had two or fewer emetic episodes were re-treated with ondansetron and chemotherapy, mainly cisplatin, for a total of 269 re-treatment courses (median, 2; range, 1 to 10). No emetic episodes occurred in 160 (59%), and two or fewer emetic episodes occurred in 217(81%) re-treatment courses.
Pediatric Studies: Four open-label, noncomparative (one US, three foreign) trials have been performed with 209 pediatric cancer patients aged 4 to 18 years given a variety of cisplatin or noncisplatin regimens. In the three foreign trials, the initial Ondansetron Injection, USP dose ranged from 0.04 to 0.87 mg/kg for a total dose of 2.16 to 12 mg. This was followed by the oral administration of ondansetron ranging from 4 to 24 mg daily for 3 days. In the US trial, Ondansetron Injection, USP was administered intravenously (only) in three doses of 0.15 mg/kg each for a total daily dose of 7.2 to 39 mg. In these studies, 58% of the 196 evaluable patients had a complete response (no emetic episodes) on day 1. Thus, prevention of vomiting in these pediatric patients was essentially the same as for patients older than 18 years of age.
An open-label, multicenter, noncomparative trial has been performed in 75 pediatric cancer patients 6 to 48 months of age receiving at least one moderately or highly emetogenic chemotherapeutic agent. Fifty-seven percent (57%) were females; 67% were white, 18% were American Hispanic, and 15% were black patients. Ondansetron Injection, USP was administered intravenously over 15 minutes in three doses of 0.15 mg/kg. The first dosewas administered 30 minutes before the start of chemotherapy, the second and third doses were administered 4 and 8 hours after the first dose, respectively. Eighteen patients (25%) received routine prophylactic dexamethasone (i.e., not given as rescue). Of the 75 evaluable patients, 56% had a complete response (no emetic episodes) on day 1. Thus, prevention of vomiting in these pediatric patients was comparable to the prevention of vomiting in patients 4 years of age and older.
Clinical trial information in pediatric cancer patients 6 months to 48 months of age is approved for GlaxoSmithKline Corporation's ondansetron injection. However, due to GlaxoSmithKline's marketing exclusivity rights, this drug product is not labeled for use in this subpopulation of pediatric patients.
Postoperative Nausea and Vomiting: Prevention of Postoperative Nausea and Vomiting: Adult Studies: Adult surgical patients who received ondansetron immediately before the induction of general balanced anesthesia (barbiturate: thiopental, methohexital, or thiamylal; opioid: alfentanil or fentanyl; nitrous oxide; neuromuscular blockade: succinylcholine/curare and/or vecuronium or atracurium; and supplemental isoflurane) were evaluated in two double blind US studies involving 554 patients. Ondansetron Injection, USP (4 mg) I.V. given over 2 to 5 minutes was significantly more effective than placebo. The results of these studies are summarized in Table 8.
Table 8. Prevention of Postoperative Nausea and Vomiting in Adult Patients
Ondansetron 4 mg I.V. Placebo P Value † Study 1
Emetic episodes:
Number of patients 136 139
Treatment response over 24-h postoperative
period
<0.001 0 Emetic episodes 103 (76%) 64 (46%)
1 Emetic episode 13 (10%) 17 (12%)
More than 1 emetic episode/rescued 20 (15%) 58 (42%)
Nausea assessments:
Number of patients 134 136
No Nausea over 24-h postoperative period 56 (42%) 39 (29%)
Study 2
Emetic episodes:
Number of patients 136 143
Treatment response over 24-h postoperative
period
0.002 0 Emetic episodes 85 (63%) 63 (44%)
1 Emetic episode 16 (12%) 29 (20%)
More than 1 emetic episode/rescued 35 (26%) 51 (36%)
Nausea assessments:
Number of patients 125 133
No nausea over 24-h postoperative period 48 (38%) 42 (32%)
The study populations in Table 8 consisted mainly of females undergoing laparoscopic procedures.
