FANSIDAR
-
sulfadoxine and
pyrimethamine tablet
Genentech, Inc.
----------
WARNING: FATALITIES ASSOCIATED WITH THE ADMINISTRATION OF FANSIDAR HAVE OCCURRED DUE TO SEVERE REACTIONS, INCLUDING STEVENS-JOHNSON SYNDROME AND TOXIC EPIDERMAL NECROLYSIS. FANSIDAR PROPHYLAXIS MUST BE DISCONTINUED AT THE FIRST APPEARANCE OF SKIN RASH, IF A SIGNIFICANT REDUCTION IN THE COUNT OF ANY FORMED BLOOD ELEMENTS IS NOTED, OR UPON THE OCCURRENCE OF ACTIVE BACTERIAL OR FUNGAL INFECTIONS.
DESCRIPTION
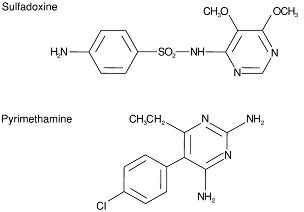
Fansidar is an antimalarial agent, each tablet containing 500 mg N1-(5,6-dimethoxy-4-pyrimidinyl) sulfanilamide (sulfadoxine) and 25 mg 2,4-diamino-5-(p-chlorophenyl)-6-ethylpyrimidine (pyrimethamine). Each tablet also contains cornstarch, gelatin, lactose, magnesium stearate and talc.
CLINICAL PHARMACOLOGY
Microbiology
Mechanism of Action
Sulfadoxine and pyrimethamine, the constituents of Fansidar, are folic acid antagonists. Sulfadoxine inhibits the activity of dihydropteroate synthase whereas pyrimethamine inhibits dihydrofolate reductase.
Activity in vitro
Sulfadoxine and pyrimethamine are active against the asexual erythrocytic stages of Plasmodium falciparum. Fansidar may also be effective against strains of P. falciparum resistant to chloroquine.
Drug Resistance
Strains of P. falciparum with decreased susceptibility to sulfadoxine and/or pyrimethamine can be selected in vitro or in vivo. P. falciparum malaria that is clinically resistant to Fansidar occurs frequently in parts of Southeast Asia and South America, and is also prevalent in East and Central Africa. Therefore, Fansidar should be used with caution in these areas. Likewise, Fansidar may not be effective for treatment of recrudescent malaria that develops after prior therapy (or prophylaxis) with Fansidar.
Pharmacokinetics
Absorption
After administration of 1 tablet, peak plasma levels for pyrimethamine (approximately 0.2 mg/L) and for sulfadoxine (approximately 60 mg/L) are reached after about 4 hours.
Distribution
The volume of distribution for sulfadoxine and pyrimethamine is 0.14 L/kg and 2.3 L/kg, respectively.
Patients taking 1 tablet a week (recommended adult dose for malaria prophylaxis) can be expected to have mean steady state plasma concentrations of about 0.15 mg/L for pyrimethamine after about four weeks and about 98 mg/L for sulfadoxine after about seven weeks. Plasma protein binding is about 90% for both pyrimethamine and sulfadoxine. Both pyrimethamine and sulfadoxine cross the placental barrier and pass into breast milk.
Metabolism
About 5% of sulfadoxine appears in the plasma as acetylated metabolite, about 2 to 3% as the glucuronide. Pyrimethamine is transformed to several unidentified metabolites.
INDICATIONS AND USAGE
Treatment of Acute Malaria
Fansidar is indicated for the treatment of acute, uncomplicated P. falciparum malaria for those patients in whom chloroquine resistance is suspected. However, strains of P. falciparum (see CLINICAL PHARMACOLOGY: Microbiology) may be encountered which have developed resistance to Fansidar, in which case alternative treatment should be administered.
Prevention of Malaria
Malaria prophylaxis with Fansidar is not routinely recommended and should only be considered for travelers to areas where chloroquine-resistant P. falciparum malaria is endemic and sensitive to Fansidar, and when alternative drugs are not available or are contraindicated (see CONTRAINDICATIONS). However, strains of P. falciparum may be encountered which have developed resistance to Fansidar.
