Label: TRIGELS-F FORTE- folic acid, ascorbic acid, cyanocobalamin, and ferrous fumarate capsule
- NHRIC Code(s): 13811-518-10
- Packager: Trigen Laboratories, LLC
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated November 24, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
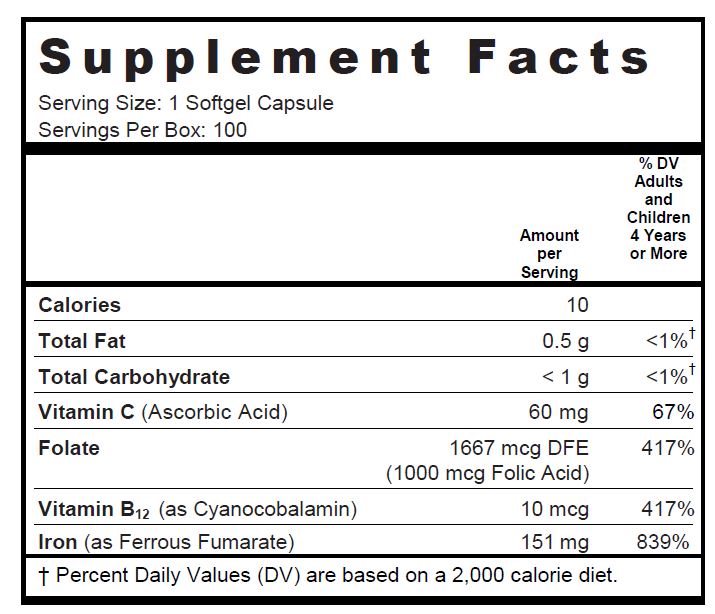
Other Ingredients: Soybean Oil, Gelatin, Glycerin, Purified Water, Yellow Beeswax, Soy Lecithin, Ethyl Vanillin, Titanium Dioxide, FD&C Red #40, FD&C Yellow #5, FD&C Blue #1
THIS PRODUCT CONTAINS SOY.
Trigels-F Forte is a prescription vitamin used for the dietary management of patients with nutritional deficiencies or are in need of nutritional supplementation. - CONTRAINDICATIONS
- WARNINGS
-
PRECAUTIONS
Do not exceed recommended dose. The type of anemia and the underlying cause or causes should be determined before starting therapy with Trigels-F Forte. Since the anemia may be a result of a systemic disturbance, such as recurrent blood loss, the underlying cause or causes should be corrected, if possible.
Folic Acid: Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where vitamin B12 is deficient. Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurologic manifestations remain progressive.
The patient’s medical conditions and consumption of other drugs, herbs, and/or supplements should be considered.
This product contains FD&C Yellow No. 5 (tartrazine) which may cause allergic-type reactions (including bronchial asthma) in certain susceptible patients. Although the overall incidence of FD&C Yellow No. 5 (tartrazine) sensitivity in the general population is low, it is frequently seen in patients who also have aspirin hypersensitivity.
-
ADVERSE REACTIONS
Allergic sensitization has been reported following both oral and parenteral administration of folic acid, as well as possibly the use of other forms of folates, including reduced folates. Mild transient diarrhea, polycythemia vera, itching, transitory exanthema and the feeling of swelling of the entire body have been associated with cyanocobalamin.
-
OVERDOSAGE
The clinical course of acute iron overdosage can be variable. Initial symptoms may include abdominal pain, nausea, vomiting, diarrhea, tarry stools melena, hematemesis, hypotension, tachycardia, metabolic acidosis, hyperglycemia, dehydration, drowsiness, pallor, cyanosis, lassitude, seizures, shock and coma.
- DESCRIPTION
- DIRECTIONS FOR USE
- HOW SUPPLIED
- STORAGE
-
HEALTH CLAIM
KEEP THIS PRODUCT OUT OF REACH OF CHILDREN.
For use on the order of a healthcare practitioner
Call your doctor about side effects. To report side effects, call Trigen Laboratories, LLC at 1-770-509-4500 or FDA at
1-800-FDA-1088 or www.fda.gov/medwatch.
PLR-TRIGELSF-00001-1 Rev. 11/2021
Manufactured for:
Trigen Laboratories, LLC
Alpharetta, GA 30005
www.trigenlab.com
- PRINCIPAL DISPLAY PANEL - 100 Softgel Blister Pack Carton
-
INGREDIENTS AND APPEARANCE
TRIGELS-F FORTE
folic acid, ascorbic acid, cyanocobalamin, and ferrous fumarate capsuleProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:13811-518 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Folic Acid (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) Folic Acid 1 mg Ascorbic Acid (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) Ascorbic Acid 60 mg Cyanocobalamin (UNII: P6YC3EG204) (CYANOCOBALAMIN - UNII:P6YC3EG204) Cyanocobalamin 10 ug Ferrous Fumarate (UNII: R5L488RY0Q) (FERROUS CATION - UNII:GW89581OWR) FERROUS CATION 151 mg Inactive Ingredients Ingredient Name Strength soybean oil (UNII: 241ATL177A) gelatin (UNII: 2G86QN327L) glycerin (UNII: PDC6A3C0OX) water (UNII: 059QF0KO0R) yellow wax (UNII: 2ZA36H0S2V) lecithin, soybean (UNII: 1DI56QDM62) ethyl vanillin (UNII: YC9ST449YJ) titanium dioxide (UNII: 15FIX9V2JP) FD&C Red No. 40 (UNII: WZB9127XOA) FD&C Yellow No. 5 (UNII: I753WB2F1M) FD&C Blue No. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:13811-518-10 10 in 1 CARTON 1 100 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 08/01/2010 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color imprint scoring 1 shape size (solid drugs) 20 mm Labeler - Trigen Laboratories, LLC (830479668)