In a placebo-controlled study conducted in 468 males undergoing outpatient procedures, a single 4-mg I.V. ondansetron dose prevented postoperative vomiting over a 24-hour study period in 79% of males receiving drug compared to 63% of males receiving placebo (P<001).
Two other placebo-controlled studies were conducted in 2,792 patients undergoing major abdominal or gynecological surgeries to evaluate a single 4-mg or 8-mg I.V ondansetron dose for prevention of postoperative nausea and vomiting over a 24-hour study period. At the 4-mg dosage, 59% of patients receiving ondansetron versus 45% receiving placebo in the firt study (P<0.001) and 41% of patients receiving ondansetron versus 30% receiving placebo in the second study (P=0.001) experienced no emetic episodes. No additional benefit was observed in patients who received I.V. ondansetron 8 mg compared to patients who received I.V. ondansetron 4 mg.
Pediatric Studies: Three double-blind, placebo-controlled studies have been performed (one US, two foreign) in 1,049 male and female patients (2 to 12 years of age) undergoing general anesthesia with nitrous oxide. The surgical procedures included tonsillectomy with or without adenoidectomy, strabismus surgery, herniorrhaphy, and orchidopexy. Patients were randomized to either single I.V. doses of ondansetron (0.1 mg/kg for pediatric patients weighing 40 kg or less, 4 mg for pediatric patients weighing more than 40 kg) or placebo. Study drug was administered over at least 30 seconds, immediately prior to or following anesthesia induction. Ondansetron was significantly more effective than placebo in preventing nausea and vomiting. The results of these studies are summarized in Table 9.
Table 9. Prevention of Postoperative Nausea and Vomiting in Pediatric Patients 2 to 12 Years of Age Treatment Response Ondansetron n(%) Placebo n(%) Over 24 hours
Study 1
Number of patients 205 210
0 Emetic episodes 140 (68%) 82 (39%)
Failure* 65 (32%) 128 (61%)
Study 2
Number of patients 112 110
0 Emetic episodes 68 (61%) 38 (35%)
Failure* 44 (39%) 72 (65%)
Study 3
Number of patients 206 206
0 Emetic episodes 123 (60%) 96 (47%)
Failure* 83 (40%) 110 (53%)
Nausea assessments†:
Number of patients 185 191
None 119 (64%) 99 (52%)
* Failure was one or more emetic episodes, rescued, or withdrawn.
† Nausea measured as none mild, or severe.
A double-blind, multicenter, placebo-controlled study was conducted in 670 pediatric patients 1 month to 24 months of age who were undergoing routine surgery under general anesthesia. Seventy-five percent (75%) were males; 64% were white, 15% were black, 13% were American Hispanic, 2% were Asian, and 6% were "other race" patients. A single 0.1-mg/kg I.V. dose of ondansetron administered within 5 minutes following induction of anesthesia was statistically significantly more effective than placebo in preventing vomiting. In the placebo group, 28% of patients experienced vomiting compared to 11% of subjects who received ondansetron (P<0.01). Overall, 32 (10%) of placebo patients and 18 (5%) of patients who received ondansetron received antiemetic rescue medication(s) or prematurely withdrew from the study.
Clinical trial information in pediatric surgical patients 1 month to 24 months of age is approved for GlaxoSmithKline Corporation's ondansetron injection. However, due to GlaxoSmithKline's marketing exclusivity rights, this drug product is not labeled for use in this subpopulation of pediatric patients.
Prevention of Further Postoperative Nausea and Vomiting:Adult Studies: Adult surgical patients receiving general balanced anesthesia (barbiturate: thiopental, methohexital, or thiamylal; opioid: alfentanil or fentanyl; nitrous oxide; neuromuscular blockade: succinylcholine/curare and/or vecuronium or atracurium; and supplemental isoflurane) who received no prophylactic antiemetics and who experienced nausea and/or vomiting within 2 hours postoperatively were evaluated in two doudle-blind US studies involving 441 patients.
