DOCETAXEL- docetaxel injection, solution, concentrate
XGen Pharmaceuticals DJB, Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use DOCETAXEL INJECTION USP (Injection Concentrate) safely and effectively. See full prescribing information for DOCETAXEL INJECTION USP (Injection Concentrate).
DOCETAXEL Injection (Injection Concentrate), Intravenous Infusion (IV). Initial U.S. Approval: 1996 See full prescribing information for complete boxed warning
INDICATIONS AND USAGEDocetaxel Injection USP (Injection Concentrate) is a microtubule inhibitor indicated for:
DOSAGE AND ADMINISTRATIONAdminister in a facility equipped to manage possible complications (e.g., anaphylaxis). Administer intravenously (IV) over 1 hr every 3 weeks. PVC equipment is not recommended. Use only a 21 gauge needle to withdraw Docetaxel Injection USP (Injection Concentrate) from the vial.
For all patients: DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONSWARNINGS AND PRECAUTIONS
ADVERSE REACTIONSMost common adverse reactions across all Docetaxel Injection USP (Injection Concentrate) indications are infections, neutropenia, anemia, febrile neutropenia, hypersensitivity, thrombocytopenia, neuropathy, dysgeusia, dyspnea, constipation, anorexia, nail disorders, fluid retention, asthenia, pain, nausea, diarrhea, vomiting, mucositis, alopecia, skin reactions, myalgia (6) To report SUSPECTED ADVERSE REACTIONS, contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch DRUG INTERACTIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 2/2018 |
FULL PRESCRIBING INFORMATION
WARNING: TOXIC DEATHS, HEPATOTOXICITY, NEUTROPENIA, HYPERSENSITIVITY REACTIONS, and FLUID RETENTION
The incidence of treatment-related mortality associated with Docetaxel Injection USP (Injection Concentrate) therapy is increased in patients with abnormal liver function, in patients receiving higher doses, and in patients with non-small cell lung carcinoma and a history of prior treatment with platinum-based chemotherapy who receive Docetaxel Injection USP (Injection Concentrate) as a single agent at a dose of 100 mg/m2[see Warnings and Precautions (5.1)].
Docetaxel Injection USP (Injection Concentrate) should not be given to patients with bilirubin > upper limit of normal (ULN), or to patients with AST and/or ALT >1.5 x ULN concomitant with alkaline phosphatase >2.5 x ULN. Patients with elevations of bilirubin or abnormalities of transaminase concurrent with alkaline phosphatase are at increased risk for the development of grade 4 neutropenia, febrile neutropenia, infections, severe thrombocytopenia, severe stomatitis, severe skin toxicity, and toxic death. Patients with isolated elevations of transaminase >1.5 x ULN also had a higher rate of febrile neutropenia grade 4 but did not have an increased incidence of toxic death. Bilirubin, AST or ALT, and alkaline phosphatase values should be obtained prior to each cycle of Docetaxel USP (Injection Concentrate) therapy [see Warnings and Precautions (5.2)].
Docetaxel Injection USP (Injection Concentrate) therapy should not be given to patients with neutrophil counts of <1500 cells/mm3. In order to monitor the occurrence of neutropenia, which may be severe and result in infection, frequent blood cell counts should be performed on all patients receiving Docetaxel Injection USP (Injection Concentrate) [see Warnings and Precautions (5.3)].
Severe hypersensitivity reactions characterized by generalized rash/erythema, hypotension and/or bronchospasm, or very rarely fatal anaphylaxis, have been reported in patients who received a 3-day dexamethasone premedication. Hypersensitivity reactions require immediate discontinuation of the Docetaxel Injection USP (Injection Concentrate) infusion and administration of appropriate therapy [see Warnings and Precautions (5.4)]. Docetaxel Injection USP (Injection Concentrate) must not be given to patients who have a history of severe hypersensitivity reactions to Docetaxel Injection USP (Injection Concentrate) or to other drugs formulated with polysorbate 80 [see Contraindications (4)].
Severe fluid retention occurred in 6.5% (6/92) of patients despite use of a 3-day dexamethasone premedication regimen. It was characterized by one or more of the following events: poorly tolerated peripheral edema, generalized edema, pleural effusion requiring urgent drainage, dyspnea at rest, cardiac tamponade, or pronounced abdominal distention (due to ascites) [see Warnings and Precautions (5.5)].
1. INDICATIONS AND USAGE
1.1 Breast Cancer
Docetaxel Injection USP (Injection Concentrate) is indicated for the treatment of patients with locally advanced or metastatic breast cancer after failure of prior chemotherapy.
Docetaxel Injection USP (Injection Concentrate) in combination with doxorubicin and cyclophosphamide is indicated for the adjuvant treatment of patients with operable node-positive breast cancer.
1.2 Non-Small Cell Lung Cancer
Docetaxel Injection USP (Injection Concentrate) as a single agent is indicated for the treatment of patients with locally advanced or metastatic non-small cell lung cancer after failure of prior platinum-based chemotherapy.
Docetaxel Injection USP (Injection Concentrate) in combination with cisplatin is indicated for the treatment of patients with unresectable, locally advanced or metastatic non-small cell lung cancer who have not previously received chemotherapy for this condition.
1.3 Prostate Cancer
Docetaxel Injection USP (Injection Concentrate) in combination with prednisone is indicated for the treatment of patients with androgen independent (hormone refractory) metastatic prostate cancer.
1.4 Gastric Adenocarcinoma
Docetaxel Injection USP (Injection Concentrate) in combination with cisplatin and fluorouracil is indicated for the treatment of patients with advanced gastric adenocarcinoma, including adenocarcinoma of the gastroesophageal junction, who have not received prior chemotherapy for advanced disease.
2. DOSAGE AND ADMINISTRATION
For all indications, toxicities may warrant dosage adjustments [see Dosage and Administration (2.7)].
Administer in a facility equipped to manage possible complications (e.g. anaphylaxis).
2.1 Breast Cancer
- For locally advanced or metastatic breast cancer after failure of prior chemotherapy, the recommended dose of Docetaxel Injection USP (Injection Concentrate) is 60 mg/m2 to 100 mg/m2 administered intravenously over 1 hour every 3 weeks.
- For the adjuvant treatment of operable node-positive breast cancer, the recommended Docetaxel Injection USP (Injection Concentrate) dose is 75 mg/m2 administered 1 hour after doxorubicin 50 mg/m2 and cyclophosphamide 500 mg/m2 every 3 weeks for 6 courses. Prophylactic G-CSF may be used to mitigate the risk of hematological toxicities [see Dosage and Administration (2.7)].
2.2 Non-Small Cell Lung Cancer
- For treatment after failure of prior platinum-based chemotherapy, Docetaxel Injection USP (Injection Concentrate) was evaluated as monotherapy, and the recommended dose is 75 mg/m2 administered intravenously over 1 hour every 3 weeks. A dose of 100 mg/m2 in patients previously treated with chemotherapy was associated with increased hematologic toxicity, infection, and treatment-related mortality in randomized, controlled trials [see Boxed Warning, Dosage and Administration (2.7), Warnings and Precautions (5), Clinical Studies (14)].
- For chemotherapy-naïve patients, Docetaxel Injection USP (Injection Concentrate) was evaluated in combination with cisplatin. The recommended dose of Docetaxel Injection USP (Injection Concentrate) is 75 mg/m2 administered intravenously over 1 hour immediately followed by cisplatin 75 mg/m2 over 30-60 minutes every 3 weeks [see Dosage and Administration (2.7)].
2.3 Prostate Cancer
- For hormone-refractory metastatic prostate cancer, the recommended dose of Docetaxel Injection USP (Injection Concentrate) is 75 mg/m2 every 3 weeks as a 1 hour intravenous infusion. Prednisone 5 mg orally twice daily is administered continuously [see Dosage and Administration (2.7)].
2.4 Gastric Adenocarcinoma
- For gastric adenocarcinoma, the recommended dose of Docetaxel Injection USP (Injection Concentrate) is 75 mg/m2 as a 1 hour intravenous infusion, followed by cisplatin 75 mg/m2, as a 1 to 3 hour intravenous infusion (both on day 1 only), followed by fluorouracil 750 mg/m2 per day given as a 24-hour continuous intravenous infusion for 5 days, starting at the end of the cisplatin infusion. Treatment is repeated every three weeks. Patients must receive premedication with antiemetics and appropriate hydration for cisplatin administration [see Dosage and Administration (2.7)].
2.5 Head and Neck Cancer
Patients must receive premedication with antiemetics, and appropriate hydration (prior to and after cisplatin administration). Prophylaxis for neutropenic infections should be administered. All patients treated on the Docetaxel Injection USP (Injection Concentrate) containing arms of the TAX323 and TAX324 studies received prophylactic antibiotics.
-
Induction chemotherapy followed by radiotherapy (TAX323)
For the induction treatment of locally advanced inoperable SCCHN, the recommended dose of Docetaxel Injection USP (Injection Concentrate) is 75 mg/m2 as a 1 hour intravenous infusion followed by cisplatin 75 mg/m2 intravenously over 1 hour, on day one, followed by fluorouracil as a continuous intravenous infusion at 750 mg/m2 per day for five days. This regimen is administered every 3 weeks for 4 cycles. Following chemotherapy, patients should receive radiotherapy. [see Dosage and Administration (2.7)].
-
Induction chemotherapy followed by chemoradiotherapy (TAX324)
For the induction treatment of patients with locally advanced (unresectable, low surgical cure, or organ preservation) SCCHN, the recommended dose of Docetaxel Injection USP (Injection Concentrate) is 75 mg/m2 as a 1 hour intravenous infusion on day 1, followed by cisplatin 100 mg/m2 administered as a 30-minute to 3 hour infusion, followed by fluorouracil 1000 mg/m2/day as a continuous infusion from day 1 to day 4. This regimen is administered every 3 weeks for 3 cycles. Following chemotherapy, patients should receive chemoradiotherapy [see Dosage and Administration (2.7)].
2.6 Premedication Regimen
All patients should be premedicated with oral corticosteroids (see below for prostate cancer) such as dexamethasone 16 mg per day (e.g., 8 mg twice daily) for 3 days starting 1 day prior to Docetaxel Injection USP (Injection Concentrate) administration in order to reduce the incidence and severity of fluid retention as well as the severity of hypersensitivity reactions [see Boxed Warning, Warnings and Precautions (5.4)].
For hormone-refractory metastatic prostate cancer, given the concurrent use of prednisone, the recommended premedication regimen is oral dexamethasone 8 mg, at 12 hours, 3 hours and 1 hour before the Docetaxel Injection USP (Injection Concentrate) infusion [see Warnings and Precautions (5.4)].
2.7 Dosage Adjustments During Treatment
Breast Cancer
Patients who are dosed initially at 100 mg/m2 and who experience either febrile neutropenia, neutrophils <500 cells/mm3 for more than 1 week, or severe or cumulative cutaneous reactions during Docetaxel Injection USP (Injection Concentrate) therapy should have the dosage adjusted from 100 mg/m2 to 75 mg/m2. If the patient continues to experience these reactions, the dosage should either be decreased from 75 mg/m2 to 55 mg/m2 or the treatment should be discontinued. Conversely, patients who are dosed initially at 60 mg/m2 and who do not experience febrile neutropenia, neutrophils <500 cells/mm3 for more than 1 week, severe or cumulative cutaneous reactions, or severe peripheral neuropathy during Docetaxel Injection USP (Injection Concentrate) therapy may tolerate higher doses. Patients who develop ≥grade 3 peripheral neuropathy should have Docetaxel Injection USP (Injection Concentrate) treatment discontinued entirely.
Combination Therapy with Docetaxel Injection USP (Injection Concentrate) in the Adjuvant Treatment of Breast Cancer
Docetaxel Injection USP (Injection Concentrate) in combination with doxorubicin and cyclophosphamide should be administered when the neutrophil count is ≥1,500 cells/mm3. Patients who experience febrile neutropenia should receive G-CSF in all subsequent cycles. Patients who continue to experience this reaction should remain on G-CSF and have their Docetaxel Injection USP (Injection Concentrate) dose reduced to 60 mg/m2. Patients who experience grade 3 or 4 stomatitis should have their Docetaxel Injection USP (Injection Concentrate) dose decreased to 60 mg/m2. Patients who experience severe or cumulative cutaneous reactions or moderate neurosensory signs and/or symptoms during Docetaxel Injection USP (Injection Concentrate) therapy should have their dosage of Docetaxel Injection USP (Injection Concentrate) reduced from 75 mg/m2 to 60 mg/m2. If the patient continues to experience these reactions at 60 mg/m2, treatment should be discontinued.
Non-Small Cell Lung Cancer
Monotherapy with Docetaxel Injection USP (Injection Concentrate) for NSCLC treatment after failure of prior platinum-based chemotherapy
Patients who are dosed initially at 75 mg/m2 and who experience either febrile neutropenia, neutrophils <500 cells/mm3 for more than one week, severe or cumulative cutaneous reactions, or other grade 3/4 non-hematological toxicities during Docetaxel Injection USP (Injection Concentrate) treatment should have treatment withheld until resolution of the toxicity and then resumed at 55 mg/m2. Patients who develop ≥grade 3 peripheral neuropathy should have Docetaxel Injection USP (Injection Concentrate) treatment discontinued entirely.
Combination therapy with Docetaxel Injection USP (Injection Concentrate) for chemotherapy-naïve NSCLC
For patients who are dosed initially at Docetaxel Injection USP (Injection Concentrate) 75 mg/m2 in combination with cisplatin, and whose nadir of platelet count during the previous course of therapy is <25,000 cells/mm3, in patients who experience febrile neutropenia, and in patients with serious non-hematologic toxicities, the Docetaxel Injection USP (Injection Concentrate) dosage in subsequent cycles should be reduced to 65 mg/m2. In patients who require a further dose reduction, a dose of 50 mg/m2 is recommended. For cisplatin dosage adjustments, see manufacturer’s prescribing information.
Prostate Cancer
Combination therapy with Docetaxel Injection USP (Injection Concentrate) for hormone-refractory metastatic prostate cancer
Docetaxel Injection USP (Injection Concentrate) should be administered when the neutrophil count is ≥1,500 cells/mm3. Patients who experience either febrile neutropenia, neutrophils <500 cells/mm3 for more than one week, severe or cumulative cutaneous reactions or moderate neurosensory signs and/or symptoms during Docetaxel Injection USP (Injection Concentrate) therapy should have the dosage of Docetaxel Injection USP (Injection Concentrate) reduced from 75 mg/m2 to 60 mg/m2. If the patient continues to experience these reactions at 60 mg/m2, the treatment should be discontinued.
Gastric or Head and Neck Cancer
Docetaxel Injection USP (Injection Concentrate) in combination with cisplatin and fluorouracil in gastric cancer or head and neck cancer
Patients treated with Docetaxel Injection USP (Injection Concentrate) in combination with cisplatin and fluorouracil must receive antiemetics and appropriate hydration according to current institutional guidelines. In both studies, G-CSF was recommended during the second and/or subsequent cycles in case of febrile neutropenia, or documented infection with neutropenia, or neutropenia lasting more than 7 days. If an episode of febrile neutropenia, prolonged neutropenia or neutropenic infection occurs despite G-CSF use, the Docetaxel Injection USP (Injection Concentrate) dose should be reduced from 75 mg/m2 to 60 mg/m2. If subsequent episodes of complicated neutropenia occur the Docetaxel Injection USP (Injection Concentrate) dose should be reduced from 60 mg/m2 to 45 mg/m2. In case of grade 4 thrombocytopenia the Docetaxel Injection USP (Injection Concentrate) dose should be reduced from 75 mg/m2 to 60 mg/m2. Patients should not be retreated with subsequent cycles of Docetaxel Injection USP (Injection Concentrate) until neutrophils recover to a level >1,500 cells/mm3 and platelets recover to a level >100,000 cells/mm3. Discontinue treatment if these toxicities persist. [see Warnings and Precautions (5.3)].
Recommended dose modifications for toxicities in patients treated with Docetaxel Injection USP (Injection Concentrate) in combination with cisplatin and fluorouracil are shown in Table 1.
| Toxicity | Dosage adjustment |
| Diarrhea grade 3 | First episode: reduce fluorouracil dose by 20%. Second episode: then reduce Docetaxel Injection USP (Injection Concentrate) dose by 20%. |
| Diarrhea grade 4 | First episode: reduce Docetaxel Injection USP (Injection Concentrate) and fluorouracil doses by 20%. Second episode: discontinue treatment. |
| Stomatitis/mucositis grade 3 | First episode: reduce fluorouracil dose by 20%. Second episode: stop fluorouracil only, at all subsequent cycles. Third episode: reduce Docetaxel Injection USP (Injection Concentrate) dose by 20%. |
| Stomatitis/mucositis grade 4 | First episode: stop fluorouracil only, at all subsequent cycles. Second episode: reduce Docetaxel Injection USP (Injection Concentrate) dose by 20%. |
Liver dysfunction:
In case of AST/ALT >2.5 to ≤5 x ULN and AP ≤2.5 x ULN, or AST/ALT >1.5 to ≤5 x ULN and AP >2.5 to ≤5 x ULN, Docetaxel Injection USP (Injection Concentrate) should be reduced by 20%.
In case of AST/ALT >5 x ULN and/or AP >5 x ULN Docetaxel Injection USP (Injection Concentrate) should be stopped.
The dose modifications for cisplatin and fluorouracil in the gastric cancer study are provided below:
Cisplatin dose modifications and delays
Peripheral neuropathy: A neurological examination should be performed before entry into the study, and then at least every 2 cycles and at the end of treatment. In the case of neurological signs or symptoms, more frequent examinations should be performed and the following dose modifications can be made according to NCIC-CTC grade:
- Grade 2: Reduce cisplatin dose by 20%.
- Grade 3: Discontinue treatment.
Ototoxicity: In the case of grade 3 toxicity, discontinue treatment.
Nephrotoxicity: In the event of a rise in serum creatinine ≥grade 2 (>1.5 x normal value) despite adequate rehydration, CrCl should be determined before each subsequent cycle and the following dose reductions should be considered (see Table 2).
For other cisplatin dosage adjustments, also refer to the manufacturers’ prescribing information.
| CrCl = Creatinine clearance | |
| Creatinine clearance result before next cycle | Cisplatin dose next cycle |
| CrCl ≥60 mL/min | Full dose of cisplatin was given. CrCl was to be repeated before each treatment cycle. |
| CrCl between 40 and 59 mL/min | Dose of cisplatin was reduced by 50% at subsequent cycle. If CrCl was >60 mL/min at end of cycle, full cisplatin dose was reinstituted at the next cycle. If no recovery was observed, then cisplatin was omitted from the next treatment cycle. |
| CrCl <40 mL/min | Dose of cisplatin was omitted in that treatment cycle only. If CrCl was still <40 mL/min at the end of cycle, cisplatin was discontinued. If CrCl was >40 and <60 mL/min at end of cycle, a 50% cisplatin dose was given at the next cycle. If CrCl was >60 mL/min at end of cycle, full cisplatin dose was given at next cycle. |
Fluorouracil dose modifications and treatment delays
For diarrhea and stomatitis, see Table 1.
