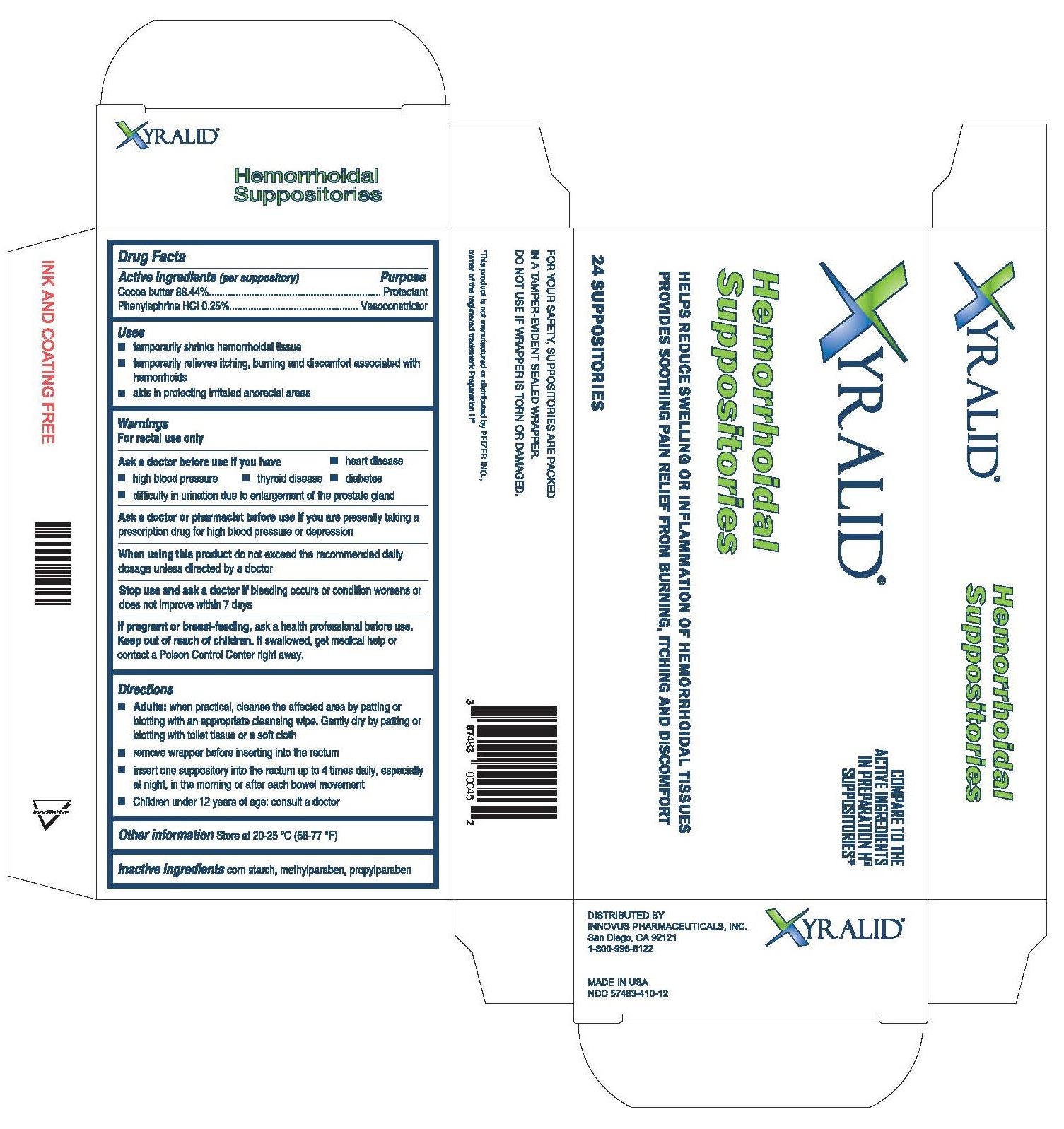
Label: XYRALID HEMORRHOIDAL- hemorrhoidal suppositories suppository
- NDC Code(s): 57483-410-12, 57483-410-13
- Packager: Innovus Pharmaceuticals, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 3, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Uses
- Warnings
- Ask a doctor or pharmacist before use if you are
- Ask a doctor before use if you have
- When using this product
- Stop use and ask a doctor if
- If pregnant or breastfeeding
- Keep out of reach of children
-
Directions
-Adults: when practical, cleanse the affected area by patting or blotting with an appropriate cleansing wipe. Gently dry by patting or blotting with a toilet tissue or a soft cloth.
-remove the wrapper before inserting into the rectum.
-insert one suppository into the rectum up to 4 times daily, especially at night, in the morning or after each bowel movement.
-Children under 12 years of age: consult a doctor
- Other information
- Inactive ingredients
- Purpose
- Carton Label
-
INGREDIENTS AND APPEARANCE
XYRALID HEMORRHOIDAL
hemorrhoidal suppositories suppositoryProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57483-410 Route of Administration RECTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength COCOA BUTTER (UNII: 512OYT1CRR) (COCOA BUTTER - UNII:512OYT1CRR) COCOA BUTTER 88.5 g in 100 g PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 0.26 g in 100 g Inactive Ingredients Ingredient Name Strength METHYLPARABEN (UNII: A2I8C7HI9T) 0.02 g in 100 g PROPYLPARABEN (UNII: Z8IX2SC1OH) 0.02 g in 100 g MODIFIED CORN STARCH (1-OCTENYL SUCCINIC ANHYDRIDE) (UNII: 461P5CJN6T) 11.2 g in 100 g Product Characteristics Color white Score Shape BULLET Size 32mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57483-410-13 4 in 1 CARTON 03/25/2018 1 NDC:57483-410-12 24 g in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part346 03/25/2018 Labeler - Innovus Pharmaceuticals, Inc. (962507187) Establishment Name Address ID/FEI Business Operations Unipack, Inc. 009248480 manufacture(57483-410)