HEADACHE RELIEF PM- acetaminophen, aspirin, diphenhydramine citrate tablet, film coated
DOLGENCORP, LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Dollar General 44-674-Delisted
Active ingredients (in each caplet)
Acetaminophen 250 mg
Aspirin 250 mg (NSAID)*
Diphenhydramine citrate 38 mg
*nonsteroidal anti-inflammatory drug
Uses
for the temporary relief of occasional headaches and minor aches and pains with accompanying sleeplessness
Warnings
Reye's syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction, which may include:
- hives
- shock
- facial swelling
- asthma (wheezing)
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- blisters
- rash
- skin reddening
If a skin reaction occurs, stop use and seek medical help right away.
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- 3 or more alcoholic drinks every day while using this product
- with other drugs containing acetaminophen
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- are age 60 or older
- have 3 or more alcoholic drinks every day while using this product
- have had stomach ulcers or bleeding problems
- take other drugs containing prescription or nonprescription NSAIDs [aspirin, ibuprofen, naproxen, or others]
- take more or for a longer time than directed
- take a blood thinning (anticoagulant) or steroid drug
Do not use
- if you have ever had an allergic reaction to acetaminophen, aspirin, or any other pain reliever/fever reducer
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- with any other product containing diphenhydramine, even one used on skin
- in children under 12 years of age
Ask a doctor before use if
- stomach bleeding warning applies to you
- you have a history of stomach problems, such as heartburn
- you have difficulty in urination due to enlargement of the prostate gland
- you have liver disease
- you have a breathing problem such as emphysema or chronic bronchitis
- you are taking a diuretic
- you have high blood pressure, heart disease, liver cirrhosis, or kidney disease
- you have glaucoma
- you have asthma
Ask a doctor or pharmacist before use if you are taking
- a prescription drug for:
- diabetes
- gout
- arthritis
- any other drug, or are under a doctor’s care for any serious condition
- sedatives or tranquilizers
- any product that contains aspirin, acetaminophen, or any other pain reliever/fever reducer
When using this product
- avoid alcoholic beverages
- drowsiness may occur
- do not drive a motor vehicle or operate machinery
Stop use and ask a doctor if
- an allergic reaction occurs. Seek medical help right away.
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- ringing in the ears or a loss of hearing occurs
- sleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of a serious underlying medical illness.
- pain gets worse or lasts for more than 10 days
- redness or swelling is present
- new symptoms occur
These could be signs of a serious condition.
Directions
-
do not take more than directed
- adults and children 12 years and over: take 2 caplets at bedtime, with a full glass of water
- do not take more than 2 caplets in 24 hours, unless directed by a doctor
- children under 12 years: do not use
Other information
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, crospovidone, FD&C blue #1 aluminum lake, hypromellose, maltodextrin, medium chain triglycerides, microcrystalline cellulose, polydextrose, povidone, sodium lauryl sulfate, sodium starch glycolate, sodium sulfate, stearic acid, talc, titanium dioxide, yellow iron oxide, zinc stearate
Principal display panel
DG™|health
Compare to active
ingredients of Excedrin®
PM Headache**
Headache Relief PM
Acetaminophen
Aspirin (NSAID)
Diphenhydramine citrate
Pain Reliever/Nighttime Sleep Aid
• Caffeine free • Non-habit forming
50 Caplets
Actual Caplet Size
TAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
**This product is not manufactured or distributed by Novartis AG,
owner of the registered trademark Excedrin® PM Headache.