In the event of grade 2 or greater plantar-palmar toxicity, fluorouracil should be stopped until recovery. The fluorouracil dosage should be reduced by 20%.
For other greater than grade 3 toxicities, except alopecia and anemia, chemotherapy should be delayed (for a maximum of 2 weeks from the planned date of infusion) until resolution to grade ≤1 and then recommenced, if medically appropriate.
For other fluorouracil dosage adjustments, also refer to the manufacturers’ prescribing information.
Combination Therapy with Strong CYP3A4 inhibitors:
Avoid using concomitant strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, clarithromycin, atazanavir, indinavir, nefazodone, nelfinavir, ritonavir, saquinavir, telithromycin and voriconazole). There are no clinical data with a dose adjustment in patients receiving strong CYP3A4 inhibitors. Based on extrapolation from a pharmacokinetic study with ketoconazole in 7 patients, consider a 50% docetaxel dose reduction if patients require co-administration of a strong CYP3A4 inhibitor. [see Drug Interactions (7),Clinical Pharmacology (12.3)].
2.8 Administration Precautions
Docetaxel Injection USP (Injection Concentrate) is a cytotoxic anticancer drug and, as with other potentially toxic compounds, caution should be exercised when handling and preparing Docetaxel Injection USP (Injection Concentrate) solutions. The use of gloves is recommended. Please refer to [see How Supplied/ Storage and Handling (16.3)].
If Docetaxel Injection USP (Injection Concentrate), initial diluted solution, or final dilution for infusion should come into contact with the skin, immediately and thoroughly wash with soap and water. If Docetaxel Injection USP (Injection Concentrate), initial diluted solution, or final dilution for infusion should come into contact with mucosa, immediately and thoroughly wash with water.
Contact of the Docetaxel Injection USP (Injection Concentrate) concentrate with plasticized PVC equipment or devices used to prepare solutions for infusion is not recommended. In order to minimize patient exposure to the plasticizer DEHP (di-2-ethylhexyl phthalate), which may be leached from PVC infusion bags or sets, the final Docetaxel Injection USP (Injection Concentrate) dilution for infusion should be stored in bottles (glass, polypropylene) or plastic bags (polypropylene, polyolefin) and administered through polyethylene-lined administration sets.
One-vial Docetaxel Injection USP (Injection Concentrate)
Docetaxel Injection USP (Injection Concentrate) requires NO prior dilution with a diluent and is ready to add to the infusion solution.
Please follow the preparation instructions provided below.
2.9 Preparation and Administration
DO NOT use the two-vial formulation (Injection Concentrate and diluent) with the one-vial formulation.
One-vial Docetaxel Injection USP (Injection Concentrate)
Docetaxel Injection USP (Injection Concentrate) (20 mg/mL) requires NO prior dilution with a diluent and is ready to add to the infusion solution. Use only a 21 gauge needle to withdraw Docetaxel Injection USP (Injection Concentrate) from the vial because larger bore needles (e.g., 18 and 19 gauge) may result in stopper coring and rubber particulates.
- Docetaxel Injection USP (Injection Concentrate) vials should be stored between 2 and 25°C (36 and 77°F). If the vials are stored under refrigeration, allow the appropriate number of vials of Docetaxel Injection USP (Injection Concentrate) vials to stand at room temperature for approximately 5 minutes before use.
- Using only a 21 gauge needle, aseptically withdraw the required amount of Docetaxel Injection USP (Injection Concentrate) (20 mg docetaxel/mL) with a calibrated syringe and inject via a single injection (one shot) into a 250 mL infusion bag or bottle of either 0.9% Sodium Chloride solution or 5% Dextrose solution to produce a final concentration of 0.3 mg/mL to 0.74 mg/mL.
If a dose greater than 200 mg of Docetaxel Injection USP (Injection Concentrate) is required, use a larger volume of the infusion vehicle so that a concentration of 0.74 mg/mL Docetaxel Injection USP (Injection Concentrate) is not exceeded. - Thoroughly mix the infusion by gentle manual rotation.
- As with all parenteral products, Docetaxel Injection USP (Injection Concentrate) should be inspected visually for particulate matter or discoloration prior to administration whenever the solution and container permit. If the Docetaxel Injection USP (Injection Concentrate) dilution for intravenous infusion is not clear or appears to have precipitation, it should be discarded.
- Docetaxel Injection USP (Injection Concentrate) infusion solution is supersaturated, therefore may crystallize over time. If crystals appear, the solution must no longer be used and shall be discarded.
The Docetaxel Injection USP (Injection Concentrate) dilution for infusion should be administered intravenously as a 1-hour infusion under ambient room temperature (below 25°C) and lighting conditions.
2.10 Stability
Docetaxel Injection USP (Injection Concentrate) final dilution for infusion, if stored between 2°C and 25°C (36°F and 77°F) is stable for 6 hours. Docetaxel Injection USP (Injection Concentrate) final dilution for infusion (in either 0.9% Sodium Chloride solution or 5% Dextrose solution) should be used within 6 hours (including the 1 hour intravenous administration).
In addition, physical and chemical in-use stability of the infusion solution prepared as recommended has been demonstrated in non-PVC bags up to 48 hours when stored between 2°C and 8°C (36 and 46°F).
3. DOSAGE FORMS AND STRENGTHS
One-vial Docetaxel Injection USP (Injection Concentrate)
Docetaxel Injection USP (Injection Concentrate) 20 mg/mL
Docetaxel Injection USP (Injection Concentrate) 20 mg/1 mL: 20 mg docetaxel in 1 mL in 50/50 (v/v) ratio polysorbate 80/dehydrated alcohol, and anhydrous citric acid.
Docetaxel Injection USP (Injection Concentrate) 80 mg/4 mL (20 mg/mL)
Docetaxel Injection USP (Injection Concentrate) 80 mg/4 mL: 80 mg docetaxel in 4 mL 50/50 (v/v) ratio polysorbate 80/dehydrated alcohol, and anhydrous citric acid.
Docetaxel Injection USP (Injection Concentration) 200 mg/10 mL (20 mg/mL)
Docetaxel Injection USP (Injection Concentrate) 200 mg/10 mL: 200 mg docetaxel in 10 mL 50/50 (v/v) ratio polysorbate 80/dehydrated alcohol, and anhydrous citric acid.
4. CONTRAINDICATIONS
- Docetaxel Injection USP (Injection Concentrate) is contraindicated in patients who have a history of severe hypersensitivity reactions to docetaxel or to other drugs formulated with polysorbate 80. Severe reactions, including anaphylaxis, have occurred [see Warnings and Precautions (5.4)].
- Docetaxel Injection USP (Injection Concentrate) should not be used in patients with neutrophil counts of <1500 cells/mm3.
5. WARNINGS AND PRECAUTIONS
5.1 Toxic Deaths
Breast Cancer
Docetaxel Injection USP (Injection Concentrate) administered at 100 mg/m2 was associated with deaths considered possibly or probably related to treatment in 2.0% (19/965) of metastatic breast cancer patients, both previously treated and untreated, with normal baseline liver function and in 11.5% (7/61) of patients with various tumor types who had abnormal baseline liver function (AST and/or ALT >1.5 times ULN together with AP >2.5 times ULN). Among patients dosed at 60 mg/m2, mortality related to treatment occurred in 0.6% (3/481) of patients with normal liver function, and in 3 of 7 patients with abnormal liver function. Approximately half of these deaths occurred during the first cycle. Sepsis accounted for the majority of the deaths.
Non-Small Cell Lung Cancer
Docetaxel Injection USP (Injection Concentrate) administered at a dose of 100 mg/m2 in patients with locally advanced or metastatic non-small cell lung cancer who had a history of prior platinum-based chemotherapy was associated with increased treatment-related mortality (14% and 5% in two randomized, controlled studies). There were 2.8% treatment-related deaths among the 176 patients treated at the 75 mg/m2 dose in the randomized trials. Among patients who experienced treatment-related mortality at the 75 mg/m2 dose level, 3 of 5 patients had an ECOG PS of 2 at study entry [see Dosage and Administration (2.2),Clinical Studies (14)].
5.2 Hepatic Impairment
Patients with combined abnormalities of transaminases and alkaline phosphatase should not be treated with Docetaxel Injection USP (Injection Concentrate) [see Boxed Warning, Use in Specific Populations (8.6), Clinical studies (14)].
5.3 Hematologic Effects
Perform frequent peripheral blood cell counts on all patients receiving Docetaxel Injection USP (Injection Concentrate). Patients should not be retreated with subsequent cycles of Docetaxel Injection USP (Injection Concentrate) until neutrophils recover to a level >1500 cells/ mm3 and platelets recover to a level > 100,000 cells/ mm3.
A 25% reduction in the dose of Docetaxel Injection USP (Injection Concentrate) is recommended during subsequent cycles following severe neutropenia (<500 cells/ mm3) lasting 7 days or more, febrile neutropenia, or a grade 4 infection in a Docetaxel Injection USP cycle [see Dosage and Administration (2.7)].
Neutropenia (<2000 neutrophils/ mm3) occurs in virtually all patients given 60 mg/m2 to 100 mg/m2 of Docetaxel Injection USP (Injection Concentrate) and grade 4 neutropenia (<500 cells/ mm3) occurs in 85% of patients given 100 mg/m2 and 75% of patients given 60 mg/m2. Frequent monitoring of blood counts is, therefore, essential so that dose can be adjusted. Docetaxel Injection USP (Injection Concentrate) should not be administered to patients with neutrophils <1500 cells/ mm3.
Febrile neutropenia occurred in about 12% of patients given 100 mg/m2 but was very uncommon in patients given 60 mg/m2. Hematologic responses, febrile reactions and infections, and rates of septic death for different regimens are dose related [see Adverse Reactions (6.1), Clinical Studies (14)].
Three breast cancer patients with severe liver impairment (bilirubin >1.7 times ULN) developed fatal gastrointestinal bleeding associated with severe drug-induced thrombocytopenia. In gastric cancer patients treated with docetaxel in combination with cisplatin and fluorouracil (TCF), febrile neutropenia and/or neutropenic infection occurred in 12% of patients receiving G-CSF compared to 28% who did not. Patients receiving TCF should be closely monitored during the first and subsequent cycles for febrile neutropenia and neutropenic infection [see Dosage and Administration (2.7), Adverse Reactions (6)].
5.4 Hypersensitivity Reactions
Patients should be observed closely for hypersensitivity reactions, especially during the first and second infusions. Severe hypersensitivity reactions characterized by generalized rash/erythema, hypotension and/or bronchospasm, or very rarely fatal anaphylaxis, have been reported in patients premedicated with 3 days of corticosteroids. Severe hypersensitivity reactions require immediate discontinuation of the Docetaxel Injection USP (Injection Concentrate) infusion and aggressive therapy. Patients with a history of severe hypersensitivity reactions should not be rechallenged with Docetaxel Injection USP (Injection Concentrate).
Hypersensitivity reactions may occur within a few minutes following initiation of a Docetaxel Injection USP (Injection Concentrate) infusion. If minor reactions such as flushing or localized skin reactions occur, interruption of therapy is not required. All patients should be premedicated with an oral corticosteroid prior to the initiation of the infusion of Docetaxel Injection USP (Injection Concentrate) [see Dosage and Administration (2.6)].
5.5 Fluid Retention
Severe fluid retention has been reported following Docetaxel Injection USP (Injection Concentrate) therapy. Patients should be premedicated with oral corticosteroids prior to each Docetaxel Injection USP (Injection Concentrate) administration to reduce the incidence and severity of fluid retention [see Dosage and Administration (2.6)]. Patients with pre-existing effusions should be closely monitored from the first dose for the possible exacerbation of the effusions.
When fluid retention occurs, peripheral edema usually starts in the lower extremities and may become generalized with a median weight gain of 2 kg.
Among 92 breast cancer patients premedicated with 3-day corticosteroids, moderate fluid retention occurred in 27.2% and severe fluid retention in 6.5%. The median cumulative dose to onset of moderate or severe fluid retention was 819 mg/m2. Nine of 92 patients (9.8%) of patients discontinued treatment due to fluid retention: 4 patients discontinued with severe fluid retention; the remaining 5 had mild or moderate fluid retention. The median cumulative dose to treatment discontinuation due to fluid retention was 1021 mg/m2. Fluid retention was completely, but sometimes slowly, reversible with a median of 16 weeks from the last infusion of Docetaxel Injection USP (Injection Concentrate) to resolution (range: 0 to 42+ weeks). Patients developing peripheral edema may be treated with standard measures, e.g., salt restriction, oral diuretic(s).
5.6 Acute Myeloid Leukemia
Treatment-related acute myeloid leukemia (AML) or myelodysplasia has occurred in patients given anthracyclines and/or cyclophosphamide, including use in adjuvant therapy for breast cancer. In the adjuvant breast cancer trial (TAX316) AML occurred in 3 of 744 patients who received Docetaxel Injection USP (Injection Concentrate), doxorubicin and cyclophosphamide (TAC) and in 1 of 736 patients who received fluorouracil, doxorubicin and cyclophosphamide [see Clinical Studies (14.2)]. In TAC-treated patients, the risk of delayed myelodysplasia or myeloid leukemia requires hematological follow-up.
5.7 Cutaneous Reactions
Localized erythema of the extremities with edema followed by desquamation has been observed. In case of severe skin toxicity, an adjustment in dosage is recommended [see Dosage and Administration (2.7)]. The discontinuation rate due to skin toxicity was 1.6% (15/965) for metastatic breast cancer patients. Among 92 breast cancer patients premedicated with 3-day corticosteroids, there were no cases of severe skin toxicity reported and no patient discontinued Docetaxel Injection USP (Injection Concentrate) due to skin toxicity.
5.8 Neurologic Reactions
Severe neurosensory symptoms (e.g. paresthesia, dysesthesia, pain) were observed in 5.5% (53/965) of metastatic breast cancer patients, and resulted in treatment discontinuation in 6.1%. When these symptoms occur, dosage must be adjusted. If symptoms persist, treatment should be discontinued [see Dosage and Administration (2.7)]. Patients who experienced neurotoxicity in clinical trials and for whom follow-up information on the complete resolution of the event was available had spontaneous reversal of symptoms with a median of 9 weeks from onset (range: 0 to 106 weeks). Severe peripheral motor neuropathy mainly manifested as distal extremity weakness occurred in 4.4% (42/965).
5.9 Eye Disorders
Cystoid macular edema (CME) has been reported in patients treated with Docetaxel Injection USP (Injection Concentrate). Patients with impaired vision should undergo a prompt and comprehensive ophthalmologic examination. If CME is diagnosed, Docetaxel Injection USP (Injection Concentrate) treatment should be discontinued and appropriate treatment initiated. Alternative non-taxane cancer treatment should be considered.
5.10 Asthenia
Severe asthenia has been reported in 14.9% (144/965) of metastatic breast cancer patients but has led to treatment discontinuation in only 1.8%. Symptoms of fatigue and weakness may last a few days up to several weeks and may be associated with deterioration of performance status in patients with progressive disease.
5.11 Alcohol Content
Cases of intoxication have been reported with some formulations of docetaxel due to the alcohol content. The alcohol content in a dose of Docetaxel Injection USP (Injection Concentrate) Injection may affect the central nervous system and should be taken into account for patients in whom alcohol intake should be avoided or minimized. Consideration should be given to the alcohol content in Docetaxel Injection USP (Injection Concentrate) Injection on the ability to drive or use machines immediately after the infusion. Each administration of Docetaxel Injection USP (Injection Concentrate) Injection at 100 mg/m2 delivers 2.0 g/m2 of ethanol. For a patient with a BSA of 2.0 m2, this would deliver 4.0 grams of ethanol [see Description (11)]. Other docetaxel products may have a different amount of alcohol.
5.12 Use in Pregnancy
Docetaxel Injection USP (Injection Concentrate) can cause fetal harm when administered to a pregnant woman. Docetaxel caused embryofetal toxicities including intrauterine mortality when administered to pregnant rats and rabbits during the period of organogenesis. Embryofetal effects in animals occurred at doses as low as 1/50 and 1/300 the recommended human dose on a body surface area basis.
There are no adequate and well-controlled studies in pregnant women using Docetaxel Injection USP (Injection Concentrate). If Docetaxel Injection USP (Injection Concentrate) is used during pregnancy, or if the patient becomes pregnant while receiving this drug, the patient should be apprised of the potential hazard to the fetus. Women of childbearing potential should be advised to avoid becoming pregnant during therapy with Docetaxel Injection USP (Injection Concentrate) [see Use in Specific Populations (8.1)].
6. ADVERSE REACTIONS
The most serious adverse reactions from Docetaxel Injection USP (Injection Concentrate) are:
- Toxic Deaths [see Boxed Warning, Warning and Precautions (5.1)]
- Hepatotoxicity [see Boxed Warning, Warnings and Precautions (5.2)]
- Neutropenia [see Boxed Warning, Warnings and Precautions (5.3)]
- Hypersensitivity [see Boxed Warning, Warnings and Precautions (5.4)]
- Fluid Retention [see Boxed Warning, Warnings and Precautions (5.5)]
- Acute Myeloid Leukemia [see Boxed Warning, Warnings and Precautions (5.6)]
- Cutaneous Reactions [see Boxed Warning, Warnings and Precautions (5.7)]
- Neurologic Reactions [see Boxed Warning, Warnings and Precautions (5.8)]
- Eye Disorders [see Boxed Warning, Warnings and Precautions (5.9)]
- Asthenia [see Boxed Warning, Warnings and Precautions (5.10)]
- Alcohol Intoxication [see Boxed Warning, Warnings and Precautions (5.11)]
The most common adverse reactions across all Docetaxel Injection USP (Injection Concentrate) indications are infections, neutropenia, anemia, febrile neutropenia, hypersensitivity, thrombocytopenia, neuropathy, dysgeusia, dyspnea, constipation, anorexia, nail disorders, fluid retention, asthenia, pain, nausea, diarrhea, vomiting, mucositis, alopecia, skin reactions, and myalgia. Incidence varies depending on the indication.
Adverse reactions are described according to indication. Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Responding patients may not experience an improvement in performance status on therapy and may experience worsening. The relationship between changes in performance status, response to therapy, and treatment-related side effects has not been established.