Patients who experienced an episode of postoperative nausea and/or vomiting were given Ondansetron Injection, USP (4 mg) I.V. over 2 to 5 minutes, and this was significantly more effective than placebo. The results of these studies are summarized in Table 10.
Table 10. Prevention of Further Postoperative Nausea and Vomiting in Adult Patients
Ondansetron 4 mg I.V. Placebo P Value Study 1
Emetic episodes:
Number of patients 104 117
Treatment response 24 h after study drug
0 Emetic episodes 49 (47%) 19 (16%) <0.001 1 Emetic episode 12 (12%) 9 (8%)
More than 1 emetic episode/rescued 43 (41%) 89 (76%)
Median time to first emetic episode (min)* 55.0 43.0
Nausea assessments:
Number of patients 98 102
Mean nausea score over 24-h postoperative period† 1.7 3.1
Study 2
Emetic episodes:
Number of patients 112 108
Treatment response 24 h after study drug
0 Emetic episodes 49 (44%) 28 (26%) 0.006 1 Emetic episode 14 (13%) 3 (3%)
More than 1 emetic episode/rescued 49 (44%) 77 (71%)
Median time to first emetic episode (min)* 60.5 34.0
Nausea assessments:
Number of patients 105 85
No nausea score over 24-h postoperative period† 1.9 2.9
* After administration of study drug.
† Nausea measured on a scale of 0-10 with 0 = no nausea, 10 = nausea as bad as it can be.
The study populations in Table 10 consisted mainly of women undergoing laparoscopic procedures.
Repeat Dosing in Adults: In patients who do not achieve adequate control of postoperative nausea and vomiting following a single, prophylatic, preinduction, I.V. dose of ondansetron 4 mg, administration of a second I.V. dose of ondansetron 4 mg postoperatively does not provide additional control of nausea and vomiting.
Pediatric Study: One double-blind, placebo-controlled, US study was performed in 351 male and female outpatients (2 to 12 years of age) who received general anesthesia with nitrous oxide and no prophylactic antiemetics. Surgical procedures were unrestricted. Patients who experienced two or more emetic episodes within 2 hours following discontinuation of nitrous oxide were randomized to either single I.V. doses of ondansetron (0.1 mg/kg for pediatric patients weighing 40 kg or less, 4 mg for pediatric patients weighing more than 40 kg) or placebo administered over at least 30 seconds. Ondansetron was significantly more effective than placebo in preventing further episodes of nausea and vomiting. The results of the study are summarized in Table 11.
*Failure was one or more emetic episodes, rescued, or withdrawn.Table 11. Prevention of Further Postoperative Nausea and Vomiting in Pediatric Patients 2 to 12 Years of Age Treatment Response Over 24 Hours Ondansetron n (%) Placebo n (%) P Value Number of patients 180 171
0 Emetic episodes 96 (53%) 29 (17%) <0.001 Failure* 84 (47%) 142 (83%)
-
INDICATIONS AND USAGE
- Prevention of nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including high-dose cisplatin. Efficacy of the 32 mg single dose beyond 24 hours in these patients has not been established.
- Prevention of postoperative nausea and/or vomiting. As with other antiemetics, routine prophylaxis is not recommended for patients in whom there is little expectation that nausea and/or vomiting will occur postoperatively. In patients where nausea and/or vomiting must be avoided postoperatively, ondansetron injection, USP is recommended even where the incidence of postoperative nausea and/or vomiting is low. For patients who do not receive prophylactic ondansetron injection, USP and experience nausea and/or vomiting postoperatively, ondansetron injection, USP may be given to prevent further episodes (see CLINICAL TRIALS).
