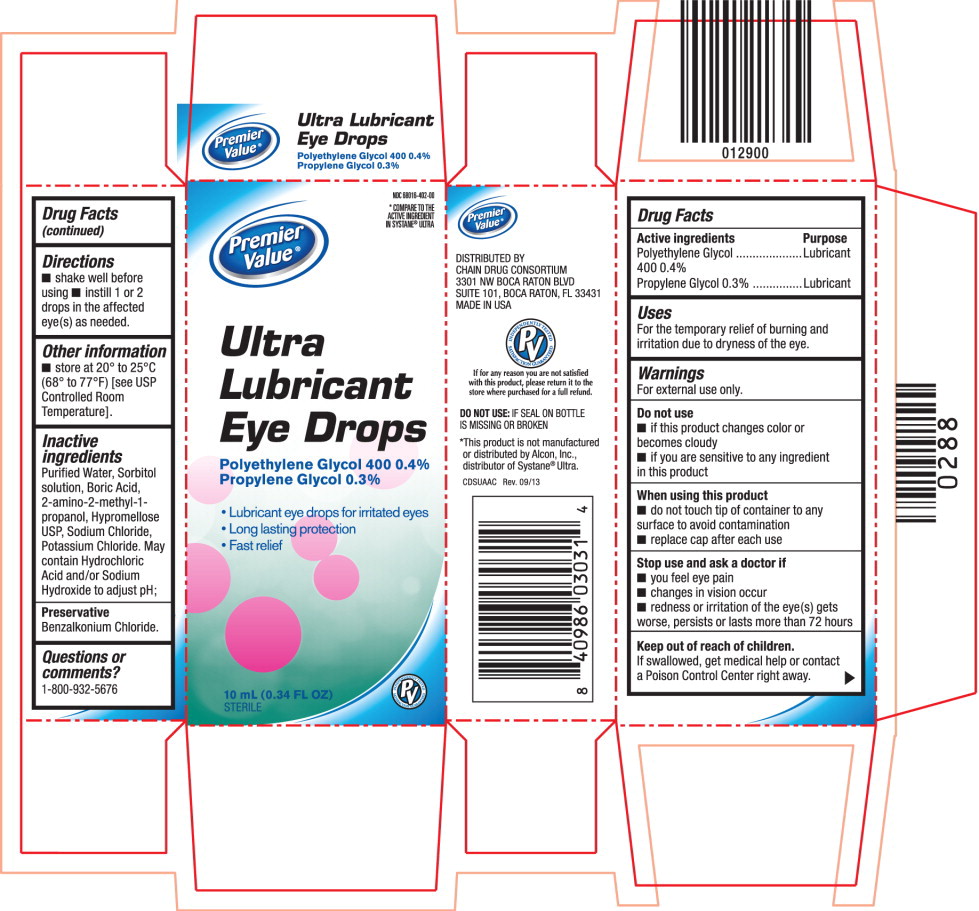
ULTRA LUBRICANT EYE DROPS- polyethylene glycol 400 and propylene glycol solution/ drops
Chain Drug Consortium
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Warnings
For external use only.
Do not use
- if this product changes color or becomes cloudy
- if you are sensitive to any ingredient in this product
When using this product
- do not touch tip of container to any surface to avoid contamination
- replace cap after each use
Inactive ingredients
Purified Water, Sorbitol solution, Boric Acid, 2-amino-2-methyl-1-propanol, Hypromellose USP, Sodium Chloride, Potassium Chloride. May contain Hydrochloric Acid and/or Sodium Hydroxide to adjust pH;
Principal Display Panel Text for Container Label:
NDC 68016-402-00
Premier
Value® Logo
Ultra Lubricant
Eye Drops
Polyethylene Glycol 400 0.4%
Propylene Glycol 0.3%
10 mL (0.34 FL OZ) STERILE
Principal Display Panel Text for Carton Label:
NDC 68016-402-00
*COMPARE TO THE
ACTIVE INGREDIENT
IN SYSTANE® ULTRA
Premier
Value® Logo
Ultra
Lubricant
Eye Drops
Polyethylene Glycol 400 0.4%
Propylene Glycol 0.3%
- Lubricant eye drops for irritated eyes
- Long lasting protection
- Fast relief
10 mL (0.34 FL OZ)
STERILE PV Brand Logo
| ULTRA LUBRICANT EYE DROPS
polyethylene glycol 400 and propylene glycol solution/ drops |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - Chain Drug Consortium (101668460) |