6.1 Clinical Trial Experience
Breast Cancer
Monotherapy with Docetaxel Injection USP (Injection Concentrate) for locally advanced or metastatic breast cancer after failure of prior chemotherapy
Docetaxel Injection USP (Injection Concentrate) 100 mg/m2: Adverse drug reactions occurring in at least 5% of patients are compared for three populations who received Docetaxel Injection USP (Injection Concentrate) administered at 100 mg/m2 as a 1-hour infusion every 3 weeks: 2045 patients with various tumor types and normal baseline liver function tests; the subset of 965 patients with locally advanced or metastatic breast cancer, both previously treated and untreated with chemotherapy, who had normal baseline liver function tests; and an additional 61 patients with various tumor types who had abnormal liver function tests at baseline. These reactions were described using COSTART terms and were considered possibly or probably related to Docetaxel Injection USP (Injection Concentrate). At least 95% of these patients did not receive hematopoietic support. The safety profile is generally similar in patients receiving Docetaxel Injection USP (Injection Concentrate) for the treatment of breast cancer and in patients with other tumor types (See Table 3).
| *Normal Baseline LFTs: Transaminases ≤1.5 times ULN or alkaline phosphatase ≤2.5 times ULN or isolated elevations of transaminases or alkaline phosphatase up to 5 times ULN **Elevated Baseline LFTs: AST and/or ALT >1.5 times ULN concurrent with alkaline phosphatase >2.5 times ULN ***Febrile Neutropenia: ANC grade 4 with fever >38°C with intravenous antibiotics and/or hospitalization |
|||
| Adverse Reaction | All Tumor Types
Normal LFTs* n=2045 % | All Tumor Types
Elevated LFTs** n=61 % | Breast Cancer
Normal LFTs* n=965 % |
| Hematologic | |||
| Neutropenia | |||
| <2000 cells/mm3 | 96 | 96 | 99 |
| <500 cells/mm3 | 75 | 88 | 86 |
| Leukopenia | |||
| <4000 cells/mm3 | 96 | 98 | 99 |
| <1000 cells/mm3 | 32 | 47 | 44 |
| Thrombocytopenia | |||
| <100,000 cells/mm3 | 8 | 25 | 9 |
| Anemia | |||
| <11 g/dL | 90 | 92 | 94 |
| <8 g/dL | 9 | 31 | 8 |
| Febrile Neutropenia*** | 11 | 26 | 12 |
| Septic Death | 2 | 5 | 1 |
| Non-Septic Death | 1 | 7 | 1 |
| Infections | |||
| Any | 22 | 33 | 22 |
| Severe | 6 | 16 | 6 |
| Fever in Absence of Infection | |||
| Any | 31 | 41 | 35 |
| Severe | 2 | 8 | 2 |
| Hypersensitivity Reactions | |||
| Regardless of Premedication | |||
| Any | 21 | 20 | 18 |
| Severe | 4 | 10 | 3 |
| With 3-day Premedication | n=92 | n=3 | n=92 |
| Any | 15 | 33 | 15 |
| Severe | 2 | 0 | 2 |
| Fluid Retention | |||
| Regardless of Premedication | |||
| Any | 47 | 39 | 60 |
| Severe | 7 | 8 | 9 |
| With 3-day Premedication | n=92 | n=3 | n=92 |
| Any | 64 | 67 | 64 |
| Severe | 7 | 33 | 7 |
| Neurosensory | |||
| Any | 49 | 34 | 58 |
| Severe | 4 | 0 | 6 |
| Cutaneous | |||
| Any | 48 | 54 | 47 |
| Severe | 5 | 10 | 5 |
| Nail Changes | |||
| Any | 31 | 23 | 41 |
| Severe | 3 | 5 | 4 |
| Gastrointestinal | |||
| Nausea | 39 | 38 | 42 |
| Vomiting | 22 | 23 | 23 |
| Diarrhea | 39 | 33 | 43 |
| Severe | 5 | 5 | 6 |
| Stomatitis | |||
| Any | 42 | 49 | 52 |
| Severe | 6 | 13 | 7 |
| Alopecia | 76 | 62 | 74 |
| Asthenia | |||
| Any | 62 | 53 | 66 |
| Severe | 13 | 25 | 15 |
| Myalgia | |||
| Any | 19 | 16 | 21 |
| Severe | 2 | 2 | 2 |
| Arthralgia | 9 | 7 | 8 |
| Infusion Site Reactions | 4 | 3 | 4 |
Hematologic Reactions
Reversible marrow suppression was the major dose-limiting toxicity of Docetaxel Injection USP (Injection Concentrate) [see Warnings and Precautions (5.3)]. The median time to nadir was 7 days, while the median duration of severe neutropenia (<500 cells/mm3) was 7 days. Among 2045 patients with solid tumors and normal baseline LFTs, severe neutropenia occurred in 75.4% and lasted for more than 7 days in 2.9% of cycles.
Febrile neutropenia (<500 cells/mm3 with fever >38°C with intravenous antibiotics and/or hospitalization) occurred in 11% of patients with solid tumors, in 12.3% of patients with metastatic breast cancer, and in 9.8% of 92 breast cancer patients premedicated with 3-day corticosteroids.
Severe infectious episodes occurred in 6.1% of patients with solid tumors, in 6.4% of patients with metastatic breast cancer, and in 5.4% of 92 breast cancer patients premedicated with 3-day corticosteroids.
Thrombocytopenia (<100,000 cells/mm3) associated with fatal gastrointestinal hemorrhage has been reported.
Hypersensitivity Reactions
Severe hypersensitivity reactions have been reported [see Boxed Warning, Warnings and Precautions (5.4)]. Minor events, including flushing, rash with or without pruritus, chest tightness, back pain, dyspnea, drug fever, or chills, have been reported and resolved after discontinuing the infusion and instituting appropriate therapy.
Fluid Retention.
Fluid retention can occur with the use of Docetaxel Injection USP (Injection Concentrate) [see Boxed Warning, Dosage and Administration (2.6), Warnings and Precautions (5.5)].
Cutaneous Reactions
Severe skin toxicity is discussed elsewhere in the label [see Warnings and Precautions (5.7)]. Reversible cutaneous reactions characterized by a rash including localized eruptions, mainly on the feet and/or hands, but also on the arms, face, or thorax, usually associated with pruritus, have been observed. Eruptions generally occurred within 1 week after Docetaxel Injection USP (Injection Concentrate) infusion, recovered before the next infusion, and were not disabling.
Severe nail disorders were characterized by hypo- or hyperpigmentation, and occasionally by onycholysis (in 0.8% of patients with solid tumors) and pain.
Neurologic Reactions
Neurologic reactions are discussed elsewhere in the label [see Warnings and Precautions (5.8)]
Gastrointestinal Reactions
Nausea, vomiting, and diarrhea were generally mild to moderate. Severe reactions occurred in 3-5% of patients with solid tumors and to a similar extent among metastatic breast cancer patients. The incidence of severe reactions was 1% or less for the 92 breast cancer patients premedicated with 3-day corticosteroids.
Severe stomatitis occurred in 5.5% of patients with solid tumors, in 7.4% of patients with metastatic breast cancer, and in 1.1% of the 92 breast cancer patients premedicated with 3-day corticosteroids.
Cardiovascular Reactions
Hypotension occurred in 2.8% of patients with solid tumors; 1.2% required treatment. Clinically meaningful events such as heart failure, sinus tachycardia, atrial flutter, dysrhythmia, unstable angina, pulmonary edema, and hypertension occurred rarely. Seven of 86 (8.1%) of metastatic breast cancer patients receiving Docetaxel Injection USP (Injection Concentrate) 100 mg/m2 in a randomized trial and who had serial left ventricular ejection fractions assessed developed deterioration of LVEF by ≥10% associated with a drop below the institutional lower limit of normal.
Infusion Site Reactions
Infusion site reactions were generally mild and consisted of hyperpigmentation, inflammation, redness or dryness of the skin, phlebitis, extravasation, or swelling of the vein.
Hepatic Reactions
In patients with normal LFTs at baseline, bilirubin values greater than the ULN occurred in 8.9% of patients. Increases in AST or ALT >1.5 times the ULN, or alkaline phosphatase >2.5 times ULN, were observed in 18.9% and 7.3% of patients, respectively. While on Docetaxel Injection USP (Injection Concentrate), increases in AST and/or ALT >1.5 times ULN concomitant with alkaline phosphatase >2.5 times ULN occurred in 4.3% of patients with normal LFTs at baseline. Whether these changes were related to the drug or underlying disease has not been established.
Hematologic and Other Toxicity: Relation to dose and baseline liver chemistry abnormalities
Hematologic and other toxicity is increased at higher doses and in patients with elevated baseline liver function tests (LFTs). In the following tables, adverse drug reactions are compared for three populations: 730 patients with normal LFTs given Docetaxel Injection USP (Injection Concentrate) at 100 mg/m2 in the randomized and single arm studies of metastatic breast cancer after failure of previous chemotherapy; 18 patients in these studies who had abnormal baseline LFTs (defined as AST and/or ALT >1.5 times ULN concurrent with alkaline phosphatase >2.5 times ULN); and 174 patients in Japanese studies given Docetaxel Injection USP (Injection Concentrate) at 60 mg/m2 who had normal LFTs (see Tables 4 and 5).
| *Normal Baseline LFTs: Transaminases ≤1.5 times ULN or alkaline phosphatase ≤2.5 times ULN or isolated elevations of transaminases or alkaline phosphatase up to 5 times ULN **Elevated Baseline LFTs: AST and/or ALT >1.5 times ULN concurrent with alkaline phosphatase >2.5 times ULN ***Incidence of infection requiring hospitalization and/or intravenous antibiotics was 8.5% (n=62) among the 730 patients with normal LFTs at baseline; 7 patients had concurrent grade 3 neutropenia, and 46 patients had grade 4 neutropenia. ****Febrile Neutropenia: For 100 mg/m2, ANC grade 4 and fever >38°C with intravenous antibiotics and/or hospitalization; for 60 mg/m2, ANC grade 3/4 and fever >38.1°C |
|||
|
Docetaxel Injection USP
(Injection Concentrate)
|
Docetaxel Injection
USP (Injection Concentrate)
|
||
| Adverse Reaction | Normal LFTs*
n=730 % |
Elevated
LFTs**
| Normal LFTs*
n=174 % |
| Neutropenia | |||
| Any <2000 cells/mm3 | 98 | 100 | 95 |
| Grade 4 <500 cells/mm3 | 84 | 94 | 75 |
| Thrombocytopenia | |||
| Any <100,000 cells/mm3 | 11 | 44 | 14 |
| Grade 4 <20,000 cells/mm3 | 1 | 17 | 1 |
| Anemia <11 g/dL | 95 | 94 | 65 |
| Infection*** | |||
| Any | 23 | 39 | 1 |
| Grade 3 and 4 | 7 | 33 | 0 |
| Febrile Neutropenia**** | |||
| By Patient | 12 | 33 | 0 |
| By Course | 2 | 9 | 0 |
| Septic Death | 2 | 6 | 1 |
| Non-Septic Death | 1 | 11 | 0 |
| *Normal Baseline LFTs: Transaminases ≤1.5 times ULN or alkaline phosphatase ≤2.5 times ULN or isolated elevations of transaminases or alkaline phosphatase up to 5 times ULN **Elevated Baseline Liver Function: AST and/or ALT >1.5 times ULN concurrent with alkaline phosphatase >2.5 times ULN ***Retention includes (by COSTART): edema (peripheral, localized, generalized, lymphedema, pulmonary edema, and edema otherwise not specified) and effusion (pleural, pericardial, and ascites); no premedication given with the 60 mg/m2dose NA = not available |
|||
|
Docetaxel Injection USP
(Injection Concentrate)
| Docetaxel Injection USP (Injection Concentrate)
60 mg/m2 |
||
| Adverse Reaction | Normal LFTs*
n=730 % |
Elevated
LFTs**
| Normal LFTs*
n=174 % |
| Acute Hypersensitivity
Reaction Regardless of Premedication | |||
| Any | 13 | 6 | 1 |
| Severe | 1 | 0 | 0 |
| Fluid Retention***
Regardless of Premedication | |||
| Any | 56 | 61 | 13 |
| Severe | 8 | 17 | 0 |
| Neurosensory | |||
| Any | 57 | 50 | 20 |
| Severe | 6 | 0 | 0 |
| Myalgia | 23 | 33 | 3 |
| Cutaneous | |||
| Any | 45 | 61 | 31 |
| Severe | 5 | 17 | 0 |
| Asthenia | |||
| Any | 65 | 44 | 66 |
| Severe | 17 | 22 | 0 |
| Diarrhea | |||
| Any | 42 | 28 | NA |
| Severe | 6 | 11 | |
| Stomatitis | |||
| Any | 53 | 67 | 19 |
| Severe | 8 | 39 | 1 |
In the three-arm monotherapy trial, TAX313, which compared Docetaxel Injection USP (Injection Concentrate) 60 mg/m2, 75 mg/m2 and 100 mg/m2 in advanced breast cancer, grade 3/4 or severe adverse reactions occurred in 49.0% of patients treated with Docetaxel Injection USP (Injection Concentrate) 60 mg/m2 compared to 55.3% and 65.9% treated with 75 mg/m2 and 100 mg/m2 respectively. Discontinuation due to adverse reactions was reported in 5.3% of patients treated with 60 mg/m2 vs. 6.9% and 16.5% for patients treated at 75 mg/m2 and 100 mg/m2 respectively. Deaths within 30 days of last treatment occurred in 4.0% of patients treated with 60 mg/m2 compared to 5.3% and 1.6% for patients treated at 75 mg/m2 and 100 mg/m2 respectively.
The following adverse reactions were associated with increasing docetaxel doses: fluid retention (26%, 38%, and 46% at 60 mg/m2, 75 mg/m2, and 100 mg/m2 respectively), thrombocytopenia (7%, 11% and 12% respectively), neutropenia (92%, 94%, and 97% respectively), febrile neutropenia (5%, 7%, and 14% respectively), treatment-related grade 3/4 infection (2%, 3%, and 7% respectively) and anemia (87%, 94%, and 97% respectively).
Combination therapy with Docetaxel Injection USP (Injection Concentrate) in the adjuvant treatment of breast cancer
The following table presents treatment emergent adverse reactions observed in 744 patients, who were treated with Docetaxel Injection USP (Injection Concentrate) 75 mg/m2 every 3 weeks in combination with doxorubicin and cyclophosphamide (see Table 6).
| * COSTART term and grading system for events related to treatment. | ||||
| Docetaxel Injection USP (Injection Concentrate) 75 mg/m2+
Doxorubicin 50 mg/m2+ Cyclophosphamide 500 mg/m2 (TAC) n=744 % |
Fluorouracil
500 mg/m2+
|
|||
| Adverse Reaction | Any | Grade 3/4 | Any | Grade 3/4 |
| Anemia | 92 | 4 | 72 | 2 |
| Neutropenia | 71 | 66 | 82 | 49 |
| Fever in absence of infection | 47 | 1 | 17 | 0 |
| Infection | 39 | 4 | 36 | 2 |
| Thrombocytopenia | 39 | 2 | 28 | 1 |
| Febrile neutropenia | 25 | N/A | 3 | N/A |
| Neutropenic infection | 12 | N/A | 6 | N/A |
| Hypersensitivity reactions | 13 | 1 | 4 | 0 |
| Lymphedema | 4 | 0 | 1 | 0 |
| Fluid Retention* | 35 | 1 | 15 | 0 |
| Peripheral edema | 27 | 0 | 7 | 0 |
| Weight gain | 13 | 0 | 9 | 0 |
| Neuropathy sensory | 26 | 0 | 10 | 0 |
| Neuro-cortical | 5 | 1 | 6 | 1 |
| Neuropathy motor | 4 | 0 | 2 | 0 |
| Neuro-cerebellar | 2 | 0 | 2 | 0 |
| Syncope | 2 | 1 | 1 | 0 |
| Alopecia | 98 | N/A | 97 | N/A |
| Skin toxicity | 27 | 1 | 18 | 0 |
| Nail disorders | 19 | 0 | 14 | 0 |
| Nausea | 81 | 5 | 88 | 10 |
| Stomatitis | 69 | 7 | 53 | 2 |
| Vomiting | 45 | 4 | 59 | 7 |
| Diarrhea | 35 | 4 | 28 | 2 |
| Constipation | 34 | 1 | 32 | 1 |
| Taste perversion | 28 | 1 | 15 | 0 |
| Anorexia | 22 | 2 | 18 | 1 |
| Abdominal Pain | 11 | 1 | 5 | 0 |
| Amenorrhea | 62 | N/A | 52 | N/A |
| Cough | 14 | 0 | 10 | 0 |
| Cardiac dysrhythmias | 8 | 0 | 6 | 0 |
| Vasodilatation | 27 | 1 | 21 | 1 |
| Hypotension | 2 | 0 | 1 | 0 |
| Phlebitis | 1 | 0 | 1 | 0 |
| Asthenia | 81 | 11 | 71 | 6 |
| Myalgia | 27 | 1 | 10 | 0 |
| Arthralgia | 19 | 1 | 9 | 0 |
| Lacrimation disorder | 11 | 0 | 7 | 0 |
| Conjunctivitis | 5 | 0 | 7 | 0 |
Of the 744 patients treated with TAC, 36.3% experienced severe treatment emergent adverse reactions compared to 26.6% of the 736 patients treated with FAC. Dose reductions due to hematologic toxicity occurred in 1% of cycles in the TAC arm versus 0.1% of cycles in the FAC arm. Six percent of patients treated with TAC discontinued treatment due to adverse reactions, compared to 1.1% treated with FAC; fever in the absence of infection and allergy being the most common reasons for withdrawal among TAC-treated patients. Two patients died in each arm within 30 days of their last study treatment; 1 death per arm was attributed to study drugs.
Fever and Infection
Fever in the absence of infection was seen in 46.5% of TAC-treated patients and in 17.1% of FAC-treated patients. Grade 3/4 fever in the absence of infection was seen in 1.3% and 0% of TAC- and FAC-treated patients respectively. Infection was seen in 39.4% of TAC-treated patients compared to 36.3% of FAC-treated patients. Grade 3/4 infection was seen in 3.9% and 2.2% of TAC-treated and FAC-treated patients respectively. There were no septic deaths in either treatment arm.
Gastrointestinal Reactions
In addition to gastrointestinal reactions reflected in the table above, 7 patients in the TAC arm were reported to have colitis/enteritis/large intestine perforation vs. one patient in the FAC arm. Five of the 7 TAC-treated patients required treatment discontinuation; no deaths due to these events occurred.
Cardiovascular Reactions
More cardiovascular reactions were reported in the TAC arm vs. the FAC arm; dysrhythmias, all grades (7.9% vs. 6.0%), hypotension, all grades (2.6% vs. 1.1%) and CHF (2.3% vs. 0.9%, at 70 months median follow-up). One patient in each arm died due to heart failure.
Acute Myeloid Leukemia (AML)
Treatment-related acute myeloid leukemia or myelodysplasia is known to occur in patients treated with anthracyclines and/or cyclophosphamide, including use in adjuvant therapy for breast cancer. AML occurs at a higher frequency when these agents are given in combination with radiation therapy. AML occurred in the adjuvant breast cancer trial (TAX316). The cumulative risk of developing treatment-related AML at 5 years in TAX316 was 0.4% for TAC-treated patients and 0.1% for FAC-treated patients. This risk of AML is comparable to the risk observed for other anthracyclines/cyclophosphamide containing adjuvant breast chemotherapy regimens.
