HYVEE BRILLIANT WHITE- fluoride paste, dentifrice
HyVee Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
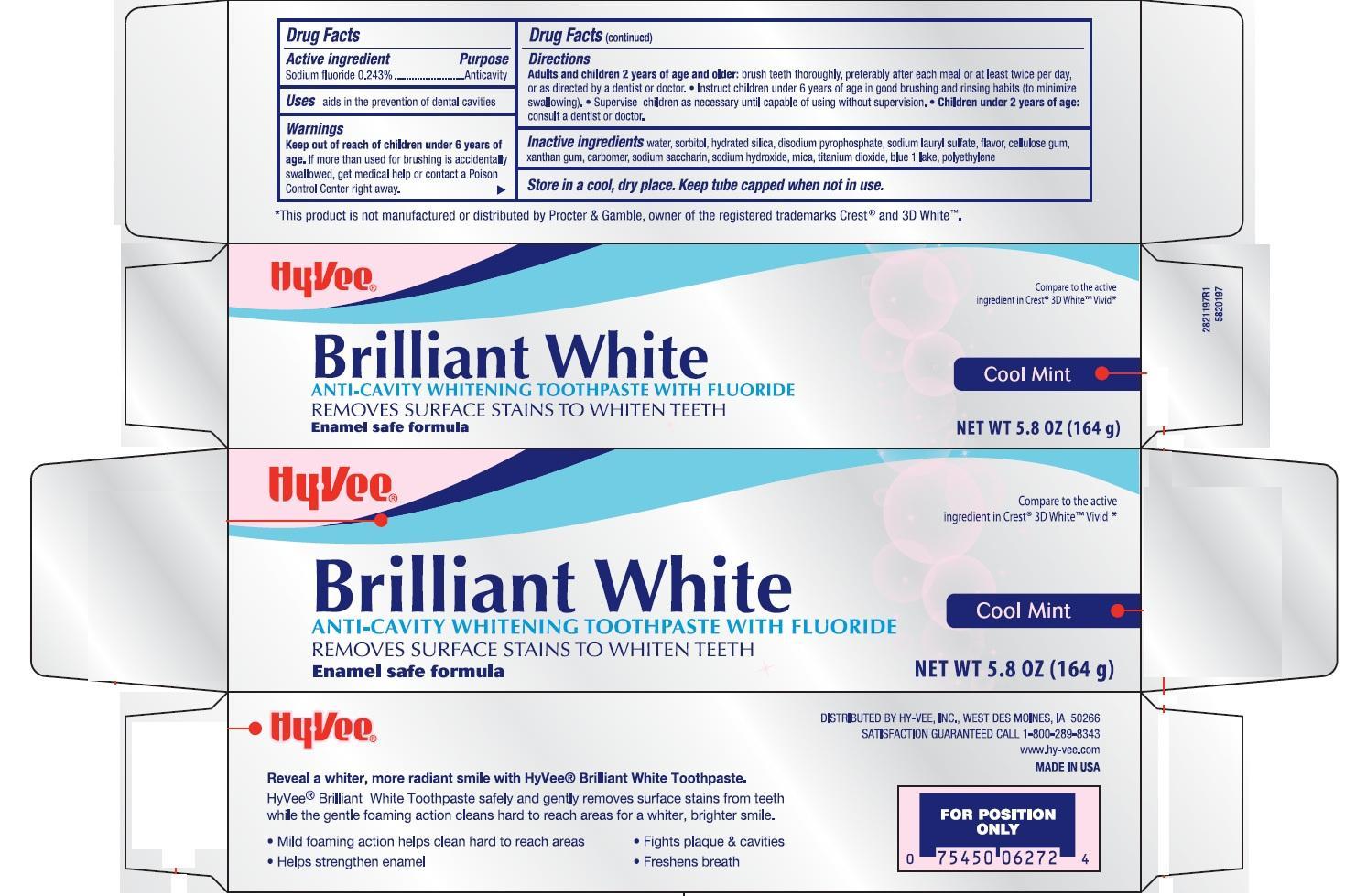
Active ingredients / Purpose
Sodium fluoride 0.243% ............Anticavity
Uses
- aids in the prevention of dental cavities
Children under 2 years of age, consult a dentist or doctor
Keep out of reach of children. If more than used for brushing is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Directions
Adults and children 2 years of age and older: brush teeth thorughly preferably after each meal or at least twice a day, or as directed by a dentist or doctor. Instruct children under 6 years of age in good brushing and rinsing habits (to avoid swallowing). Supervise children as necessary until capable of using without supervision.
Apply at least a 1-inch strip of the product onto a soft bristle toothbrush.
Children under 12 years of age: consult a dentist or a doctor.
Other Information
Store in a cool dry place. Keep tube capped when not in use.
Inactive ingredients
water, sorbitol, hydrated silica, disodium pyrophosphate, sodium lauryl sulfate, flavor, cellulose gum, xanthan gum, carbomer, sodium saccharin, sodium hydroxide, mica, titanium dioxide, blue 1 lake, polyethylene
mm1.jpg
mm2.jpg