- CONTRAINDICATIONS
- WARNINGS
-
PRECAUTIONS
Ondansetron is not a drug that stimulates gastric or intestinal peristalsis. It should not be used instead of nasogastric suction. The use of ondansetron in patients following abdominal surgery or in patients with chemotherapy-induced nausea and vomiting may mask a progressive ileus and/or gastric distention.
Rarely and predominantly with intravenous ondansetron, transient ECG changes including QT interval prolongation have been reported.
Drug Interactions: Ondansetron does not itself appear to induce or inhibit the cytochrome P-450 drug-metabolizing enzyme system of the liver (see CLINICAL PHARMACOLOGY: Pharmacokinetics). Because ondansetron is metabolized by hepatic cytochrome P-450 drug-metabolizing enzymes (CYP3A4, CYP2D6, CYP1A2), inducers or inhibitors of these enzymes may change the clearance, and hence, the half-life of ondansetron. On the basis of limited available data, no dosage adjustment is recommended for patients on these drugs.
Phenytoin, Carbamazepine, and Rifampicin: In patients treated with potent inducers of CYP3A4 (i.e., phenytoin, carbamazepine, and rifampicin), the clearance of ondansetron was significantly increased and ondansetron blood concentrations were decreased. However, on the basis of available data, no dosage adjustment for ondansetron is recommended for patients on these drugs.1,3Tramadol: Although no pharmacokinetic drug interaction between ondansetron and tramadol has been observed, data from 2 small studies indicate that ondansetron may be associated with an increase in patient controlled administration of tramadol.4,5
Chemotherapy: Tumor response to chemotherapy in the P 388 mouse leukemia model is not affected by ondansetron. In humans, carmustine, etoposide, and cisplatin do not affect the pharmacokinetics of ondansetron.
In a crossover study in 76 pediatric patients, I.V. ondansetron did not increase blood levels of high-dose methotrexate.
Carcinogenesis, Mutagenesis, Impairment of Fertility: Carcinogenic effects were not seen in 2-years studies in rats and mice with oral ondansetron doses up to 10 and 30 mg/kg per day, respectively. Ondansetron was not mutagenic in standard test for mutagenicity. Oral administration of ondansetron up to 15 mg/kg per day did not affect fertility or general reproductive performance of male and female rats.
Pregnancy: Teratogenic Effects: Pregnancy Category B. Reproduction studies have been performed in pregnant rats and rabbits at I.V. doses up to 4 mg/kg per day and have revealed no evidence of impaired fertility or harm to the fetus due to ondansetron. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nursing Mothers: Ondansetron is excreted in the breast milk of rats. It is not known whether ondansetron is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when ondansetron is administered to a nursing woman.
Pediatric Use: Little information is available about the use of ondansetron in pediatric surgical patients younger than 1 month of age. (See CLINICAL TRIALS section for studies of ondansetron in prevention of postoperative nausea and vomiting in patients 1 month of age and older.) Little information is available about the use of ondansetron in pediatric cancer patients younger than 6 months of age. (See CLINICAL TRIALS section for studies of ondansetron in chemotherapy-induced nausea and vomiting in pediatric patients 6 months of age and older.) (See DOSAGE AND ADMINISTRATION.)
The clearance of ondansetron in pediatric patients 1 month to 4 months of age is slower and the half-life is ~2.5 fold longer than patients who are >4 to 24 months of age. As a precaution, it is recommended that patients less than 4 months of age receiving this drug be closely monitored. (See CLINICAL PHARMACOLOGY: Pharmacokinetics).
The frequency and type of adverse events reported in pediatric patients receiving ondansetron were similar to those in patients receiving placebo. (See ADVERSE EVENTS.)
Geriatric Use: Of the total number of subjects enrolled in cancer chemotherapy-induced and postoperative nausea and vomiting in US- and foreign-controlled clinical trials, 862 were 65 years of age and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. Dosage adjustment is not needed in patients over the age of 65 (see CLINICAL PHARMACOLOGY).