Lung Cancer
Monotherapy with Docetaxel Injection USP (Injection Concentrate) for unresectable, locally advanced or metastatic NSCLC previously treated with platinum-based chemotherapy
Docetaxel Injection USP (Injection Concentrate) 75 mg/m2: Treatment emergent adverse drug reactions are shown in Table 7. Included in this table are safety data for a total of 176 patients with non-small cell lung carcinoma and a history of prior treatment with platinum-based chemotherapy who were treated in two randomized, controlled trials. These reactions were described using NCI Common Toxicity Criteria regardless of relationship to study treatment, except for the hematologic toxicities or where otherwise noted.
| * Normal Baseline LFTs: Transaminases ≤1.5 times ULN or alkaline phosphatase ≤2.5 times ULN or isolated elevations of transaminases or alkaline phosphatase up to 5 times ULN ** Febrile Neutropenia: ANC grade 4 with fever >38°C with intravenous antibiotics and/or hospitalization *** COSTART term and grading system ‡ Not Applicable § Not Done |
|||
| Adverse Reaction | Docetaxel Injection USP (Injection Concentrate)
75 mg/m2 n=176 % | Best Supportive Care
n=49 % | Vinorelbine/Ifosfamide
n=119 % |
| Neutropenia | |||
| Any | 84 | 14 | 83 |
| Grade 3/4 | 65 | 12 | 57 |
| Leukopenia | |||
| Any | 84 | 6 | 89 |
| Grade 3/4 | 49 | 0 | 43 |
| Thrombocytopenia | |||
| Any | 8 | 0 | 8 |
| Grade 3/4 | 3 | 0 | 2 |
| Anemia | |||
| Any | 91 | 55 | 91 |
| Grade 3/4 | 9 | 12 | 14 |
| Febrile Neutropenia** | 6 | NA‡ | 1 |
| Infection | |||
| Any | 34 | 29 | 30 |
| Grade 3/4 | 10 | 6 | 9 |
| Treatment Related Mortality | 3 | NA‡ | 3 |
| Hypersensitivity Reactions | |||
| Any | 6 | 0 | 1 |
| Grade 3/4 | 3 | 0 | 0 |
| Fluid Retention | |||
| Any | 34 | ND§ | 23 |
| Severe | 3 | 3 | |
| Neurosensory | |||
| Any | 23 | 14 | 29 |
| Grade 3/4 | 2 | 6 | 5 |
| Neuromotor | |||
| Any | 16 | 8 | 10 |
| Grade 3/4 | 5 | 6 | 3 |
| Skin | |||
| Any | 20 | 6 | 17 |
| Grade 3/4 | 1 | 2 | 1 |
| Gastrointestinal | |||
| Nausea | |||
| Any | 34 | 31 | 31 |
| Grade 3/4 | 5 | 4 | 8 |
| Vomiting | |||
| Any | 22 | 27 | 22 |
| Grade 3/4 | 3 | 2 | 6 |
| Diarrhea | |||
| Any | 23 | 6 | 12 |
| Grade 3/4 | 3 | 0 | 4 |
| Alopecia | 56 | 35 | 50 |
| Asthenia | |||
| Any | 53 | 57 | 54 |
| Severe*** | 18 | 39 | 23 |
| Stomatitis | |||
| Any | 26 | 6 | 8 |
| Grade 3/4 | 2 | 0 | 1 |
| Pulmonary | |||
| Any | 41 | 49 | 45 |
| Grade 3/4 | 21 | 29 | 19 |
| Nail Disorder | |||
| Any | 11 | 0 | 2 |
| Severe*** | 1 | 0 | 0 |
| Myalgia | |||
| Any | 6 | 0 | 3 |
| Severe*** | 0 | 0 | 0 |
| Arthralgia | |||
| Any | 3 | 2 | 2 |
| Severe*** | 0 | 0 | 1 |
| Taste Perversion | |||
| Any | 6 | 0 | 0 |
| Severe*** | 1 | 0 | 0 |
Combination therapy with Docetaxel Injection USP (Injection Concentrate) in chemotherapy-naïve advanced unresectable or metastatic NSCLC
Table 8 presents safety data from two arms of an open label, randomized controlled trial (TAX326) that enrolled patients with unresectable stage IIIB or IV non-small cell lung cancer and no history of prior chemotherapy. Adverse reactions were described using the NCI Common Toxicity Criteria except where otherwise noted.
| * Replaces NCI term "Allergy" ** COSTART term and grading system |
||
| Adverse Reaction |
Docetaxel Injection USP (Injection Concentrate) 75mg/m2
+ Cisplatin
|
Vinorelbine 25 mg/m2 +
Cisplatin 100 mg/m2
|
| Neutropenia | ||
| Any | 91 | 90 |
| Grade 3/4 | 74 | 78 |
| Febrile Neutropenia | 5 | 5 |
| Thrombocytopenia | ||
| Any | 15 | 15 |
| Grade 3/4 | 3 | 4 |
| Anemia | ||
| Any | 89 | 94 |
| Grade 3/4 | 7 | 25 |
| Infection | ||
| Any | 35 | 37 |
| Grade 3/4 | 8 | 8 |
| Fever in absence of infection | ||
| Any | 33 | 29 |
| Grade 3/4 | < 1 | 1 |
| Hypersensitivity Reaction* | ||
| Any | 12 | 4 |
| Grade 3/4 | 3 | < 1 |
| Fluid Retention** | ||
| Any | 54 | 42 |
| All severe or life-threatening events | 2 | 2 |
| Pleural effusion | ||
| Any | 23 | 22 |
| All severe or life-threatening events | 2 | 2 |
| Peripheral edema | ||
| Any | 34 | 18 |
| All severe or life-threatening events | <1 | <1 |
| Weight Gain | ||
| Any | 15 | 9 |
| All severe or life-threatening events | <1 | <1 |
| Neurosensory | ||
| Any | 47 | 42 |
| Grade 3/4 | 4 | 4 |
| Neuromotor | ||
| Any | 19 | 17 |
| Grade 3/4 | 3 | 6 |
| Skin | ||
| Any | 16 | 14 |
| Grade 3/4 | <1 | 1 |
| Nausea | ||
| Any | 72 | 76 |
| Grade 3/4 | 10 | 17 |
| Vomiting | ||
| Any | 55 | 61 |
| Grade 3/4 | 8 | 16 |
| Diarrhea | ||
| Any | 47 | 25 |
| Grade 3/4 | 7 | 3 |
| Anorexia** | ||
| Any | 42 | 40 |
| All severe or life-threatening events | 5 | 5 |
| Stomatitis | ||
| Any | 24 | 21 |
| Grade 3/4 | 2 | 1 |
| Alopecia | ||
| Any | 75 | 42 |
| Grade 3 | <1 | 0 |
| Asthenia** | ||
| Any | 74 | 75 |
| All severe or life-threatening events | 12 | 14 |
| Nail Disorder** | ||
| Any | 14 | <1 |
| All severe events | <1 | 0 |
| Myalgia** | ||
| Any | 18 | 12 |
| All severe events | <1 | <1 |
Deaths within 30 days of last study treatment occurred in 31 patients (7.6%) in the docetaxel+cisplatin arm and 37 patients (9.3%) in the vinorelbine+cisplatin arm. Deaths within 30 days of last study treatment attributed to study drug occurred in 9 patients (2.2%) in the docetaxel+cisplatin arm and 8 patients (2.0%) in the vinorelbine+cisplatin arm.
The second comparison in the study, vinorelbine+cisplatin versus Docetaxel Injection USP (Injection Concentrate)+carboplatin (which did not demonstrate a superior survival associated with Docetaxel Injection USP (Injection Concentrate), [see Clinical Studies (14.3)]) demonstrated a higher incidence of thrombocytopenia, diarrhea, fluid retention, hypersensitivity reactions, skin toxicity, alopecia and nail changes on the Docetaxel Injection USP (Injection Concentrate)+carboplatin arm, while a higher incidence of anemia, neurosensory toxicity, nausea, vomiting, anorexia and asthenia was observed on the vinorelbine+cisplatin arm.
Prostate Cancer
Combination therapy with Docetaxel Injection USP (Injection Concentrate) in patients with prostate cancer
The following data are based on the experience of 332 patients, who were treated with Docetaxel Injection USP (Injection Concentrate) 75 mg/m2 every 3 weeks in combination with prednisone 5 mg orally twice daily (see Table 9).
| * Related to treatment | ||||
| Docetaxel Injection USP (Injection Concentrate) 75
mg/m2 every 3 weeks + prednisone 5 mg twice daily n=332 % | Mitoxantrone 12 mg/m2 every
3 weeks + prednisone 5 mg twice daily n=335 % |
|||
| Adverse Reaction | Any | Grade 3/4 | Any | Grade 3/4 |
| Anemia | 67 | 5 | 58 | 2 |
| Neutropenia | 41 | 32 | 48 | 22 |
| Thrombocytopenia | 3 | 1 | 8 | 1 |
| Febrile neutropenia | 3 | N/A | 2 | N/A |
| Infection | 32 | 6 | 20 | 4 |
| Epistaxis | 6 | 0 | 2 | 0 |
| Allergic Reactions | 8 | 1 | 1 | 0 |
| Fluid Retention* | 24 | 1 | 5 | 0 |
| Weight Gain* | 8 | 0 | 3 | 0 |
| Peripheral Edema* | 18 | 0 | 2 | 0 |
| Neuropathy Sensory | 30 | 2 | 7 | 0 |
| Neuropathy Motor | 7 | 2 | 3 | 1 |
| Rash/Desquamation | 6 | 0 | 3 | 1 |
| Alopecia | 65 | N/A | 13 | N/A |
| Nail Changes | 30 | 0 | 8 | 0 |
| Nausea | 41 | 3 | 36 | 2 |
| Diarrhea | 32 | 2 | 10 | 1 |
| Stomatitis/Pharyngitis | 20 | 1 | 8 | 0 |
| Taste Disturbance | 18 | 0 | 7 | 0 |
| Vomiting | 17 | 2 | 14 | 2 |
| Anorexia | 17 | 1 | 14 | 0 |
| Cough | 12 | 0 | 8 | 0 |
| Dyspnea | 15 | 3 | 9 | 1 |
| Cardiac left ventricular function | 10 | 0 | 22 | 1 |
| Fatigue | 53 | 5 | 35 | 5 |
| Myalgia | 15 | 0 | 13 | 1 |
| Tearing | 10 | 1 | 2 | 0 |
| Arthralgia | 8 | 1 | 5 | 1 |
Gastric Cancer
Combination therapy with Docetaxel Injection USP (Injection Concentrate) in gastric adenocarcinoma
Data in the following table are based on the experience of 221 patients with advanced gastric adenocarcinoma and no history of prior chemotherapy for advanced disease, who were treated with Docetaxel Injection USP (Injection Concentrate) 75 mg/m2 in combination with cisplatin and fluorouracil (see Table 10).
| Clinically important treatment emergent adverse reactions were determined based upon frequency, severity, and clinical impact of the adverse reaction. * Related to treatment |
||||
|
Docetaxel Injection USP (Injection Concentrate)
75 mg/m2+
| Cisplatin 100 mg/m2 +
fluorouracil 1000 mg/m2 n=224 |
|||
| Adverse Reaction | Any
% | Grade 3/4
% | Any
% | Grade 3/4
% |
| Anemia | 97 | 18 | 93 | 26 |
| Neutropenia | 96 | 82 | 83 | 57 |
| Fever in the absence of infection | 36 | 2 | 23 | 1 |
| Thrombocytopenia | 26 | 8 | 39 | 14 |
| Infection | 29 | 16 | 23 | 10 |
| Febrile neutropenia | 16 | N/A | 5 | N/A |
| Neutropenic infection | 16 | N/A | 10 | N/A |
| Allergic reactions | 10 | 2 | 6 | 0 |
| Fluid retention* | 15 | 0 | 4 | 0 |
| Edema* | 13 | 0 | 3 | 0 |
| Lethargy | 63 | 21 | 58 | 18 |
| Neurosensory | 38 | 8 | 25 | 3 |
| Neuromotor | 9 | 3 | 8 | 3 |
| Dizziness | 16 | 5 | 8 | 2 |
| Alopecia | 67 | 5 | 41 | 1 |
| Rash/itch | 12 | 1 | 9 | 0 |
| Nail changes | 8 | 0 | 0 | 0 |
| Skin desquamation | 2 | 0 | 0 | 0 |
| Nausea | 73 | 16 | 76 | 19 |
| Vomiting | 67 | 15 | 73 | 19 |
| Anorexia | 51 | 13 | 54 | 12 |
| Stomatitis | 59 | 21 | 61 | 27 |
| Diarrhea | 78 | 20 | 50 | 8 |
| Constipation | 25 | 2 | 34 | 3 |
| Esophagitis/dysphagia/odynophagia | 16 | 2 | 14 | 5 |
| Gastrointestinal pain/cramping | 11 | 2 | 7 | 3 |
| Cardiac dysrhythmias | 5 | 2 | 2 | 1 |
| Myocardial ischemia | 1 | 0 | 3 | 2 |
| Tearing | 8 | 0 | 2 | 0 |
| Altered hearing | 6 | 0 | 13 | 2 |
Head and Neck Cancer
Combination therapy with Docetaxel Injection USP (Injection Concentrate) in head and neck cancer
Table 11 summarizes the safety data obtained from patients that received induction chemotherapy with Docetaxel Injection USP (Injection Concentrate) 75 mg/m2 in combination with cisplatin and fluorouracil followed by radiotherapy (TAX323; 174 patients) or chemoradiotherapy (TAX324; 251 patients). The treatment regimens are described in Section 14.6.
| Clinically important treatment emergent adverse reactions based upon frequency, severity, and clinical impact. | ||||||||
| * Febrile neutropenia: grade ≥2 fever concomitant with grade 4 neutropenia requiring intravenous antibiotics and/or hospitalization. | ||||||||
| ** Related to treatment. | ||||||||
| *** Includes superficial and deep vein thrombosis and pulmonary embolism | ||||||||
| TAX323 (n=355) | TAX324 (n=494) |
|||||||
| Docetaxel Injection USP (Injection Concentrate) arm (n=174) |
Comparator arm (n=181) | Docetaxel Injection USP (Injection Concentrate) arm (n=251) |
Comparator arm (n=243) |
|||||
| Adverse Reaction (by Body System) | Any % | Grade 3/4 % | Any % | Grade 3/4 % | Any % | Grade 3/4 % | Any % | Grade 3/4 % |
| Neutropenia | 93 | 76 | 87 | 53 | 95 | 84 | 84 | 56 |
| Anemia | 89 | 9 | 88 | 14 | 90 | 12 | 86 | 10 |
| Thrombocytopenia | 24 | 5 | 47 | 18 | 28 | 4 | 31 | 11 |
| Infection | 27 | 9 | 26 | 8 | 23 | 6 | 28 | 5 |
| Febrile neutropenia* | 5 | N/A | 2 | N/A | 12 | N/A | 7 | N/A |
| Neutropenic infection | 14 | N/A | 8 | N/A | 12 | N/A | 8 | N/A |
| Cancer pain | 21 | 5 | 16 | 3 | 17 | 9 | 20 | 11 |
| Lethargy | 41 | 3 | 38 | 3 | 61 | 5 | 56 | 10 |
| Fever in the absence of infection | 32 | 1 | 37 | 0 | 30 | 4 | 28 | 3 |
| Myalgia | 10 | 1 | 7 | 0 | 7 | 0 | 7 | 2 |
| Weight loss | 21 | 1 | 27 | 1 | 14 | 2 | 14 | 2 |
| Allergy | 6 | 0 | 3 | 0 | 2 | 0 | 0 | 0 |
| Fluid retention** | 20 | 0 | 14 | 1 | 13 | 1 | 7 | 2 |
| Edema only | 13 | 0 | 7 | 0 | 12 | 1 | 6 | 1 |
| Weight gain only | 6 | 0 | 6 | 0 | 0 | 0 | 1 | 0 |
| Dizziness | 2 | 0 | 5 | 1 | 16 | 4 | 15 | 2 |
| Neurosensory | 18 | 1 | 11 | 1 | 14 | 1 | 14 | 0 |
| Altered hearing | 6 | 0 | 10 | 3 | 13 | 1 | 19 | 3 |
| Neuromotor | 2 | 1 | 4 | 1 | 9 | 0 | 10 | 2 |
| Alopecia | 81 | 11 | 43 | 0 | 68 | 4 | 44 | 1 |
| Rash/itch | 12 | 0 | 6 | 0 | 20 | 0 | 16 | 1 |
| Dry skin | 6 | 0 | 2 | 0 | 5 | 0 | 3 | 0 |
| Desquamation | 4 | 1 | 6 | 0 | 2 | 0 | 5 | 0 |
| Nausea | 47 | 1 | 51 | 7 | 77 | 14 | 80 | 14 |
| Stomatitis | 43 | 4 | 47 | 11 | 66 | 21 | 68 | 27 |
| Vomiting | 26 | 1 | 39 | 5 | 56 | 8 | 63 | 10 |
| Diarrhea | 33 | 3 | 24 | 4 | 48 | 7 | 40 | 3 |
| Constipation | 17 | 1 | 16 | 1 | 27 | 1 | 38 | 1 |
| Anorexia | 16 | 1 | 25 | 3 | 40 | 12 | 34 | 12 |
| Esophagitis/dysphagia/ Odynophagia | 13 | 1 | 18 | 3 | 25 | 13 | 26 | 10 |
| Taste, sense of smell altered | 10 | 0 | 5 | 0 | 20 | 0 | 17 | 1 |
| Gastrointestinal pain/cramping | 8 | 1 | 9 | 1 | 15 | 5 | 10 | 2 |
| Heartburn | 6 | 0 | 6 | 0 | 13 | 2 | 13 | 1 |
| Gastrointestinal bleeding | 4 | 2 | 0 | 0 | 5 | 1 | 2 | 1 |
| Cardiac dysrhythmia | 2 | 2 | 2 | 1 | 6 | 3 | 5 | 3 |
| Venous*** | 3 | 2 | 6 | 2 | 4 | 2 | 5 | 4 |
| Ischemia myocardial | 2 | 2 | 1 | 0 | 2 | 1 | 1 | 1 |
| Tearing | 2 | 0 | 1 | 0 | 2 | 0 | 2 | 0 |
| Conjunctivitis | 1 | 0 | 1 | 0 | 1 | 0 | 0.4 | 0 |
6.2 Post-Marketing Experiences
The following adverse reactions have been identified from clinical trials and/or post-marketing surveillance. Because they are reported from a population of unknown size, precise estimates of frequency cannot be made.
Body as a whole: diffuse pain, chest pain, radiation recall phenomenon.
Cardiovascular: atrial fibrillation, deep vein thrombosis, ECG abnormalities, thrombophlebitis, pulmonary embolism, syncope, tachycardia, myocardial infarction.