CONTRAINDICATIONS
- Repeated prophylactic (prolonged) use of Fansidar is contraindicated in patients with renal or hepatic failure or with blood dyscrasias;
- Hypersensitivity to pyrimethamine, sulfonamides, or any other ingredient of Fansidar;
- Patients with documented megaloblastic anemia due to folate deficiency;
- Infants less than 2 months of age;
- Prophylactic use of Fansidar in pregnancy at term and during the nursing period.
WARNINGS
FATALITIES ASSOCIATED WITH THE ADMINISTRATION OF FANSIDAR HAVE OCCURRED DUE TO SEVERE REACTIONS, INCLUDING STEVENS-JOHNSON SYNDROME AND TOXIC EPIDERMAL NECROLYSIS. FANSIDAR PROPHYLAXIS MUST BE DISCONTINUED AT THE FIRST APPEARANCE OF SKIN RASH, IF A SIGNIFICANT REDUCTION IN THE COUNT OF ANY FORMED BLOOD ELEMENTS IS NOTED, OR UPON THE OCCURRENCE OF ACTIVE BACTERIAL OR FUNGAL INFECTIONS.
Fatalities associated with the administration of sulfonamides, although rare, have occurred due to severe reactions, including fulminant hepatic necrosis, agranulocytosis, aplastic anemia and other blood dyscrasias. Fansidar prophylactic regimen has been reported to cause leukopenia during a treatment of 2 months or longer. This leukopenia is generally mild and reversible.
PRECAUTIONS
General
Oral Fansidar has not been evaluated for the treatment of cerebral malaria or other severe manifestations of complicated malaria, including hyperparasitemia, pulmonary edema or renal failure. Patients with severe malaria are not candidates for oral therapy. In the event of recrudescent P. falciparum infections after treatment with Fansidar or failure of chemoprophylaxis with Fansidar, patients should be treated with a different blood schizonticide.
Fansidar should be given with caution to patients with impaired renal or hepatic function, to those with possible folate deficiency and to those with severe allergy or bronchial asthma. As with some sulfonamide drugs, in glucose-6-phosphate dehydrogenase-deficient individuals, hemolysis may occur. Urinalysis with microscopic examination and renal function tests should be performed during therapy of those patients who have impaired renal function. Excessive sun exposure should be avoided.
Information for the Patient
Patients should be warned that at the first appearance of a skin rash, they should stop use of Fansidar and seek medical attention immediately. Adequate fluid intake must be maintained in order to prevent crystalluria and stone formation.
Patients should also be warned that the appearance of sore throat, fever, arthralgia, cough, shortness of breath, pallor, purpura, jaundice or glossitis may be early indications of serious disorders which require prophylactic treatment to be stopped and medical treatment to be sought.
Females should be cautioned against becoming pregnant and should not breastfeed their infants during Fansidar therapy or prophylactic treatment.
Patients should be warned to keep Fansidar out of reach of children.
Patients also should be advised:
- that malaria can be a life-threatening infection;
- that Fansidar is being prescribed to help prevent or treat this serious infection;
- that no chemoprophylactic regimen is 100% effective, and protective clothing, insect repellents, and bednets are important components of malaria prophylaxis;
- to seek medical attention for any febrile illness that occurs after return from a malarious area and inform their physician that they may have been exposed to malaria;
- that in a small percentage of cases, patients are unable to take this medication because of side effects, and it may be necessary to change medications;
- that when used as prophylaxis, the first dose of Fansidar should be taken 1 or 2 days prior to arrival in an endemic area;
- that if the patient experiences any symptom that may affect the patient's ability to take this drug as prescribed, the physician should be contacted and alternative antimalarial medication should be considered.
Laboratory Tests
Regularly scheduled complete blood counts, liver enzyme tests and analysis of urine for crystalluria should be performed whenever Fansidar is administered for more than three months.