-
ADVERSE REACTIONS
Chemotherapy-Induced Nausea and Vomiting: The adverse events in Table 12 have been reported in adults receiving ondansetron at a dosage of three 0.15-mg/kg doses or as a single 32-mg dose in clinical trials. These patients were receiving concomitant chemotherapy, primarily cisplatin, and I.V. fluids. Most were receiving a diuretic.
*See NeurologicalTable 12. Principal Adverse Events in Comparative Trials in Adults
Number of Adult Patients With Event
ONDANSETRON
INJECTION, USP
0.15 mg/kg x 3
n=419ONDANSETRON
INJECTION, USP
32 mg/kg x 1
n=220Meloclopramide n=156 Placebo n=34 Diarrhea 16% 8% 44% 18% Headache 17% 25% 7% 15% Fever 8% 7% 5% 3% Akalhisia 0% 0% 6% 0% Acute dystonic reactions* 0% 0% 5% 0%
The following have been reported during controlled clinical trials:Cardiovascular: Rare cases of angina (chest pain), electrocardiographic alterations, hypotension and tachycardia have been reported. In many cases the relationship to Ondansetron Injection, USP was unclear.
Gastrointestinal: Constipation has been reported in 11% of chemotherapy patients receiving multiday ondansetron.
Hepatic: In comparative trials in cisplatin chemotherapy patients with normal baseline values of aspartate transaminase (AST) and alanine transaminase (ALT) these enzymes have been reported to exceed twice the upper limit of normal in approximately 5% of patients. The increases were transient and did not appear to be related to dose or duration of therapy. On repeat exposure, similar transient elevations in transaminase values occurred in some courses, but symptomatic hepatic disease did not occur.
Integumentary: Rash has occurred in approximately 1% of patients receiving ondansetron.
Neurological: There have been rare reports consistent with, but not diagnostic of, extrapyramidal reactions in patients receiving ondansetron injection, USP, and rare cases of grand mal seizure. The relationship to ondansetron injection, USP was unclear.
Other: Rare cases of hypokalemia have been reported. The relationship to ondansetron injection, USP was unclear.
Postoperative Nausea and Vomiting: The adverse events in Table 13 have been reported in 2% of adults receiving ondansetron at a dosage of 4 mg I.V. over 2 to 5 minutes in clinical trials. Rates of these events were not significantly different in the ondansetron and placebo groups. These patients were receiving multiple concomitant perioperative and postoperative medications.
Table 13. Adverse Events in 2% of Adults Receiving Ondansetron at a Dosage of 4 mg I.V. over 2 to 5 Minutes in Clinical Trials
ONDANSETRON INJECTION, USP 4 mg I.V. n=547 patients Placebo n=547 patients Headache 92 (17%) 77 (14%) Dizziness 67 (12%) 88 (16%) Musculoskeletal pain 57 (10%) 59 (11%) Drowsiness/sedation 44 (8%) 37 (7%) Shivers 38 (7%) 39 (7%) Malaise/fatigue 25 (5%) 30 (5%) Injection site reaction 21 (4%) 18 (3%) Urinary retention 17 (3%) 15 (3%) Postoperative C02-related pain* 12 (2%) 16 (3%) Chest pain (unspecified) 12 (2%) 15 (3%) Anxiety/agitation 11 (2%) 16 (3%) Dysuria 11 (2%) 9 (2%) Hypotension 10 (2%) 12 (2%) Fever 10 (2%) 6 (1%) Cold sensation 9 (2%) 8 (1%) Pruritus 9 (2%) 3 (<1%) Paresthesia 9 (2%) 2 (<1%) *Sites of pain included abdomen, stomach, joints, rib cage, shoulder.