Cutaneous: very rare cases of cutaneous lupus erythematosus and rare cases of bullous eruptions such as erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis, and Scleroderma-like changes usually preceded by peripheral lymphedema. In some cases multiple factors may have contributed to the development of these effects. Severe hand and foot syndrome has been reported. Cases of permanent alopecia have been reported
Gastrointestinal: abdominal pain, anorexia, constipation, duodenal ulcer, esophagitis, gastrointestinal hemorrhage, gastrointestinal perforation, ischemic colitis, colitis, intestinal obstruction, ileus, neutropenic enterocolitis and dehydration as a consequence to gastrointestinal events have been reported.
Hematologic: bleeding episodes. Disseminated intravascular coagulation (DIC), often in association with sepsis or multiorgan failure, has been reported. Cases of acute myeloid leukemia and myelodysplasic syndrome have been reported in association with Docetaxel Injection USP (Injection Concentrate) when used in combination with other chemotherapy agents and/or radiotherapy.
Hypersensitivity: rare cases of anaphylactic shock have been reported. Very rarely these cases resulted in a fatal outcome in patients who received premedication.
Hepatic: rare cases of hepatitis, sometimes fatal primarily in patients with pre-existing liver disorders, have been reported.
Neurologic: confusion, rare cases of seizures or transient loss of consciousness have been observed, sometimes appearing during the infusion of the drug.
Ophthalmologic: conjunctivitis, lacrimation or lacrimation with or without conjunctivitis. Excessive tearing which may be attributable to lacrimal duct obstruction has been reported. Rare cases of transient visual disturbances (flashes, flashing lights, scotomata) typically occurring during drug infusion and in association with hypersensitivity reactions have been reported. These were reversible upon discontinuation of the infusion. Cases of cystoid macular edema (CME) have been reported in patients treated with Docetaxel Injection USP (Injection Concentrate).
Hearing: rare cases of ototoxicity, hearing disorders and/or hearing loss have been reported, including cases associated with other ototoxic drugs.
Respiratory: dyspnea, acute pulmonary edema, acute respiratory distress syndrome/pneumonitis, interstitial lung disease, interstitial pneumonia, respiratory failure, and pulmonary fibrosis have rarely been reported and may be associated with fatal outcome. Rare cases of radiation pneumonitis have been reported in patients receiving concomitant radiotherapy.
Renal: renal insufficiency and renal failure have been reported, the majority of these cases were associated with concomitant nephrotoxic drugs.
Metabolism and nutrition disorders: cases of hyponatremia have been reported.
7. DRUG INTERACTIONS
Docetaxel is a CYP3A4 substrate. In vitro studies have shown that the metabolism of docetaxel may be modified by the concomitant administration of compounds that induce, inhibit, or are metabolized by cytochrome P450 3A4.
In vivo studies showed that the exposure of docetaxel increased 2.2-fold when it was coadministered with ketoconazole, a potent inhibitor of CYP3A4. Protease inhibitors, particularly ritonavir, may increase the exposure of docetaxel. Concomitant use of Docetaxel Injection USP (Injection Concentrate) and drugs that inhibit CYP3A4 may increase exposure to docetaxel and should be avoided. In patients receiving treatment with Docetaxel Injection USP (Injection Concentrate), close monitoring for toxicity and a Docetaxel Injection USP (Injection Concentrate) dose reduction could be considered if systemic administration of a potent CYP3A4 inhibitor cannot be avoided [see Dosage and Administration (2.7) and Clinical Pharmacology (12.3)].
8. USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category D [see 'Warnings and Precautions' section]
Based on its mechanism of action and findings in animals, Docetaxel Injection USP (Injection Concentrate) can cause fetal harm when administered to a pregnant woman. If Docetaxel Injection USP (Injection Concentrate) is used during pregnancy, or if the patient becomes pregnant while receiving this drug, the patient should be apprised of the potential hazard to the fetus. Women of childbearing potential should be advised to avoid becoming pregnant during therapy with Docetaxel Injection USP (Injection Concentrate).
Docetaxel Injection USP (Injection Concentrate) can cause fetal harm when administered to a pregnant woman. Studies in both rats and rabbits at doses ≥0.3 and 0.03 mg/kg/day, respectively (about 1/50 and 1/300 the daily maximum recommended human dose on a mg/m2 basis), administered during the period of organogenesis, have shown that Docetaxel Injection USP (Injection Concentrate) is embryotoxic and fetotoxic (characterized by intrauterine mortality, increased resorption, reduced fetal weight, and fetal ossification delay). The doses indicated above also caused maternal toxicity.
8.3 Nursing Mothers
It is not known whether docetaxel is excreted in human milk. Because many drugs are excreted in human milk, and because of the potential for serious adverse reactions in nursing infants from Docetaxel Injection USP (Injection Concentrate), a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
The alcohol content in Docetaxel Injection USP (Injection Concentrate) Injection should be taken into account when given to pediatric patients [see Warnings and Precautions (5.11)].
The efficacy of Docetaxel Injection USP (Injection Concentrate) in pediatric patients as monotherapy or in combination has not been established. The overall safety profile of Docetaxel Injection USP (Injection Concentrate) in pediatric patients receiving monotherapy or TCF was consistent with the known safety profile in adults.
Docetaxel Injection USP (Injection Concentrate) has been studied in a total of 289 pediatric patients: 239 in 2 trials with monotherapy and 50 in combination treatment with cisplatin and 5-fluoruracil (TCF).
Docetaxel Injection USP (Injection Concentrate) Monotherapy
Docetaxel Injection USP (Injection Concentrate) monotherapy was evaluated in a dose-finding phase 1 trial in 61 pediatric patients (median age 12.5 years, range 1-22 years) with a variety of refractory solid tumors. The recommended dose was 125 mg/m2 as a 1-hour intravenous infusion every 21 days. The primary dose limiting toxicity was neutropenia.
The recommended dose for Docetaxel Injection USP (Injection Concentrate) monotherapy was evaluated in a phase 2 single-arm trial in 178 pediatric patients (median age 12 years, range 1-26 years) with a variety of recurrent/refractory solid tumors. Efficacy was not established with tumor response rates ranging from one complete response (CR) (0.6%) in a patient with undifferentiated sarcoma to four partial responses (2.2%) seen in one patient each with Ewing Sarcoma, neuroblastoma, osteosarcoma, and squamous cell carcinoma.
Docetaxel Injection USP (Injection Concentrate) in Combination
Docetaxel Injection USP (Injection Concentrate) was studied in combination with cisplatin and 5-fluorouracil (TCF) versus cisplatin and 5-fluorouracil (CF) for the induction treatment of nasopharyngeal carcinoma (NPC) in pediatric patients prior to chemoradiation consolidation. Seventy-five patients (median age 16 years, range 9 to 21 years) were randomized (2:1) to Docetaxel Injection USP (Injection Concentrate) (75 mg/m2) in combination with cisplatin (75 mg/m2) and 5-fluorouracil (750 mg/m2) (TCF) or to cisplatin (80 mg/m2) and 5-fluorouracil (1000 mg/m2/day) (CF). The primary endpoint was the CR rate following induction treatment of NPC. One patient out of 50 in the TCF group (2%) had a complete response while none of the 25 patients in the CF group had a complete response.
Pharmacokinetics:
Pharmacokinetic parameters for docetaxel were determined in 2 pediatric solid tumor trials. Following docetaxel administration at 55 mg/m2 to 235 mg/m2 in a 1-hour intravenous infusion every 3 weeks in 25 patients aged 1 to 20 years (median 11 years), docetaxel clearance was 17.3±10.9 L/h/m2.
Docetaxel was administered in combination with cisplatin and 5-fluorouracil (TCF), at dose levels of 75 mg/m2 in a 1-hour intravenous infusion day 1 in 28 patients aged 10 to 21 years (median 16 years, 17 patients were older than 16). Docetaxel clearance was 17.9±8.75 L/h/m2, corresponding to an AUC of 4.20±2.57 μg.h/mL.
In summary, the body surface area adjusted clearance of docetaxel monotherapy and TCF combination in children were comparable to those in adults [see Clinical Pharmacology (12.3)].
8.5 Geriatric Use
In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy in elderly patients.
Non-Small Cell Lung Cancer
In a study conducted in chemotherapy-naïve patients with NSCLC (TAX326), 148 patients (36%) in the Docetaxel Injection USP (Injection Concentrate)+cisplatin group were 65 years of age or greater. There were 128 patients (32%) in the vinorelbine+cisplatin group 65 years of age or greater. In the Docetaxel Injection USP (Injection Concentrate)+cisplatin group, patients less than 65 years of age had a median survival of 10.3 months (95% CI: 9.1 months, 11.8 months) and patients 65 years or older had a median survival of 12.1 months (95% CI: 9.3 months, 14 months). In patients 65 years of age or greater treated with Docetaxel Injection USP (Injection Concentrate)+cisplatin, diarrhea (55%), peripheral edema (39%) and stomatitis (28%) were observed more frequently than in the vinorelbine+cisplatin group (diarrhea 24%, peripheral edema 20%, stomatitis 20%). Patients treated with Docetaxel Injection USP (Injection Concentrate)+cisplatin who were 65 years of age or greater were more likely to experience diarrhea (55%), infections (42%), peripheral edema (39%) and stomatitis (28%) compared to patients less than the age of 65 administered the same treatment (43%, 31%, 31% and 21%, respectively).
When Docetaxel Injection USP (Injection Concentrate) was combined with carboplatin for the treatment of chemotherapy-naïve, advanced non-small cell lung carcinoma, patients 65 years of age or greater (28%) experienced higher frequency of infection compared to similar patients treated with Docetaxel Injection USP (Injection Concentrate)+cisplatin, and a higher frequency of diarrhea, infection and peripheral edema than elderly patients treated with vinorelbine+cisplatin.
Prostate Cancer
Of the 333 patients treated with Docetaxel Injection USP (Injection Concentrate) every three weeks plus prednisone in the prostate cancer study (TAX327), 209 patients were 65 years of age or greater and 68 patients were older than 75 years. In patients treated with Docetaxel Injection USP (Injection Concentrate) every three weeks, the following treatment emergent adverse reactions occurred at rates ≥10% higher in patients 65 years of age or greater compared to younger patients: anemia (71% vs. 59%), infection (37% vs. 24%), nail changes (34% vs. 23%), anorexia (21% vs. 10%), weight loss (15% vs. 5%) respectively.
Breast Cancer
In the adjuvant breast cancer trial (TAX316), Docetaxel Injection USP (Injection Concentrate) in combination with doxorubicin and cyclophosphamide was administered to 744 patients of whom 48 (6%) were 65 years of age or greater. The number of elderly patients who received this regimen was not sufficient to determine whether there were differences in safety and efficacy between elderly and younger patients.
Gastric Cancer
Among the 221 patients treated with Docetaxel Injection USP (Injection Concentrate) in combination with cisplatin and fluorouracil in the gastric cancer study, 54 were 65 years of age or older and 2 patients were older than 75 years. In this study, the number of patients who were 65 years of age or older was insufficient to determine whether they respond differently from younger patients. However, the incidence of serious adverse reactions was higher in the elderly patients compared to younger patients. The incidence of the following adverse reactions (all grades, regardless of relationship): lethargy, stomatitis, diarrhea, dizziness, edema, febrile neutropenia/neutropenic infection occurred at rates ≥10% higher in patients who were 65 years of age or older compared to younger patients. Elderly patients treated with TCF should be closely monitored.
Head and Neck Cancer
Among the 174 and 251 patients who received the induction treatment with Docetaxel Injection USP (Injection Concentrate) in combination with cisplatin and fluorouracil (TPF) for SCCHN in the TAX323 and TAX324 studies, 18 (10%) and 32 (13%) of the patients were 65 years of age or older, respectively.
These clinical studies of Docetaxel Injection USP (Injection Concentrate) in combination with cisplatin and fluorouracil in patients with SCCHN did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients. Other reported clinical experience with this treatment regimen has not identified differences in responses between elderly and younger patients.
8.6 Hepatic Impairment
Patients with bilirubin >ULN should not receive Docetaxel Injection USP (Injection Concentrate). Also, patients with AST and/or ALT >1.5 x ULN concomitant with alkaline phosphatase >2.5 x ULN should not receive Docetaxel Injection USP (Injection Concentrate) [see Boxed Warning, Warnings and Precautions (5.2), Clinical Pharmacology (12.3)].
The alcohol content in Docetaxel Injection USP (Injection Concentrate) Injection should be taken into account when given to patients with hepatic impairment [see Warnings and Precautions (5.11)].
10. OVERDOSAGE
There is no known antidote for Docetaxel Injection USP (Injection Concentrate) overdosage. In case of overdosage, the patient should be kept in a specialized unit where vital functions can be closely monitored. Anticipated complications of overdosage include: bone marrow suppression, peripheral neurotoxicity, and mucositis. Patients should receive therapeutic G-CSF as soon as possible after discovery of overdose. Other appropriate symptomatic measures should be taken, as needed.
In two reports of overdose, one patient received 150 mg/m2 and the other received 200 mg/m2 as 1-hour infusions. Both patients experienced severe neutropenia, mild asthenia, cutaneous reactions, and mild paresthesia, and recovered without incident.
In mice, lethality was observed following single intravenous doses that were ≥154 mg/kg (about 4.5 times the human dose of 100 mg/m2 on a mg/m2 basis); neurotoxicity associated with paralysis, non-extension of hind limbs, and myelin degeneration was observed in mice at 48 mg/kg (about 1.5 times the human dose of 100 mg/m2 basis). In male and female rats, lethality was observed at a dose of 20 mg/kg (comparable to the human dose of 100 mg/m2 on a mg/m2 basis) and was associated with abnormal mitosis and necrosis of multiple organs.
11. DESCRIPTION
Docetaxel is an antineoplastic agent belonging to the taxoid family. It is prepared by semisynthesis beginning with a precursor derived from a plant cell fermentation process. The chemical name for docetaxel is (2R,3S)-N-carboxy-3-phenylisoserine,N-tert-butyl ester, 13-ester with 5β-20epoxy-1,2α,4,7β,10β,13α-hexahydroxytax-11-en-9-one 4-acetate 2-benzoate. Docetaxel has the following structural formula:
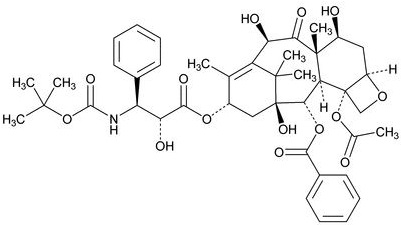
structure
Docetaxel is a white to off-white powder with an empirical formula of C43H53NO14, and a molecular weight of 807.88. It is highly lipophilic and practically insoluble in water.
One-vial Docetaxel Injection USP (Injection Concentrate)
Docetaxel Injection USP (Injection Concentrate) is a sterile, non-pyrogenic, pale yellow to brownish-yellow solution at 20 mg/mL concentration.
Each mL contains 20 mg docetaxel (anhydrous) in 50/50 (v/v) ratio polysorbate 80/dehydrated alcohol, and anhydrous citric acid.
Docetaxel Injection USP (Injection Concentrate) is available in single use vials containing 20 mg (1 mL) or 80 mg (4 mL) or 200 mg (10 mL) docetaxel (anhydrous).
Docetaxel Injection USP (Injection Concentrate) requires NO prior dilution with a diluent and is ready to add to the infusion solution.
12. CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Docetaxel is an antineoplastic agent that acts by disrupting the microtubular network in cells that is essential for mitotic and interphase cellular functions. Docetaxel binds to free tubulin and promotes the assembly of tubulin into stable microtubules while simultaneously inhibiting their disassembly. This leads to the production of microtubule bundles without normal function and to the stabilization of microtubules, which results in the inhibition of mitosis in cells. Docetaxel's binding to microtubules does not alter the number of protofilaments in the bound microtubules, a feature which differs from most spindle poisons currently in clinical use.
12.3 Human Pharmacokinetics
Absorption: The pharmacokinetics of docetaxel have been evaluated in cancer patients after administration of 20 mg/m2 to 115 mg/m2 in phase 1 studies. The area under the curve (AUC) was dose proportional following doses of 70 mg/m2 to 115 mg/m2 with infusion times of 1 to 2 hours. Docetaxel's pharmacokinetic profile is consistent with a three-compartment pharmacokinetic model, with half-lives for the α, β, and γ phases of 4 min, 36 min, and 11.1 hr, respectively. Mean total body clearance was 21 L/h/m2.
Distribution: The initial rapid decline represents distribution to the peripheral compartments and the late (terminal) phase is due, in part, to a relatively slow efflux of docetaxel from the peripheral compartment. Mean steady state volume of distribution was 113 L. In vitro studies showed that docetaxel is about 94% protein bound, mainly to α1-acid glycoprotein, albumin, and lipoproteins. In three cancer patients, the in vitro binding to plasma proteins was found to be approximately 97%. Dexamethasone does not affect the protein binding of docetaxel.
Metabolism: In vitro drug interaction studies revealed that docetaxel is metabolized by the CYP3A4 isoenzyme, and its metabolism may be modified by the concomitant administration of compounds that induce, inhibit, or are metabolized by cytochrome P450 3A4 [see Drug Interactions (7)].
Elimination: A study of 14C-docetaxel was conducted in three cancer patients. Docetaxel was eliminated in both the urine and feces following oxidative metabolism of the tert-butyl ester group, but fecal excretion was the main elimination route. Within 7 days, urinary and fecal excretion accounted for approximately 6% and 75% of the administered radioactivity, respectively. About 80% of the radioactivity recovered in feces is excreted during the first 48 hours as 1 major and 3 minor metabolites with very small amounts (less than 8%) of unchanged drug.
Effect of Age: A population pharmacokinetic analysis was carried out after Docetaxel Injection USP (Injection Concentrate) treatment of 535 patients dosed at 100 mg/m2. Pharmacokinetic parameters estimated by this analysis were very close to those estimated from phase 1 studies. The pharmacokinetics of docetaxel were not influenced by age.
Effect of Gender: The population pharmacokinetics analysis described above also indicated that gender did not influence the pharmacokinetics of docetaxel.
Hepatic Impairment: The population pharmacokinetic analysis described above indicated that in patients with clinical chemistry data suggestive of mild to moderate liver impairment (AST and/or ALT >1.5 times ULN concomitant with alkaline phosphatase >2.5 times ULN), total body clearance was lowered by an average of 27%, resulting in a 38% increase in systemic exposure (AUC). This average, however, includes a substantial range and there is, at present, no measurement that would allow recommendation for dose adjustment in such patients. Patients with combined abnormalities of transaminase and alkaline phosphatase should not be treated with Docetaxel Injection USP (Injection Concentrate). Patients with severe hepatic impairment have not been studied. [see Warnings and Precautions (5.2) and Use in Specific Populations (8.6)]
Effect of Race: Mean total body clearance for Japanese patients dosed at the range of 10 mg/m2 to 90 mg/m2 was similar to that of European/American populations dosed at 100 mg/m2, suggesting no significant difference in the elimination of docetaxel in the two populations.