Drug Interactions
There have been reports which may indicate an increase in incidence and severity of adverse reactions when chloroquine is used with Fansidar as compared to the use of Fansidar alone. Fansidar is compatible with quinine and with antibiotics. However, antifolic drugs such as sulfonamides, trimethoprim, or trimethoprim-sulfamethoxazole combinations should not be used while the patient is receiving Fansidar for antimalarial prophylaxis. Fansidar has not been reported to interfere with antidiabetic agents.
If signs of folic acid deficiency develop, Fansidar should be discontinued. When recovery of depressed platelets or white blood cell counts in patients with drug-induced folic acid deficiency is too slow, folinic acid (leucovorin) may be administered in doses of 5 to 15 mg intramuscularly daily for 3 days or longer.
Carcinogenesis, Mutagenesis, Impairment Of Fertility
Pyrimethamine was not found carcinogenic in female mice or in male and female rats. The carcinogenic potential of pyrimethamine in male mice could not be assessed from the study because of markedly reduced life-span. Pyrimethamine was found to be mutagenic in laboratory animals and also in human bone marrow following 3 or 4 consecutive daily doses totaling 200 mg to 300 mg. Pyrimethamine was not found mutagenic in the Ames test. Testicular changes have been observed in rats treated with 105 mg/kg/day of Fansidar and with 15 mg/kg/day of pyrimethamine alone. Fertility of male rats and the ability of male or female rats to mate were not adversely affected at dosages of up to 210 mg/kg/day of Fansidar. The pregnancy rate of female rats was not affected following their treatment with 10.5 mg/kg/day, but was significantly reduced at dosages of 31.5 mg/kg/day or higher, a dosage approximately 30 times the weekly human prophylactic dose or higher.
Pregnancy
Teratogenic Effects: Pregnancy Category C
Fansidar has been shown to be teratogenic in rats when given in weekly doses approximately 12 times the weekly human prophylactic dose. Teratology studies with pyrimethamine plus sulfadoxine (1:20) in rats showed the minimum oral teratogenic dose to be approximately 0.9 mg/kg pyrimethamine plus 18 mg/kg sulfadoxine. In rabbits, no teratogenic effects were noted at oral doses as high as 20 mg/kg pyrimethamine plus 400 mg/kg sulfadoxine.
There are no adequate and well-controlled studies in pregnant women. However, due to the teratogenic effect shown in animals and because pyrimethamine plus sulfadoxine may interfere with folic acid metabolism, Fansidar therapy should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Women of childbearing potential who are traveling to areas where malaria is endemic should be warned against becoming pregnant, and should be advised to practice contraception during prophylaxis with Fansidar and for three months after the last dose.
Pediatric Use
Fansidar should not be given to infants less than 2 months of age because of inadequate development of the glucuronide-forming enzyme system.
Geriatric Use
Clinical studies of Fansidar did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy. This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
ADVERSE REACTIONS
For completeness, all major reactions to sulfonamides and to pyrimethamine are included below, even though they may not have been reported with Fansidar (see WARNINGS and PRECAUTIONS: Information for the Patient).
Hematological Changes
Agranulocytosis, aplastic anemia, megaloblastic anemia, thrombocytopenia, leukopenia, hemolytic anemia, purpura, hypoprothrombinemia, methemoglobinemia, and eosinophilia.
Skin and Miscellaneous Sites Allergic Reactions
Erythema multiforme, Stevens-Johnson syndrome, generalized skin eruptions, toxic epidermal necrolysis, urticaria, serum sickness, pruritus, exfoliative dermatitis, anaphylactoid reactions, periorbital edema, conjunctival and scleral injection, photosensitization, arthralgia, allergic myocarditis, slight hair loss, Lyell's syndrome, and allergic pericarditis.
Gastrointestinal Reactions
Glossitis, stomatitis, nausea, emesis, abdominal pains, hepatitis, hepatocellular necrosis, diarrhea, pancreatitis, feeling of fullness, and transient rise of liver enzymes.
