MOTION SICKNESS RELIEF- dimenhydrinate tablet
Cardinal Health 110, LLC. DBA Leader
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Leader 44-198
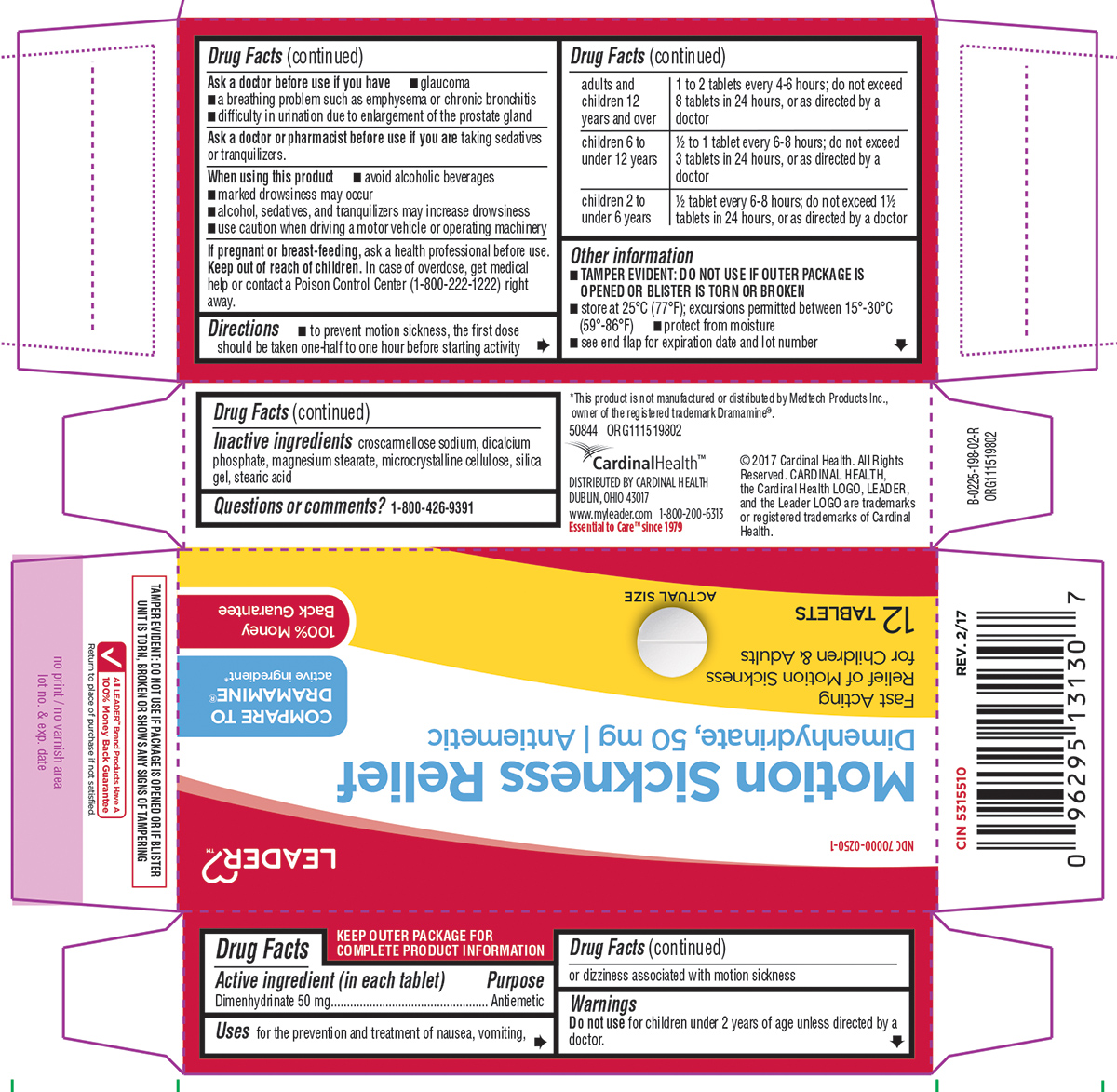
Uses
for the prevention and treatment of nausea, vomiting, or dizziness associated with motion sickness
Warnings
Ask a doctor before use if you have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- difficulty in urination due to enlargement of the prostate gland
Directions
- to prevent motion sickness, the first dose should be taken one-half to one hour before starting activity
| adults and children 12 years and over | 1 to 2 tablets every 4-6 hours; do not exceed 8 tablets in 24 hours, or as directed by a doctor |
| children 6 to under 12 years | 1/2 to 1 tablet every 6-8 hours; do not exceed 3 tablets in 24 hours, or as directed by a doctor |
| children 2 to under 6 years | 1/2 tablet every 6-8 hours; do not exceed 1 1/2 tablets in 24 hours, or as directed by a doctor |
Other information
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- protect from moisture
- see end flap for expiration date and lot number
Inactive ingredients
croscarmellose sodium, dicalcium phosphate, magnesium stearate, microcrystalline cellulose, silica gel, stearic acid
Principal Display Panel
NDC 70000-0250-1
Motion Sickness Relief
Dimenhydrinate, 50 mg | Antiemetic
Fast acting
Relief of Motion Sickness
for Children & Adults
12 TABLETS
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
All LEADER™ Brand Products Have A
100% Money Back Guarantee
Return to place of purchase if not satisfied.
*This product is not manufactured or distributed by Medtech Products Inc., owner of the registered trademark Dramamine®.
50844 ORG111519802
DISTRIBUTED BY CARDINAL HEALTH
DUBLIN, OHIO 43017
www.myleader.com 1-800-200-6313
Essential to Care™ since 1979
©2017 Cardinal Health. All Rights Reserved. CARDINAL HEALTH, the Cardinal Health LOGO, LEADER, and the Leader LOGO are trademarks or registered trademarks of Cardinal Health.
Leader 44-198
| MOTION SICKNESS RELIEF
dimenhydrinate tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Cardinal Health 110, LLC. DBA Leader (063997360) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 038154464 | pack(70000-0250) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867837 | pack(70000-0250) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 832867894 | manufacture(70000-0250) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 868734088 | pack(70000-0250) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| LNK International, Inc. | 967626305 | pack(70000-0250) | |