Effect of Ketoconazole: The effect of ketoconazole (a strong CYP3A4 inhibitor) on the pharmacokinetics of docetaxel was investigated in 7 cancer patients. Patients were randomized to receive either docetaxel (100 mg/m2 intravenous) alone or docetaxel (10 mg/m2 intravenous) in combination with ketoconazole (200 mg orally once daily for 3 days) in a crossover design with a 3-week washout period. The results of this study indicated that the mean dose-normalized AUC of docetaxel was increased 2.2-fold and its clearance was reduced by 49% when docetaxel was co-administration with ketoconazole [see Dosage and Administration (2.7) and Drug-Drug Interactions (7)].
Effect of Combination Therapies:
- Dexamethasone: Docetaxel total body clearance was not modified by pretreatment with dexamethasone.
- Cisplatin: Clearance of docetaxel in combination therapy with cisplatin was similar to that previously observed following monotherapy with docetaxel. The pharmacokinetic profile of cisplatin in combination therapy with docetaxel was similar to that observed with cisplatin alone.
- Cisplatin and Fluorouracil: The combined administration of docetaxel, cisplatin and fluorouracil in 12 patients with solid tumors had no influence on the pharmacokinetics of each individual drug.
- Prednisone: A population pharmacokinetic analysis of plasma data from 40 patients with hormone-refractory metastatic prostate cancer indicated that docetaxel systemic clearance in combination with prednisone is similar to that observed following administration of docetaxel alone.
- Cyclophosphamide and Doxorubicin: A study was conducted in 30 patients with advanced breast cancer to determine the potential for drug-drug-interactions between docetaxel (75 mg/m2), doxorubicin (50 mg/m2), and cyclophosphamide (500 mg/m2) when administered in combination. The coadministration of docetaxel had no effect on the pharmacokinetics of doxorubicin and cyclophosphamide when the three drugs were given in combination compared to coadministration of doxorubicin and cyclophosphamide only. In addition, doxorubicin and cyclophosphamide had no effect on docetaxel plasma clearance when the three drugs were given in combination compared to historical data for docetaxel monotherapy.
13. NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with docetaxel have not been performed.
Docetaxel was clastogenic in the in vitro chromosome aberration test in CHO-K1 cells and in the in vivo micronucleus test in mice administered doses of 0.39 to 1.56 mg/kg (about 1/60th to 1/15th the recommended human dose on a mg/m2 basis). Docetaxel was not mutagenic in the Ames test or the CHO/HGPRT gene mutation assays.
Docetaxel did not reduce fertility in rats when administered in multiple intravenous doses of up to 0.3 mg/kg (about 1/50th the recommended human dose on a mg/m2 basis), but decreased testicular weights were reported. This correlates with findings of a 10-cycle toxicity study (dosing once every 21 days for 6 months) in rats and dogs in which testicular atrophy or degeneration was observed at intravenous doses of 5 mg/kg in rats and 0.375 mg/kg in dogs (about 1/3rd and 1/15th the recommended human dose on a mg/m2 basis, respectively). An increased frequency of dosing in rats produced similar effects at lower dose levels.
14. CLINICAL STUDIES
14.1 Locally Advanced or Metastatic Breast Cancer
The efficacy and safety of Docetaxel Injection USP (Injection Concentrate) have been evaluated in locally advanced or metastatic breast cancer after failure of previous chemotherapy (alkylating agent-containing regimens or anthracycline-containing regimens).
Randomized Trials
In one randomized trial, patients with a history of prior treatment with an anthracycline-containing regimen were assigned to treatment with Docetaxel Injection USP (Injection Concentrate) (100 mg/m2 every 3 weeks) or the combination of mitomycin (12 mg/m2 every 6 weeks) and vinblastine (6 mg/m2 every 3 weeks). Two hundred three patients were randomized to Docetaxel Injection USP (Injection Concentrate) and 189 to the comparator arm. Most patients had received prior chemotherapy for metastatic disease; only 27 patients on the Docetaxel Injection USP arm and 33 patients on the comparator arm entered the study following relapse after adjuvant therapy. Three-quarters of patients had measurable, visceral metastases. The primary endpoint was time to progression. The following table summarizes the study results (See Table 12).
| * For the risk ratio, a value less than 1.00 favors docetaxel. | |||
| Efficacy Parameter | Docetaxel (n=203) | Mitomycin/Vinblastine (n=189) | p-value |
| Median Survival | 11.4 months | 8.7 months | |
| Risk Ratio*, Mortality (Docetaxel: Control) 95% CI (Risk Ratio) | 0.73 0.58–0.93 | p=0.01 Log Rank |
|
| Median Time to Progression | 4.3 months | 2.5 months | |
| Risk Ratio*, Progression (Docetaxel: Control) 95% CI (Risk Ratio) | 0.75 0.61–0.94 | p=0.01 Log Rank |
|
| Overall Response Rate Complete Response Rate | 28.1% 3.4% | 9.5% 1.6% | p<0.0001 Chi Square |
In a second randomized trial, patients previously treated with an alkylating-containing regimen were assigned to treatment with Docetaxel Injection USP (Injection Concentrate) (100 mg/m2) or doxorubicin (75 mg/m2) every 3 weeks. One hundred sixty-one patients were randomized to Docetaxel Injection USP (Injection Concentrate) and 165 patients to doxorubicin. Approximately one-half of patients had received prior chemotherapy for metastatic disease, and one-half entered the study following relapse after adjuvant therapy. Three-quarters of patients had measurable, visceral metastases. The primary endpoint was time to progression. The study results are summarized below (See Table 13).
| * For the risk ratio, a value less than 1.00 favors docetaxel. | |||
| Efficacy Parameter | Docetaxel (n=161) | Doxorubicin (n=165) | p-value |
| Median Survival | 14.7 months | 14.3 months | |
| Risk Ratio*, Mortality (Docetaxel: Control) 95% CI (Risk Ratio) | 0.89 0.68–1.16 | p=0.39 Log Rank |
|
| Median Time to Progression | 6.5 months | 5.3 months | |
| Risk Ratio*, Progression (Docetaxel: Control) 95% CI (Risk Ratio) | 0.93 0.71–1.16 | p=0.45 Log Rank |
|
| Overall Response Rate Complete Response Rate | 45.3% 6.8% | 29.7% 4.2% | p=0.004 Chi Square |
In another multicenter open-label, randomized trial (TAX313), in the treatment of patients with advanced breast cancer who progressed or relapsed after one prior chemotherapy regimen, 527 patients were randomized to receive Docetaxel Injection USP (Injection Concentrate) monotherapy 60 mg/m2 (n=151), 75 mg/m2 (n=188) or 100 mg/m2 (n=188). In this trial, 94% of patients had metastatic disease and 79% had received prior anthracycline therapy. Response rate was the primary endpoint. Response rates increased with Docetaxel Injection USP (Injection Concentrate) dose: 19.9% for the 60 mg/m2 group compared to 22.3% for the 75 mg/m2 and 29.8% for the 100 mg/m2 group; pair-wise comparison between the 60 mg/m2 and 100 mg/m2 groups was statistically significant (p=0.037).
Single Arm Studies
Docetaxel Injection USP (Injection Concentrate) at a dose of 100 mg/m2 was studied in six single arm studies involving a total of 309 patients with metastatic breast cancer in whom previous chemotherapy had failed. Among these, 190 patients had anthracycline-resistant breast cancer, defined as progression during an anthracycline-containing chemotherapy regimen for metastatic disease, or relapse during an anthracycline-containing adjuvant regimen. In anthracycline-resistant patients, the overall response rate was 37.9% (72/190; 95% C.I.: 31.0-44.8) and the complete response rate was 2.1%.
Docetaxel Injection USP (Injection Concentrate) was also studied in three single arm Japanese studies at a dose of 60 mg/m2, in 174 patients who had received prior chemotherapy for locally advanced or metastatic breast cancer. Among 26 patients whose best response to an anthracycline had been progression, the response rate was 34.6% (95% C.I.: 17.2-55.7), similar to the response rate in single arm studies of 100 mg/m2.
14.2 Adjuvant Treatment of Breast Cancer
A multicenter, open-label, randomized trial (TAX316) evaluated the efficacy and safety of Docetaxel Injection USP (Injection Concentrate) for the adjuvant treatment of patients with axillary-node-positive breast cancer and no evidence of distant metastatic disease. After stratification according to the number of positive lymph nodes (1-3, 4+), 1491 patients were randomized to receive either Docetaxel Injection USP (Injection Concentrate) 75 mg/m2 administered 1-hour after doxorubicin 50 mg/m2 and cyclophosphamide 500 mg/m2 (TAC arm), or doxorubicin 50 mg/m2 followed by fluorouracil 500 mg/m2 and cyclosphosphamide 500 mg/m2 (FAC arm). Both regimens were administered every 3 weeks for 6 cycles. Docetaxel Injection USP (Injection Concentrate) was administered as a 1-hour infusion; all other drugs were given as intravenous bolus on day 1. In both arms, after the last cycle of chemotherapy, patients with positive estrogen and/or progesterone receptors received tamoxifen 20 mg daily for up to 5 years. Adjuvant radiation therapy was prescribed according to guidelines in place at participating institutions and was given to 69% of patients who received TAC and 72% of patients who received FAC.
Results from a second interim analysis (median follow-up 55 months) are as follows: In study TAX316, the docetaxel-containing combination regimen TAC showed significantly longer disease-free survival (DFS) than FAC (hazard ratio=0.74; 2-sided 95% CI=0.60, 0.92, stratified log rank p=0.0047). The primary endpoint, disease-free survival, included local and distant recurrences, contralateral breast cancer and deaths from any cause. The overall reduction in risk of relapse was 25.7% for TAC-treated patients. (See Figure 1).
At the time of this interim analysis, based on 219 deaths, overall survival was longer for TAC than FAC (hazard ratio=0.69, 2-sided 95% CI=0.53, 0.90). (See Figure 2). There will be further analysis at the time survival data mature.
Figure 1 - TAX316 Disease Free Survival K-M curve
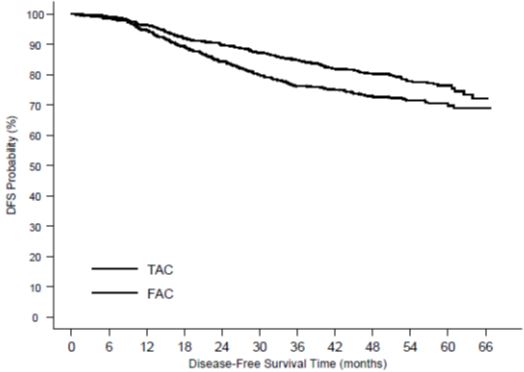
fig 1
Figure 2 - TAX316 Overall Survival K-M Curve
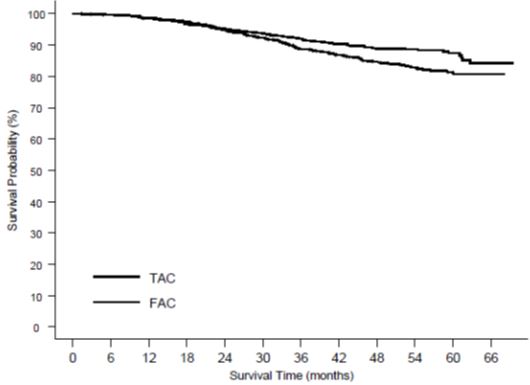
fig 2
The following table describes the results of subgroup analyses for DFS and OS (See Table 14).
| * A hazard ratio of less than 1 indicates that TAC is associated with a longer disease free survival or overall survival compared to FAC. | |||||
| Disease Free Survival | Overall Survival | ||||
| Patient subset | Number of patients | Hazard ratio* | 95% CI | Hazard ratio* | 95% CI |
| No. of positive nodes | |||||
| Overall 1–3 4+ | 744 467 277 | 0.74 0.64 0.84 | (0.60, 0.92) (0.47, 0.87) (0.63, 1.12) | 0.69 0.45 0.93 | (0.53, 0.90) (0.29, 0.70) (0.66, 1.32) |
| Receptor status | |||||
| Positive Negative | 566 178 | 0.76 0.68 | (0.59, 0.98) (0.48, 0.97) | 0.69 0.66 | (0.48, 0.99) (0.44, 0.98) |
14.3 Non-Small Cell Lung Cancer (NSCLC)
The efficacy and safety of Docetaxel Injection USP (Injection Concentrate) has been evaluated in patients with unresectable, locally advanced or metastatic non-small cell lung cancer whose disease has failed prior platinum-based chemotherapy or in patients who are chemotherapy-naïve.
Monotherapy with Docetaxel Injection USP (Injection Concentrate) for NSCLC Previously Treated with Platinum-Based Chemotherapy
Two randomized, controlled trials established that a Docetaxel Injection USP (Injection Concentrate) dose of 75 mg/m2 was tolerable and yielded a favorable outcome in patients previously treated with platinum-based chemotherapy (see below). Docetaxel Injection USP (Injection Concentrate) at a dose of 100 mg/m2, however, was associated with unacceptable hematologic toxicity, infections, and treatment-related mortality and this dose should not be used [see Boxed Warning, Dosage and Administration (2.7), Warnings and Precautions (5.3)].
One trial (TAX317), randomized patients with locally advanced or metastatic non-small cell lung cancer, a history of prior platinum-based chemotherapy, no history of taxane exposure, and an ECOG performance status ≤2 to Docetaxel Injection USP (Injection Concentrate) or best supportive care. The primary endpoint of the study was survival. Patients were initially randomized to Docetaxel Injection USP 100 mg/m2 or best supportive care, but early toxic deaths at this dose led to a dose reduction to Docetaxel Injection USP 75 mg/m2. A total of 104 patients were randomized in this amended study to either Docetaxel Injection USP 75 mg/m2 or best supportive care.
In a second randomized trial (TAX320), 373 patients with locally advanced or metastatic non-small cell lung cancer, a history of prior platinum-based chemotherapy, and an ECOG performance status ≤2 were randomized to Docetaxel Injection USP (Injection Concentrate) 75 mg/m2, Docetaxel Injection USP (Injection Concentrate) 100 mg/m2 and a treatment in which the investigator chose either vinorelbine 30 mg/m2 days 1, 8, and 15 repeated every 3 weeks or ifosfamide 2 g/m2 days 1-3 repeated every 3 weeks. Forty percent of the patients in this study had a history of prior paclitaxel exposure. The primary endpoint was survival in both trials. The efficacy data for the Docetaxel Injection USP (Injection Concentrate) 75 mg/m2 arm and the comparator arms are summarized in Table 15 and Figures 3 and 4 showing the survival curves for the two studies.
| * Vinorelbine/Ifosfamide ** p≤0.05 † uncorrected for multiple comparisons †† a value less than 1.00 favors docetaxel |
||||
| TAX317 | TAX320 | |||
| Docetaxel 75 mg/m2 n=55 | Best Supportive Care n=49 | Docetaxel 75 mg/m2 n=125 | Control (V/I*) n=123 |
|
| Overall Survival Log-rank Test | p=0.01 | p=0.13 |
||
| Risk Ratio††, Mortality (Docetaxel: Control) 95% CI (Risk Ratio) | 0.56 (0.35, 0.88) | 0.82 (0.63, 1.06) |
||
| Median Survival 95% CI | 7.5 months ** (5.5, 12.8) | 4.6 months (3.7, 6.1) | 5.7 months (5.1, 7.1) | 5.6 months (4.4, 7.9) |
| % 1-year Survival 95% CI | 37%**†
(24, 50) | 12% (2, 23) | 30%**†
(22, 39) | 20% (13, 27) |
| Time to Progression 95% CI | 12.3 weeks** (9.0, 18.3) | 7.0 weeks (6.0, 9.3) | 8.3 weeks (7.0, 11.7) | 7.6 weeks (6.7, 10.1) |
| Response Rate 95% CI | 5.5% (1.1, 15.1) | Not Applicable | 5.7% (2.3, 11.3) | 0.8% (0.0, 4.5) |
Only one of the two trials (TAX317) showed a clear effect on survival, the primary endpoint; that trial also showed an increased rate of survival to one year. In the second study (TAX320) the rate of survival at one year favored Docetaxel Injection USP 75 mg/m2.
Figure 3 - TAX317 Survival K-M Curves - Docetaxel Injection USP (Injection Concentrate) 75
mg/m2 vs. Best Supportive Care
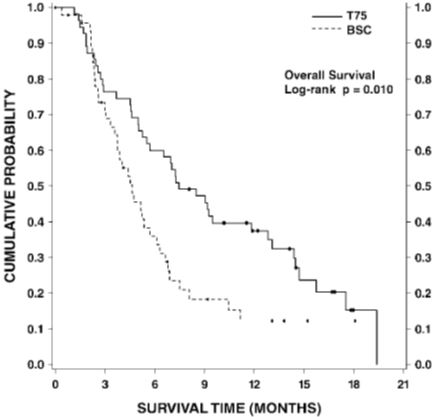
Fig 3
Figure 4 - TAX320 Survival K-M Curves - Docetaxel Injection USP (Injection Concentrate)
75 mg/m2 vs. Vinorelbine or Ifosfamide Control
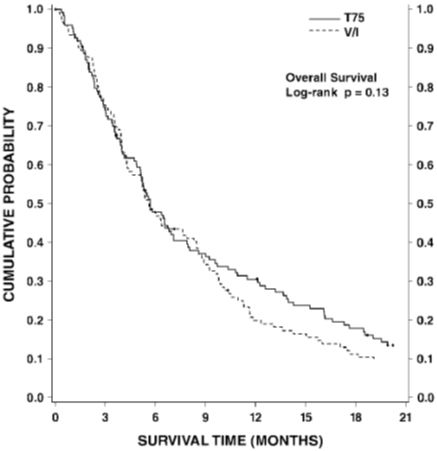
Fig 4
Patients treated with Docetaxel Injection USP (Injection Concentrate) at a dose of 75 mg/m2 experienced no deterioration in performance status and body weight relative to the comparator arms used in these trials.
Combination Therapy with Docetaxel Injection USP (Injection Concentrate) for Chemotherapy-Naïve NSCLC
In a randomized controlled trial (TAX326), 1218 patients with unresectable stage IIIB or IV NSCLC and no prior chemotherapy were randomized to receive one of three treatments: Docetaxel Injection USP (Injection Concentrate) 75 mg/m2 as a 1 hour infusion immediately followed by cisplatin 75 mg/m2 over 30 to 60 minutes every 3 weeks; vinorelbine 25 mg/m2 administered over 6-10 minutes on days 1, 8, 15, 22 followed by cisplatin 100 mg/m2 administered on day 1 of cycles repeated every 4 weeks; or a combination of Docetaxel Injection USP (Injection Concentrate) and carboplatin.
