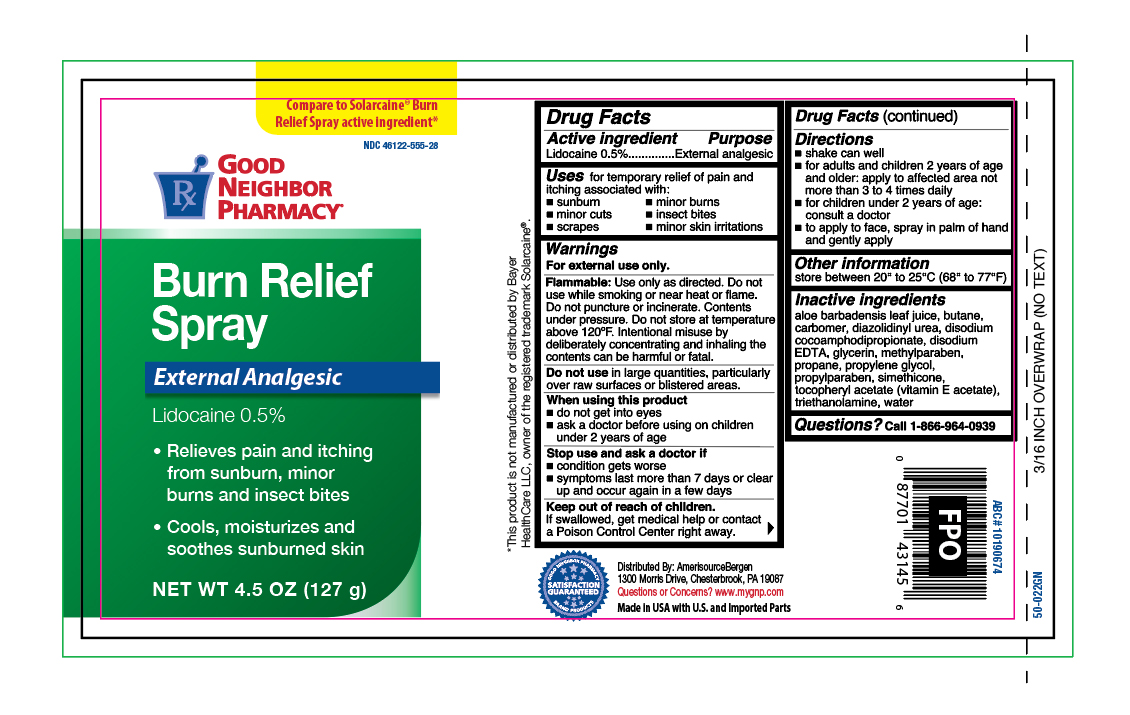
Label: BURN RELIEF- lidocaine aerosol, spray
- NDC Code(s): 46122-555-28
- Packager: AmerisourceBergen Drug Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 26, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
-
Warnings
For external use only.
Flammable:
Do not use while smoking or near heat or flame. Do not puncture or incinerate. Contents under pressure. Do not store at temperature above 120ºF. Intentional misuse by delibrately concentrating and inhaling the contents can be harmful or fatal.
When using this product
- do not get into eyes
- ask a doctor before using on children under 2 years of age
- Directions
- Other information
- Inactive ingredients
- Questions?
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
BURN RELIEF
lidocaine aerosol, sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:46122-555 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 0.64 g in 127 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) BUTANE (UNII: 6LV4FOR43R) CARBOMER HOMOPOLYMER TYPE C (UNII: 4Q93RCW27E) PROPYLPARABEN (UNII: Z8IX2SC1OH) DISODIUM COCOAMPHODIPROPIONATE (UNII: 6K8PRP397M) METHYLPARABEN (UNII: A2I8C7HI9T) WATER (UNII: 059QF0KO0R) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) EDETATE DISODIUM (UNII: 7FLD91C86K) PROPANE (UNII: T75W9911L6) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) TROLAMINE (UNII: 9O3K93S3TK) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:46122-555-28 127 g in 1 CAN; Type 0: Not a Combination Product 10/25/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 10/25/2018 Labeler - AmerisourceBergen Drug Corporation (007914906)