Central Nervous System Reactions
Headache, peripheral neuritis, mental depression, convulsions, ataxia, hallucinations, tinnitus, vertigo, insomnia, apathy, fatigue, muscle weakness, nervousness, and polyneuritis.
Genitourinary
Renal failure, interstitial nephritis, BUN and serum creatinine elevation, toxic nephrosis with oliguria and anuria, and crystalluria.
Miscellaneous Reactions
Drug fever, chills, periarteritis nodosa and LE phenomenon have occurred.
The sulfonamides bear certain chemical similarities to some goitrogens, diuretics (acetazolamide and the thiazides), and oral hypoglycemic agents. Diuresis and hypoglycemia have occurred rarely in patients receiving sulfonamides. Cross-sensitivity may exist with these agents. Rats appear to be especially susceptible to the goitrogenic effects of sulfonamides, and long-term administration has produced thyroid malignancies in the species.
OVERDOSAGE
Acute intoxication may be manifested by headache, nausea, anorexia, vomiting and central nervous system stimulation (including convulsions), followed by megaloblastic anemia, leukopenia, thrombocytopenia, glossitis and crystalluria. In acute intoxication, emesis and gastric lavage followed by purges may be of benefit. The patient should be adequately hydrated to prevent renal damage. The renal, hepatic, and hematopoietic systems should be monitored for at least 1 month after an overdosage. If the patient is having convulsions, the use of parenteral diazepam or a barbiturate is indicated. For depressed platelet or white blood cell counts, folinic acid (leucovorin) should be administered in a dosage of 5 mg to 15 mg intramuscularly daily for 3 days or longer.
DOSAGE AND ADMINISTRATION
(See INDICATIONS AND USAGE)
The dosage should be swallowed whole, and not chewed, with plenty of fluids after a meal.
Treatment of Acute Malaria
| Adults | 2 to 3 tablets taken as a single dose. |
|---|---|
| Pediatric patients (>2 months to 18 years) | The dosage for treatment of malaria in children is based upon body weight: |
| Weight (kg) | Number of Tablets Taken as a Single Dose |
| >45 | 3 |
| 31 to 45 | 2 |
| 21 to 30 | 1 ½ |
| 11 to 20 | 1 |
| 5 to 10 | ½ |
Prevention of Malaria
The malaria risk must be carefully weighed against the risk of serious adverse drug reactions (see INDICATIONS AND USAGE). If Fansidar is prescribed for prophylaxis, it is important that the physician inquires about sulfonamide intolerance and points out the risk and the need for immediate drug withdrawal if skin reactions do occur.
The first dose of Fansidar should be taken 1 or 2 days before arrival in an endemic area; administration should be continued during the stay and for 4 to 6 weeks after return.
| Once Weekly | Once Every 2 Weeks |
|
|---|---|---|
| Adults | 1 tablet | 2 tablets |
| Pediatric patients (>2 months to 18 years) | The dosage for prevention of malaria in children is based upon body weight: | |
| Weight (kg) | Number of Tablets Taken Once Weekly |
|
| >45 | 1 ½ | |
| 31 to 45 | 1 | |
| 21 to 30 | ¾ | |
| 11 to 20 | ½ | |
| 5 to 10 | ¼ | |
Prophylaxis with Fansidar should not be continued for more than two years, since no experience of more prolonged administration is available to date.
HOW SUPPLIED
Scored tablets, containing 500 mg sulfadoxine and 25 mg pyrimethamine — unit dose packages of 25 (NDC-0004-0161-03). Imprint on tablets: FANSIDAR ((ROCHE LOGO)) ROCHE.
Manufactured by:
F. Hoffmann-La Roche Ltd.
Basel, Switzerland
Distributed by:

27898797
Copyright © 1996-2004 by Roche Laboratories Inc. All rights reserved.
| FANSIDAR
sulfadoxine and pyrimethamine tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA018557 | 10/28/1981 | 04/18/2012 |
| Labeler - Genentech, Inc. (080129000) |
Revised: 08/2012 Genentech, Inc.