Pediatric Use: The adverse events in Table 14 were the most commonly reported adverse events in pediatric patients receiving ondansetron (a single 0.1 mg/kg dose for pediatric patients weighing 40 kg or less, or 4 mg for pediatric patients weighing more than 40 kg) administered intravenously over at least 30 seconds. Rates of these events were not significantly different in the ondansetron and placebo groups. These patients were receiving multiple concomitant perioperative and postoperative medications.
Table 14. Frequency of Adverse Events From Controlled Studies in Pediatric Patients 2 to 12 Years of Age Adverse Event Ondansetron n=755 Patients Placebo n=731 Patients Wound problem 80 (11%) 86 (12%) Anxiety/agitation 49 (6%) 47 (6%) Headache 44 (6%) 43 (6%) Drowsiness/sedation 41 (5%) 56 (8%) Pyrexia 32 (4%) 41 (6%) The adverse events in Table 15 were the most commonly reported adverse events in pediatric patients, 1 month to 24 months of age, receiving a single 0.1-mg/kg I.V. dose of ondansetron. The incidence and type of adverse events were similar in both the ondansetron and placebo groups. These patients were receiving multiple concomitant perioperative and postoperative medications.
Table 15. Frequency of Adverse Events (Greater Than or Equal to 2% in Either Treatment Group) in Pediatric Patients 1 Month to 24 Months of Age Adverse Event Ondansetron n=336 Patients Placebo n=334 Patients Pyrexia 14 (4%) 14 (4%) Bronchospasm 2 (<1%) 6 (2%) Post-procedural pain 4 (1%) 6 (2%) Diarrhea 6 (2%) 3 (<1%) Adverse events information reported in pediatric surgical patients 1 month to 24 months of age is approved for GlaxoSmithKline Corporation's ondansetron injection. However, due to GlaxoSmithKline's marketing exclusivity rights, this drug product is not labeled for use in this subpopulation of pediatric patients
Observed During Clinical Practice: In addition to adverse events reported from clinical trials, the following events have been identified during post-approval use of intravenous formulations of ondansetron injection, USP. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to Ondansetron Injection, USP.
Cardiovascular: Arrhythmias (including ventricular and supraventricular tachycardia, premature ventricular contractions, and atrial fibrillation), bradycardia, electrocardiographic alterations (including second-degree heart block and ST segment depression, palpitations, and syncope.
General: Flushing. Rare cases of hypersensitivity reactions, sometimes severe (e.g., anaphylaxis/anaphylactoid reactions, angioedema, bronchospasm, cardiopulmunary arrest, hypotension, laryngeal edema, laryngospasm, shock, shortness of breath, stridor) have also been reported.
Hepatobiliary: Liver enzyme abnormalities have been reported. Liver failure and death have been reported in patients with cancer receiving concurrent medications including potentially hepatotoxic cytotoxic chemotherapy and antibiotics. The etiology of the liver failure is unclear.
Local Reactions: Pain, redness, and burning at site of injection. Lower Respiratory: Hiccups Neurological: Oculogyric crisis, appearing alone, as well as with other dystonic reactions. Skin: Urticaria Special Senses: Transient dizziness during or shortly after I.V. infusion. Eye Disorders: Transient blurred vision, in some cases associated with abnormalities of accommodation. Cases of transient blindness, predominantly during intravenous administration, have been reported. These cases of transient blindness were reported to resolve within a few minutes up to 48 hours. - DRUG ABUSE AND DEPENDENCE
-
OVERDOSAGE
There is no specific antidote for ondansetron overdose. Patients should be managed with appropriate supportive therapy. Individual doses as large as 150 mg and total daily dosages (three doses) as large as 252 mg have been administered intravenously without significant adverse events. These doses are more than 10 times the recommended daily dose.
In addition to the adverse events listed above, the following events have been described in the setting of ondansetron overdose: "Sudden blindness" (amaurosis) of 2 or 3 minutes duration plus severe constipation occurred in one patient that was administered 72 mg of ondansetron intravenously as a single dose. Hypotension (and faintness) occurred in another patient that took 48 mg of oral ondansetron. Following infusion of 32 mg over only a 4 minutes period, a vasovagal episode with transient second-degree heart block was observed. In all instances, the events resolved completely.