The primary efficacy endpoint was overall survival. Treatment with Docetaxel Injection USP (Injection Concentrate)+cisplatin did not result in a statistically significantly superior survival compared to vinorelbine+cisplatin (see table below). The 95% confidence interval of the hazard ratio (adjusted for interim analysis and multiple comparisons) shows that the addition of Docetaxel Injection USP (Injection Concentrate) to cisplatin results in an outcome ranging from a 6% inferior to a 26% superior survival compared to the addition of vinorelbine to cisplatin. The results of a further statistical analysis showed that at least (the lower bound of the 95% confidence interval) 62% of the known survival effect of vinorelbine when added to cisplatin (about a 2-month increase in median survival; Wozniak et al. JCO, 1998) was maintained. The efficacy data for the Docetaxel Injection USP (Injection Concentrate)+cisplatin arm and the comparator arm are summarized in Table 16.
| a From the superiority test (stratified log rank) comparing Docetaxel Injection USP (Injection Concentrate)+cisplatin to vinorelbine+cisplatin | ||
| b Hazard ratio of Docetaxel Injection USP (Injection Concentrate)+cisplatin vs. vinorelbine+cisplatin. A hazard ratio of less than 1 indicates that Docetaxel Injection USP (Injection Concentrate)+cisplatin is associated with a longer survival. |
||
| c Adjusted for interim analysis and multiple comparisons. | ||
| Comparison | Docetaxel Injection USP (Injection Concentrate) +Cisplatin n=408 | Vinorelbine+Cisplatin n=405 |
| Kaplan-Meier Estimate of Median Survival | 10.9 months | 10.0 months |
| p-value a | 0.122 | |
| Estimated Hazard Ratio b | 0.88 | |
| Adjusted 95% CI c | (0.74, 1.06) | |
The second comparison in the same three-arm study, vinorelbine+cisplatin versus Docetaxel Injection USP (Injection Concentrate)+carboplatin, did not demonstrate superior survival associated with the Docetaxel Injection USP (Injection Concentrate) arm (Kaplan-Meier estimate of median survival was 9.1 months for Docetaxel Injection USP (Injection Concentrate)+carboplatin compared to 10.0 months on the vinorelbine+cisplatin arm) and the Docetaxel Injection USP (Injection Concentrate)+carboplatin arm did not demonstrate preservation of at least 50% of the survival effect of vinorelbine added to cisplatin. Secondary endpoints evaluated in the trial included objective response and time to progression. There was no statistically significant difference between Docetaxel Injection USP (Injection Concentrate)+cisplatin and vinorelbine+cisplatin with respect to objective response and time to progression (see Table 17).
| a Adjusted for multiple comparisons. | |||
| b Kaplan-Meier estimates. | |||
| Endpoint |
Docetaxel Injection USP (Injection +Cisplatin | Vinorelbine+Cisplatin | p-value |
| Objective Response Rate (95% CI) a | 31.6% (26.5%, 36.8%) | 24.4% (19.8%, 29.2%) | Not Significant |
| Median Time to Progression b
(95% CI) a | 21.4 weeks (19.3, 24.6) | 22.1 weeks (18.1, 25.6) | Not Significant |
14.4 Hormone Refractory Prostate Cancer
The safety and efficacy of Docetaxel Injection USP (Injection Concentrate) in combination with prednisone in patients with androgen independent (hormone refractory) metastatic prostate cancer were evaluated in a randomized multicenter active control trial. A total of 1006 patients with Karnofsky Performance Status (KPS) ≥60 were randomized to the following treatment groups:
- Docetaxel Injection USP (Injection Concentrate) 75 mg/m2 every 3 weeks for 10 cycles.
- Docetaxel Injection USP (Injection Concentrate) 30 mg/m2 administered weekly for the first 5 weeks in a 6-week cycle for 5 cycles.
- Mitoxantrone 12 mg/m2 every 3 weeks for 10 cycles.
All 3 regimens were administered in combination with prednisone 5 mg twice daily, continuously. In the Docetaxel Injection USP (Injection Concentrate) every three week arm, a statistically significant overall survival advantage was demonstrated compared to mitoxantrone. In the Docetaxel Injection USP (Injection Concentrate) weekly arm, no overall survival advantage was demonstrated compared to the mitoxantrone control arm. Efficacy results for the Docetaxel Injection USP (Injection Concentrate) every 3 week arm versus the control arm are summarized in Table 18 and Figure 5.
| * Stratified log rank test. Threshold for statistical significance = 0.0175 because of 3 arms. | ||
|
Docetaxel Injection USP (Injection Concentrate)
+ Prednisone
| Mitoxantrone+ Prednisone
every 3 weeks |
|
| Number of patients | 335 | 337 |
| Median survival (months) | 18.9 | 16.5 |
| 95% CI | (17.0–21.2) | (14.4–18.6) |
| Hazard ratio | 0.761 | -- |
| 95% CI | (0.619–0.936) | -- |
| p-value* | 0.0094 | -- |
Figure 5 - TAX327 Survival K-M Curves
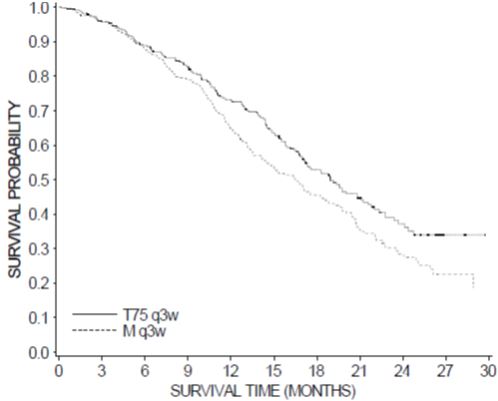
fig 5
14.5 Gastric Adenocarcinoma
A multicenter, open-label, randomized trial was conducted to evaluate the safety and efficacy of Docetaxel Injection USP (Injection Concentrate) for the treatment of patients with advanced gastric adenocarcinoma, including adenocarcinoma of the gastroesophageal junction, who had not received prior chemotherapy for advanced disease. A total of 445 patients with KPS >70 were treated with either Docetaxel Injection USP (Injection Concentrate) (T) (75 mg/m2 on day 1) in combination with cisplatin (C) (75 mg/m2 on day 1) and fluorouracil (F) (750 mg/m2 per day for 5 days) or cisplatin (100 mg/m2 on day 1) and fluorouracil (1000 mg/m2 per day for 5 days). The length of a treatment cycle was 3 weeks for the TCF arm and 4 weeks for the CF arm. The demographic characteristics were balanced between the two treatment arms. The median age was 55 years, 71% were male, 71% were Caucasian, 24% were 65 years of age or older, 19% had a prior curative surgery and 12% had palliative surgery. The median number of cycles administered per patient was 6 (with a range of 1-16) for the TCF arm compared to 4 (with a range of 1-12) for the CF arm. Time to progression (TTP) was the primary endpoint and was defined as time from randomization to disease progression or death from any cause within 12 weeks of the last evaluable tumor assessment or within 12 weeks of the first infusion of study drugs for patients with no evaluable tumor assessment after randomization. The hazard ratio (HR) for TTP was 1.47 (CF/TCF, 95% CI: 1.19-1.83) with a significantly longer TTP (p=0.0004) in the TCF arm. Approximately 75% of patients had died at the time of this analysis. Overall survival was significantly longer (p=0.0201) in the TCF arm with a HR of 1.29 (95% CI: 1.04-1.61). Efficacy results are summarized in Table 19 and Figures 6 and 7.
| *Unstratified log-rank test | ||
| †For the hazard ratio (TCF/CF), values less than 1.00 favor the Docetaxel Injection USP (Injection Concentrate) arm. | ||
| Endpoint | TCF n=221 | CF n=224 |
| Median TTP (months) (95%CI) | 5.6 (4.86–5.91) | 3.7 (3.45–4.47) |
| Hazard ratio†
(95%CI) *p-value | 0.68 (0.55–0.84) 0.0004 |
|
| Median survival (months) (95%CI) | 9.2 (8.38–10.58) | 8.6 (7.16–9.46) |
| Hazard ratio†
(95%CI) *p-value | 0.77 (0.62–0.96) 0.0201 |
|
| Overall Response Rate (CR+PR) (%) | 36.7 | 25.4 |
| p-value | 0.0106 | |
Subgroup analyses were consistent with the overall results across age, gender and race.
Figure 6 - Gastric Cancer Study (TAX325) Time to Progression K-M Curve
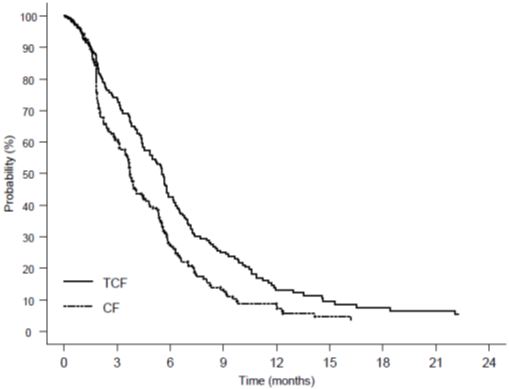
fig 6
Figure 7 - Gastric Cancer Study (TAX325) Survival K-M Curve
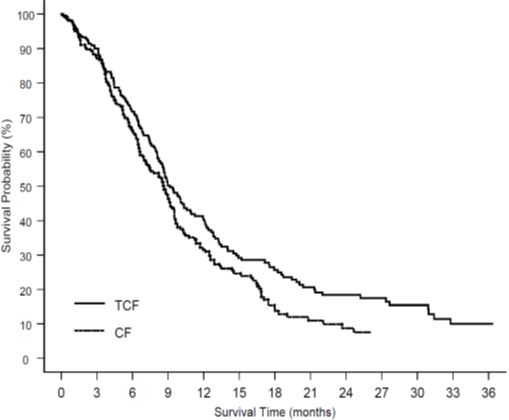
fig 7
14.6 Head and Neck Cancer
Induction chemotherapy followed by radiotherapy (TAX323)
The safety and efficacy of Docetaxel Injection USP (Injection Concentrate) in the induction treatment of patients with squamous cell carcinoma of the head and neck (SCCHN) was evaluated in a multicenter, open-label, randomized trial (TAX323). In this study, 358 patients with inoperable locally advanced SCCHN, and WHO performance status 0 or 1, were randomized to one of two treatment arms. Patients on the Docetaxel Injection USP (Injection Concentrate) arm received Docetaxel Injection USP (Injection Concentrate) (T) 75 mg/m2 followed by cisplatin (P) 75 mg/m2 on Day 1, followed by fluorouracil (F) 750 mg/m2 per day as a continuous infusion on Days 1-5. The cycles were repeated every three weeks for 4 cycles. Patients whose disease did not progress received radiotherapy (RT) according to institutional guidelines (TPF/RT). Patients on the comparator arm received cisplatin (P) 100 mg/m2 on Day 1, followed by fluorouracil (F) 1000 mg/m2/day as a continuous infusion on Days 1-5. The cycles were repeated every three weeks for 4 cycles. Patients whose disease did not progress received RT according to institutional guidelines (PF/RT). At the end of chemotherapy, with a minimal interval of 4 weeks and a maximal interval of 7 weeks, patients whose disease did not progress received radiotherapy (RT) according to institutional guidelines. Locoregional therapy with radiation was delivered either with a conventional fraction regimen (1.8 Gy-2.0 Gy once a day, 5 days per week for a total dose of 66 to 70 Gy) or with an accelerated/hyperfractionated regimen (twice a day, with a minimum interfraction interval of 6 hours, 5 days per week, for a total dose of 70 to 74 Gy, respectively). Surgical resection was allowed following chemotherapy, before or after radiotherapy.
The primary endpoint in this study, progression-free survival (PFS), was significantly longer in the TPF arm compared to the PF arm, p=0.0077 (median PFS: 11.4 vs. 8.3 months respectively) with an overall median follow up time of 33.7 months. Median overall survival with a median follow-up of 51.2 months was also significantly longer in favor of the TPF arm compared to the PF arm (median OS: 18.6 vs. 14.2 months respectively). Efficacy results are presented in Table 20 and Figures 8 and 9.
| A Hazard ratio of less than 1 favors Docetaxel Injection USP (Injection Concentrate) +Cisplatin+Fluorouracil | ||
| * Stratified log-rank test based on primary tumor site | ||
| ** Stratified log-rank test, not adjusted for multiple comparisons | ||
| *** Chi square test, not adjusted for multiple comparisons | ||
| ENDPOINT | Docetaxel Injection USP (Injection Concentrate) + Cisplatin+ Fluorouracil n=177 |
Cisplatin+ Fluorouracil n=181 |
| Median progression free survival (months) (95%CI) | 11.4 (10.1–14.0) | 8.3 (7.4–9.1) |
| Adjusted Hazard ratio (95%CI) *p-value | 0.71 (0.56–0.91) 0.0077 |
|
| Median survival (months) (95%CI) | 18.6 (15.7–24.0) | 14.2 (11.5–18.7) |
| Hazard ratio (95%CI) **p-value | 0.71 (0.56–0.90) 0.0055 |
|
| Best overall response (CR + PR) to chemotherapy (%) (95%CI) | 67.8 (60.4–74.6) | 53.6 (46.0–61.0) |
| ***p-value | 0.006 | |
| Best overall response (CR + PR) to study treatment [chemotherapy +/- radiotherapy] (%) (95%CI) | 72.3 (65.1–78.8) | 58.6 (51.0–65.8) |
| ***p-value | 0.006 | |
Figure 8 - TAX323 Progression-Free Survival K-M Curve
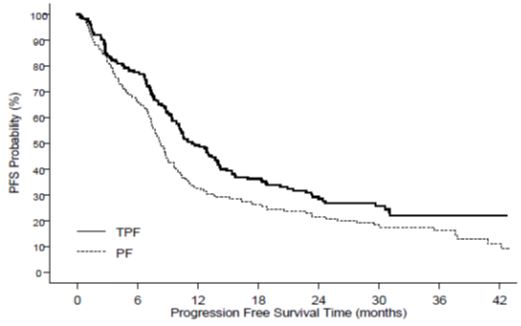
fig 8
Figure 9 - TAX323 Overall Survival K-M Curve
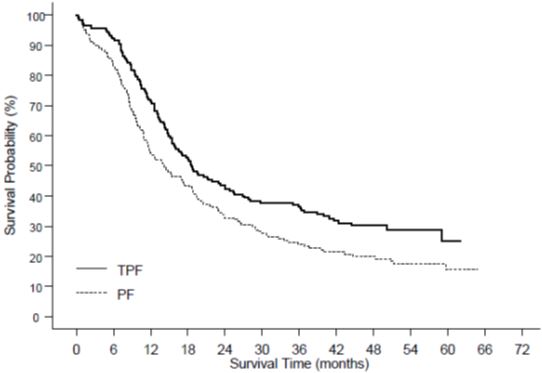
fig 9
Induction chemotherapy followed by chemoradiotherapy (TAX324)
The safety and efficacy of Docetaxel Injection USP (Injection Concentrate) in the induction treatment of patients with locally advanced (unresectable, low surgical cure, or organ preservation) SCCHN was evaluated in a randomized, multicenter open-label trial (TAX324). In this study, 501 patients, with locally advanced SCCHN, and a WHO performance status of 0 or 1, were randomized to one of two treatment arms. Patients on the Docetaxel Injection USP (Injection Concentrate) arm received Docetaxel Injection USP (Injection Concentrate) (T) 75 mg/m2 by intravenous infusion on day 1 followed by cisplatin (P) 100 mg/m2 administered as a 30-minute to three-hour intravenous infusion, followed by the continuous intravenous infusion of fluorouracil (F) 1000 mg/m2/day from day 1 to day 4. The cycles were repeated every 3 weeks for 3 cycles. Patients on the comparator arm received cisplatin (P) 100 mg/m2 as a 30-minute to three-hour intravenous infusion on day 1 followed by the continuous intravenous infusion of fluorouracil (F) 1000 mg/m2/day from day 1 to day 5. The cycles were repeated every 3 weeks for 3 cycles.
All patients in both treatment arms who did not have progressive disease were to receive 7 weeks of chemoradiotherapy (CRT) following induction chemotherapy 3 to 8 weeks after the start of the last cycle. During radiotherapy, carboplatin (AUC 1.5) was given weekly as a one-hour intravenous infusion for a maximum of 7 doses. Radiation was delivered with megavoltage equipment using once daily fractionation (2 Gy per day, 5 days per week for 7 weeks for a total dose of 70-72 Gy). Surgery on the primary site of disease and/or neck could be considered at anytime following completion of CRT. The primary efficacy endpoint, overall survival (OS), was significantly longer (log-rank test, p=0.0058) with the Docetaxel Injection USP (Injection Concentrate)-containing regimen compared to PF [median OS: 70.6 versus 30.1 months respectively, hazard ratio (HR)=0.70, 95% confidence interval (CI)= 0.54 -0.90]. Overall survival results are presented in Table 21 and Figure 10.
| A Hazard ratio of less than 1 favors Docetaxel Injection USP (Injection Concentrate) +cisplatin+fluorouracil | ||
| * un-adjusted log-rank test | ||
| NE - not estimable | ||
| ENDPOINT | Docetaxel Injection USP (Injection Concentrate) + Cisplatin+ Fluorouracil n=255 | Cisplatin+ Fluorouracil n=246 |
| Median overall survival (months) (95% CI) | 70.6 (49.0–NE) | 30.1 (20.9–51.5) |
| Hazard ratio: (95% CI) *p-value | 0.70 (0.54–0.90) 0.0058 |
|
Figure 10 - TAX324 Overall Survival K-M Curve
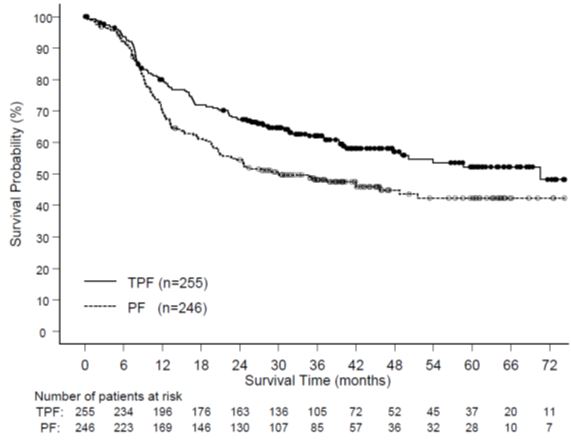
fig 10
15. REFERENCES
1. NIOSH Alert: Preventing occupational exposures to antineoplastic and other hazardous drugs in healthcare settings. 2004. U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 2004-165.
2. OSHA Technical Manual, TED 1-0.15A, Section VI: Chapter 2. Controlling Occupational Exposure to Hazardous Drugs. OSHA, 1999.
3. American Society of Health-System Pharmacists. (2006) ASHP Guidelines on Handling Hazardous Drugs. Am J Health-Syst Pharm. 2006;63:1172-1193
4. Polovich, M., White, J. M., & Kelleher, L.O. (eds.) 2005. Chemotherapy and biotherapy guidelines and recommendations for practice (2nd. ed.) Pittsburgh, PA: Oncology Nursing Society
16. HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
One-vial Docetaxel Injection USP (Injection Concentrate)
Docetaxel Injection USP (Injection Concentrate) (20 mg/mL) requires NO prior dilution with a diluent and is ready to add to the infusion solution.
Docetaxel Injection USP (Injection Concentrate) is supplied in a single use vial as a sterile, pyrogen-free, solution.