50844 ORG041867415
DISTRIBUTED BY DOLGENCORP, LLC
100 MISSION RIDGE
GOODLETTSVILLE, TN 37072
DG Health 44-674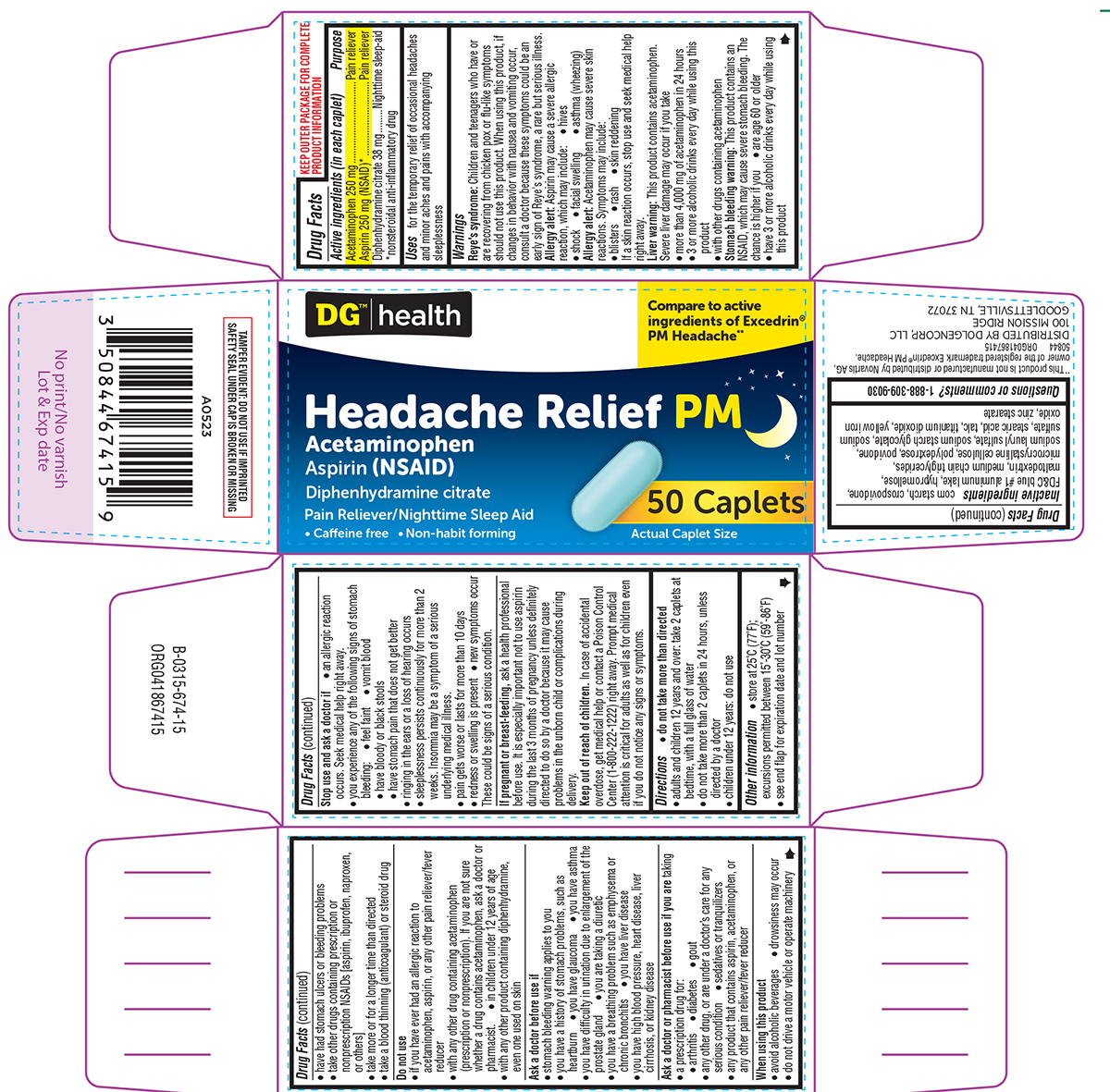
| HEADACHE RELIEF PM
acetaminophen, aspirin, diphenhydramine citrate tablet, film coated |
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
| Labeler - DOLGENCORP, LLC (068331990) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | pack(55910-674) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | manufacture(55910-674) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 868734088 | pack(55910-674) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 967626305 | pack(55910-674) | |