-
DOSAGE AND ADMINISTRATION
Prevention of Chemotherapy-Induced Nausea and Vomiting:
Adult Dosing: The recommended I.V dosage of ondansetron injection, USP for adults is a single 32 mg dose or three 0.15 mg/kg doses. A single 32 mg dose is infused over 15 minutes beginning 30 minutes before the start of emetogenic chemotherapy. The recommended infusion rate should not be exceeded (see OVERDOSAGE).
With the three-dose (0.15 mg/kg) regimen, the first dose is infused over 15 minutes beginning 30 minutes before the start of emetogenic chemotherapy. Subsequent doses (0.15 mg/kg) are administered 4 and 8 hours after the first dose of ondansetron injection, USP.
Ondansetron injection, USP should not be mixed with solutions for which physical and chemical compatibility have not been established. In particular, this applies to alkaline solutions as precipitate may form.
Vial: DILUTE BEFORE USE FOR PREVENTION OF CHEMOTHERAPY-INDUCED NAUSEA AND VOMITING. Ondansetron injection, USP should be diluted in 50 mL of 5% dextrose injection or 0.9% sodium chloride injection before administration.
Pediatric Dosing: On the basis of the available information (see CLINICAL TRIALS: Pediatric Studies and CLINICAL PHARMACOLOGY: Pharmacokinetics), the dosage in pediatric cancer patients 4 to 18 years of age should be three 0.15 mg/kg doses. The first dose is to be administered 30 minutes before the start of moderately to highly emetogenic chemotherapy, subsequent doses (0.15 mg/kg) are administered 4 and 8 hours after the first dose of ondansetron injection, USP. The drug should be infused intravenously over 15 minutes. Little information is available about dosage in pediatric cancer patients younger than 6 months of age.
Dosing information for pediatric cancer patients 6 months to 48 months of age is approved for GlaxoSmithKline Corporation's ondansetron injection. However, due to GlaxoSmithKline's marketing exclusivity rights, this drug product is not labeled for use in this subpopulation of pediatric patients.
Vial: DILUTE BEFORE USE. Ondansetron injection, USP should be diluted in 50 mL of 5% dextrose injection or 0.9% sodium chloride injection before administration.
Geriatric Dosing: The dosage recommendation is the same as for the general population.
Prevention of Postoperative Nausea and Vomiting:Adult Dosing: The recommended I.V. dosage of ondansetron injection, USP for adults is 4 mg undiluted administered intravenously in not less than 30 seconds, preferably over 2 to 5 minutes, immediately before induction of anesthesia, or postoperatively if the patient experiences nausea and/or vomiting occurring shortly after surgery. Alternatively 4 mg undiluted may be administered intramuscularly as a single injection for adults. While recommended as a fixed dose for patients weighing more than 40 kg, few patients above 80 kg have been studied. In patients who do not achieve adequate control of postoperative nausea and vomiting following a single, prophylactic, pre-induction I.V. dose of ondansetron 4 mg, administration of a second I.V. dose of 4 mg ondansetron postoperatively does not provide additional control of nausea and vomiting.
Vial: REQUIRES NO DILUTION FOR ADMINISTRATION FOR POSTOPERATIVE NAUSEA AND VOMITING.
Pediatric Dosing; The recommended I.V. dosage of ondansetron injection, USP for pediatric patients (1 month to 12 years of age) is a single 0.1 mg/kg dose for patients weighing 40 kg or less, or a single 4 mg dose for patients weighing more than 40 kg. The rate of administration should not be less than 30 seconds, preferably over 2 to 5 minutes immediately prior to or following anesthesia induction, or postoperatively if the patient experiences nausea and/or vomiting occurring shortly after surgery. Prevention of further nausea and vomiting was only studied in patients who had not received prophylactic ondansetron injection, USP.