Docetaxel Injection USP (Injection Concentrate) 20 mg/mL (NDC 25021-222-01)
Docetaxel Injection USP (Injection Concentrate) 20 mg/1 mL: 20 mg docetaxel in 1 mL in 50/50 (v/v) ratio polysorbate 80/dehydrated alcohol, and anhydrous citric acid.
Docetaxel Injection USP (Injection Concentrate) 80 mg/4 mL (20 mg/mL) (NDC 25021-222-04)
Docetaxel Injection USP (Injection Concentrate) 80 mg/4 mL: 80 mg docetaxel in 4 mL 50/50 (v/v) ratio polysorbate 80/dehydrated alcohol, and anhydrous citric acid.
Docetaxel Injection USP (Injection Concentrate) 200 mg/10 mL (20 mg/mL) (NDC 25021-222-10)
Docetaxel Injection USP (Injection Concentrate) 200 mg/10 mL: 200 mg docetaxel in 10 mL 50/50 (v/v) ratio polysorbate 80/dehydrated alcohol, and anhydrous citric acid.
16.2 Storage
Store between 2°C and 25°C (36°F and 77°F). Retain in the original package to protect from light. Freezing does not adversely affect the product.
16.3 Handling and Disposal
Procedures for proper handling and disposal of anticancer drugs should be considered. Several guidelines on this subject have been published [see References (15)].
17. PATIENT COUNSELING INFORMATION
See FDA-Approved Patient Labeling
- Docetaxel Injection USP (Injection Concentrate) may cause fetal harm. Advise patients to avoid becoming pregnant while receiving this drug. Women of childbearing potential should use effective contraceptives if receiving Docetaxel Injection USP (Injection Concentrate) [see Warnings and Precautions (5.12) and Use in Specific Populations (8.1)].
- Obtain detailed allergy and concomitant drug information from the patient prior to Docetaxel Injection USP (Injection Concentrate) administration.
- Explain the significance of oral corticosteroids such as dexamethasone administration to the patient to help facilitate compliance. Instruct patients to report if they were not compliant with oral corticosteroid regimen.
- Instruct patients to immediately report signs of a hypersensitivity reaction.
- Tell patients to watch for signs of fluid retention such as peripheral edema in the lower extremities, weight gain and dyspnea.
- Explain the significance of routine blood cell counts. Instruct patients to monitor their temperature frequently and immediately report any occurrence of fever.
- Instruct patients to report myalgia, cutaneous, or neurologic reactions.
- Explain to patients the possible effects of the alcohol content in Docetaxel Injection USP (Injection Concentrate) Injection, including possible effects on the central nervous system. Patients in whom alcohol should be avoided or minimized should consider the alcohol content of Docetaxel Injection USP (Injection Concentrate) Injection. Alcohol could impair their ability to drive or use machines immediately after infusion.
- Explain to patients that side effects such as nausea, vomiting, diarrhea, constipation, fatigue, excessive tearing, infusion site reactions, and hair loss (cases of permanent hair loss have been reported) are associated with docetaxel administration.
Patient Information
Patient Information
Docetaxel Injection USP (Injection Concentrate) (doe” se tax’ el)
(docetaxel) Injection Concentrate
Intravenous Infusion
Read this Patient Information before you receive your first treatment with Docetaxel Injection USP (Injection Concentrate) and each time before you are treated. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment.
What is the most important information I should know about Docetaxel Injection USP (Injection Concentrate)?
Docetaxel Injection USP (Injection Concentrate) can cause serious side effects, including death.
-
The chance of death in people who receive Docetaxel Injection USP (Injection Concentrate) is higher if you:
- have liver problems
- receive high doses of Docetaxel Injection USP (Injection Concentrate)
- have non-small cell lung cancer and have been treated with chemotherapy medicines that contain platinum
- Docetaxel Injection USP (Injection Concentrate) can affect your blood cells. Your doctor should do routine blood tests during treatment with Docetaxel Injection USP (Injection Concentrate). This will include regular checks of your white blood cell counts. If your white blood cells are too low, your doctor may not treat you with Docetaxel Injection USP (Injection Concentrate) until you have enough white blood cells. People with low white blood counts can develop life-threatening infections. The earliest sign of infection may be fever. Follow your doctor's instructions for how often to take your temperature while taking Docetaxel Injection USP (Injection Concentrate). Call your doctor right away if you have a fever.
-
Serious allergic reactions can happen in people who take Docetaxel Injection USP (Injection Concentrate). Serious allergic reactions are medical emergencies that can lead to death and must be treated right away.
Tell your doctor right away if you have any of these signs of a serious allergic reaction:- trouble breathing
- sudden swelling of your face, lips, tongue, throat, or trouble swallowing
- hives (raised bumps), rash, or redness all over your body
- Your body may hold too much fluid (severe fluid retention) during treatment with Docetaxel Injection USP (Injection Concentrate). This can be life threatening. To decrease the chance of this happening, you must take another medicine, a corticosteroid, before each Docetaxel Injection USP (Injection Concentrate) treatment. You must take the corticosteroid exactly as your doctor tells you. Tell your doctor or nurse before your Docetaxel Injection USP (Injection Concentrate) treatment if you forget to take the corticosteroid dose or do not take it as your doctor tells you.
What is Docetaxel Injection USP (Injection Concentrate)?
Docetaxel Injection USP (Injection Concentrate) is a prescription anti-cancer medicine used to treat certain people with:
- breast cancer
- non-small cell lung cancer
- prostate cancer
- stomach cancer
- head and neck cancer
It is not known if Docetaxel Injection USP (Injection Concentrate) is effective in children.
Who should not receive Docetaxel Injection USP (Injection Concentrate)?
Do not receive Docetaxel Injection USP (Injection Concentrate) if you:
- have had a severe allergic reaction to:
- docetaxel, the active ingredient in Docetaxel Injection USP (Injection Concentrate), or
- any other medicines that contain polysorbate 80. Ask your doctor or pharmacist if you are not sure.
- have a low white blood cell count.
What should I tell my doctor before receiving Docetaxel Injection USP (Injection Concentrate)?
Before you receive Docetaxel Injection USP (Injection Concentrate), tell your doctor if you:
- are allergic to any medicines. See "Who should not receive Docetaxel Injection USP (Injection Concentrate)?" Also, see the end of this leaflet for a complete list of the ingredients in Docetaxel Injection USP (Injection Concentrate).
- have liver problems
- have any other medical conditions
- are pregnant or plan to become pregnant. Docetaxel Injection USP (Injection Concentrate) can harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if Docetaxel Injection USP (Injection Concentrate) passes into your breast milk. You and your doctor should decide if you will receive Docetaxel Injection USP (Injection Concentrate) or breastfeed.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Docetaxel Injection USP (Injection Concentrate) may affect the way other medicines work, and other medicines may affect the way Docetaxel Injection USP (Injection Concentrate) works.
Know the medicines you take. Keep a list of them to show doctor and pharmacist when you get a new medicine.
How will I receive Docetaxel Injection USP (Injection Concentrate)?
- Docetaxel Injection USP (Injection Concentrate) will be given to you as an intravenous (IV) injection into your vein, usually over 1 hour.
- Docetaxel Injection USP (Injection Concentrate) is usually given every 3 weeks.
- Your doctor will decide how long you will receive treatment with Docetaxel Injection USP (Injection Concentrate).
- Your doctor will check your blood cell counts and other blood tests during your treatment with Docetaxel Injection USP (Injection Concentrate) to check for side effects of Docetaxel Injection USP (Injection Concentrate).
- Your doctor may stop your treatment, change the timing of your treatment, or change the dose of your treatment if you have certain side effects while receiving Docetaxel Injection USP (Injection Concentrate).
What are the possible side effects of Docetaxel Injection USP (Injection Concentrate)?
Docetaxel Injection USP (Injection Concentrate) may cause serious side effects including death.
- See "What is the most important information I should know about Docetaxel Injection USP (Injection Concentrate)?"
- Acute Myeloid Leukemia (AML), a type of blood cancer, can happen in people who take Docetaxel Injection USP (Injection Concentrate) along with certain other medicines.
- Other Blood Disorders. Changes in blood counts due to leukemia and other blood disorders may occur years after treatment with Docetaxel Injection USP (Injection Concentrate).
- Skin Reactions including redness and swelling of your arms and legs with peeling of your skin.
- Neurologic Symptoms including numbness, tingling, or burning in your hands and feet.
- Vision Problems including blurred vision or loss of vision.
-
Docetaxel Injection USP (Injection Concentrate) Injection contains alcohol.
The alcohol content in Docetaxel Injection USP (Injection Concentrate) Injection may impair your ability to drive or use machinery right after receiving Docetaxel Injection USP (Injection Concentrate) Injection. Consider whether you should drive, operate machinery or do other dangerous activities right after you receive Docetaxel Injection USP (Injection Concentrate) Injection treatment.
The most common side effects of Docetaxel Injection USP (Injection Concentrate) include:
- changes in your sense of taste
- feeling short of breath
- constipation
- decreased appetite
- changes in your fingernails or toenails
- swelling of your hands, face or feet
- feeling weak or tired
- joint and muscle pain
- nausea and vomiting
- diarrhea
- mouth or lips sores
- hair loss: in most cases normal hair growth should return. In some cases (frequency not known) permanent hair loss has been observed.
- rash
- redness of the eye, excess tearing
- skin reactions at the site of Docetaxel Injection USP (Injection Concentrate) administration such as increased skin pigmentation, redness, tenderness, swelling, warmth or dryness of the skin.
- tissue damage if Docetaxel Injection USP (Injection Concentrate) leaks out of the vein into the tissues
Tell your doctor if you have any side effect that bothers you or does not go away.
These are not all the possible side effects of Docetaxel Injection USP (Injection Concentrate). For more information ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about Docetaxel Injection USP (Injection Concentrate)
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. This Patient Information leaflet summarizes the most important information about Docetaxel Injection USP (Injection Concentrate). If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about Docetaxel Injection USP (Injection Concentrate) that is written for health professionals.
For more information, call 1-800-295-0482.
What are the ingredients in Docetaxel Injection USP (Injection Concentrate)?
Active ingredient: docetaxel
Inactive ingredients include: ethanol, polysorbate 80, and anhydrous citric acid
| Every three-week injection of Docetaxel Injection USP (Injection Concentrate) for breast, non-small cell lung and stomach, and head and neck cancers Take your oral corticosteroid medicine as your doctor tells you. |
| Oral corticosteroid dosing: |
| Day 1 Date:_________ Time: ______ AM _______ PM Day 2 Date:_________ Time: ______ AM _______ PM (Docetaxel Injection USP (Injection Concentrate) Treatment Day) Day 3 Date:_________ Time: ______ AM _______ PM |
| Every three-week injection of Docetaxel Injection USP (Injection Concentrate) for prostate cancer Take your oral corticosteroid medicine as your doctor tells you. |
| Oral corticosteroid dosing: |
| Date: _________ Time: ___________ Date: _________ Time: ___________ (Docetaxel Injection USP (Injection Concentrate) Treatment Day) Time: ___________ |
This Patient Information has been approved by the U.S. Food and Drug Administration.
Manufactured in Argentina. Manufactured by Eriochem S.A., Argentina
Manufactured for X-GEN Pharmaceuticals, Inc.
Big Flats, NY 14814
Revised: 12/2017
MST-PR-4175-02
PRINCIPAL DISPLAY PANEL - 20 mg/1 mL Vial Label
PRINCIPAL DISPLAY PANEL - NDC: 39822-2120-1 20 mg/mL Vial Label

PRINCIPAL DISPLAY PANEL - 20 mg/1 mL Carton
PRINCIPAL DISPLAY PANEL - NDC: 39822-2120-1 20 mg/mL Carton Label
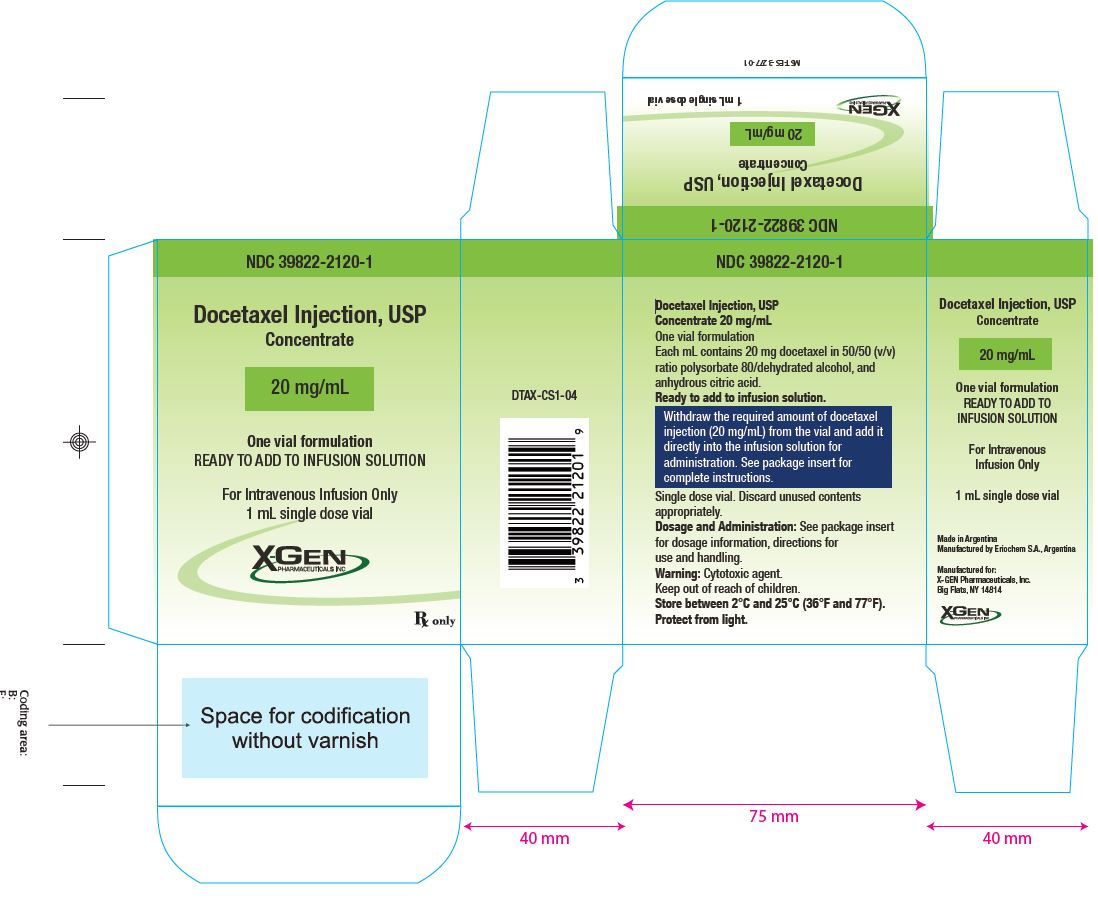
PRINCIPAL DISPLAY PANEL - 80 mg/4 mL Vial Label
PRINCIPAL DISPLAY PANEL - NDC:39822-2180-1 80 mg/4 mL Vial Label

PRINCIPAL DISPLAY PANEL - 80 mg/4 mL Carton
PRINCIPAL DISPLAY PANEL - NDC: 39822-2180-1 80 mg/4 mL Carton Label
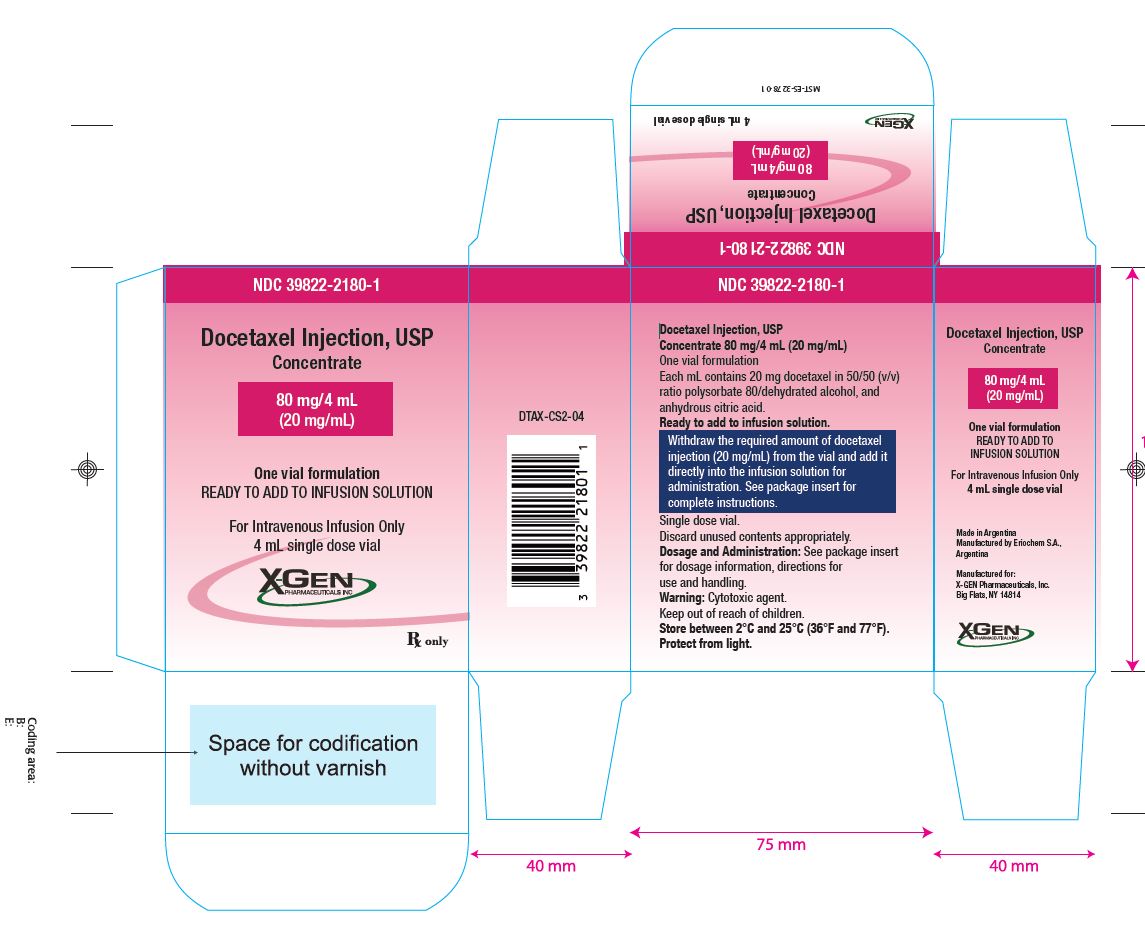
| DOCETAXEL
docetaxel injection, solution, concentrate |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| DOCETAXEL
docetaxel injection, solution, concentrate |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| DOCETAXEL
docetaxel injection, solution, concentrate |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - XGen Pharmaceuticals DJB, Inc. (117380305) |