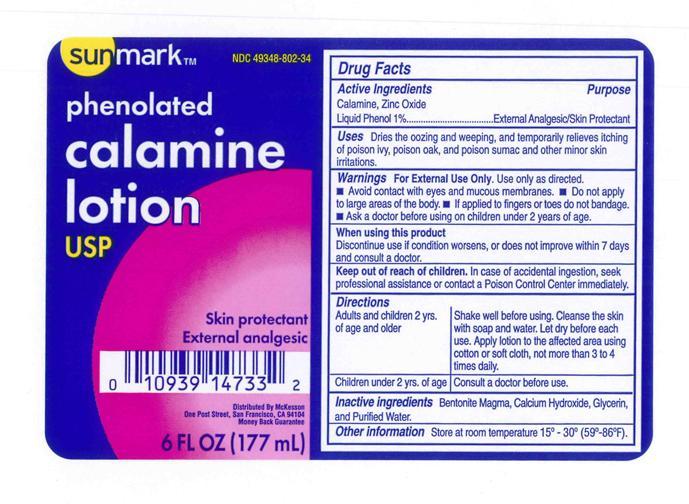
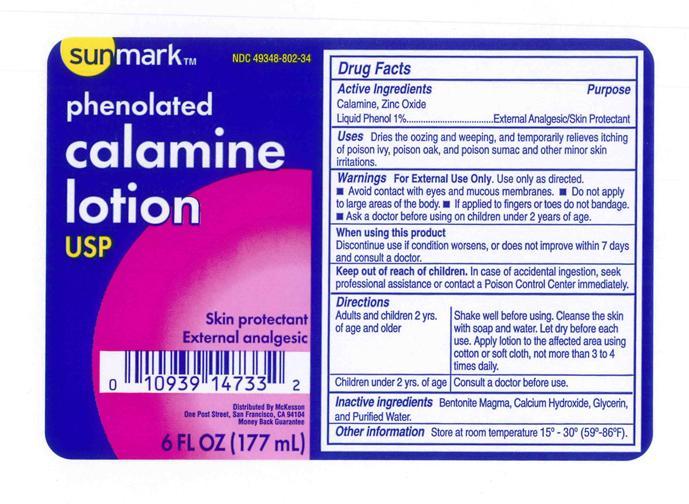
Label: SUNMARK PHENOLATED CALAMINE- calamine and zinc oxide and phenol lotion
- NDC Code(s): 49348-802-34
- Packager: Strategic Sourcing Services LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 19, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Active Ingredient
- Purpose
- Active Ingredient
- Purpose
- Uses
- Warnings
- Ask a doctor
- Keep out of reach of children.
- directions (Shake well before using)
- Other Information.
- Inactive Ingredients.
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
SUNMARK PHENOLATED CALAMINE
calamine and zinc oxide and phenol lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49348-802 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 160 mg in 1 mL PHENOL (UNII: 339NCG44TV) (PHENOL - UNII:339NCG44TV) PHENOL 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENTONITE (UNII: A3N5ZCN45C) CALCIUM HYDROXIDE (UNII: PF5DZW74VN) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49348-802-34 177 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 12/12/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M016 03/25/1998 Labeler - Strategic Sourcing Services LLC (116956644) Registrant - Pharma Nobis, LLC (118564114) Establishment Name Address ID/FEI Business Operations Pharma Nobis, LLC 118564114 analysis(49348-802) , manufacture(49348-802) , pack(49348-802) , label(49348-802)