Dosing information for pediatric surgical patients 1 month to 24 months of age is approved for Glaxo SmithKline Corporation's ondansetron injection. However, due to GlaxoSmithKline's marketing exclusivity rights, this drug product is not labeled for use in this subpopulation of pediatric patients.
Vial: REQUIRES NO DILUTION FOR ADMINISTRATION FOR POSTOPERATIVE NAUSEA AND VOMITING.
Geriatric Dosing: The dosage recommendation is the same as for the general population.
Dosage Adjustment for Patients with Impaired Renal Function: The dosage recommendation is the same as for the general population. There is no experience beyond the first-day administration of ondansetron.
Dosage Adjustment for Patients with Impaired Hepatic Function: In patients with severe hepatic impairment (Child-Pugh2 score of 10 or greater), a single maximal daily dose of 8 mg to be infused over 15 minutes beginning 30 minutes before the start of the emetogenic chemotherapy is recommended. There is no experience beyond first-day administration of ondansetron.
Stability: Ondansetron injection, USP is stable at room temperature under normal lighting conditions for 48 hours after dilution with the following I.V. fluids: 0.9% sodium chloride injection, 5 % dextrose injection, 5% dextrose and 0.9% sodium chloride injection, 5% dextrose and 0.45% sodium chloride injection, and 3% sodium chloride injection.
Although ondansetron injection, USP is chemically and physically stable when diluted as recommended, sterile precautions should be observed because diluents generally do not contain preservative. After dilution, do not use beyond 24 hours.
Note: Parenteral drug products should be inspected visually for particulate matter and discoloration before administration whenever solution and container permit.
Precaution: Occasionally, ondansetron precipitates at the stopper/vial interface in vials stored upright. Potency and safety are not affected. If a precipitate is observed, resolubilize by shaking the vial vigorously.
- HOW SUPPLIED
-
REFERENCES
- Britto MR, Hussey EK, Mydlow P, et al. Effect of enzyme inducers on ondansetron (OND) metabolism in humans. Clin Pharmacol Ther. 1997;61:228.
- Pugh RNH, Murray-Lyon IM, Dawson JL, Pietroni MC, Williams R. Transection of the oesophagus for bleeding oesophageal varices. Brit J Surg. 1973;60:646-649.
- Villikka K, Kivisto KT, Neuvonen PJ. The effect of rifampin on the pharmacokinetics of oral and intravenous ondansetron. Clin Pharmacol Ther. 1999;65:377-381.
- De Witte JL. Schoenmaekers B, Sessler DI, et al. AnesthAnalg. 2001;92:1319-1321.
- Arcioni R, della Rocca M, RomanÒ R, et al. Anesth Analg. 2002;94:1553-1557.
Manufactured by:
HIKMA FARMACêUTICA (PORTUGAL), S.A.
Estrada do Rio da MÒ, n0 8, 8A e 8B - Fervenca,
2705 - 906 Terrugem SNT
PORTUGALfor:
WEST-WARD PHARMACEUTICAL Corp.
465 Industrial Way West
EATONTOWN NJ 07724
USAIss.: Sept.2006
Relabeling and Repackaging by:
Physicians Total Care, Inc.
Tulsa, OK 74146
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ONDANSETRON
ondansetron hydrochloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-5888(NDC:62778-027) Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ONDANSETRON HYDROCHLORIDE (UNII: NMH84OZK2B) (ONDANSETRON - UNII:4AF302ESOS) ONDANSETRON HYDROCHLORIDE 2 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-5888-0 5 in 1 CARTON 1 2 mL in 1 VIAL Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076781 05/08/2008 Labeler - Physicians Total Care, Inc. (194123980) Establishment Name Address ID/FEI Business Operations Physicians Total Care, Inc. 194123980 relabel, repack


