SEI BELLA AGE-DEFYING LIQUID FOUNDATION BEIGE- octinoxate 7.5% lotion
Melaleuca Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Sei Bella Age-Defying Liquid Foundation - Beige
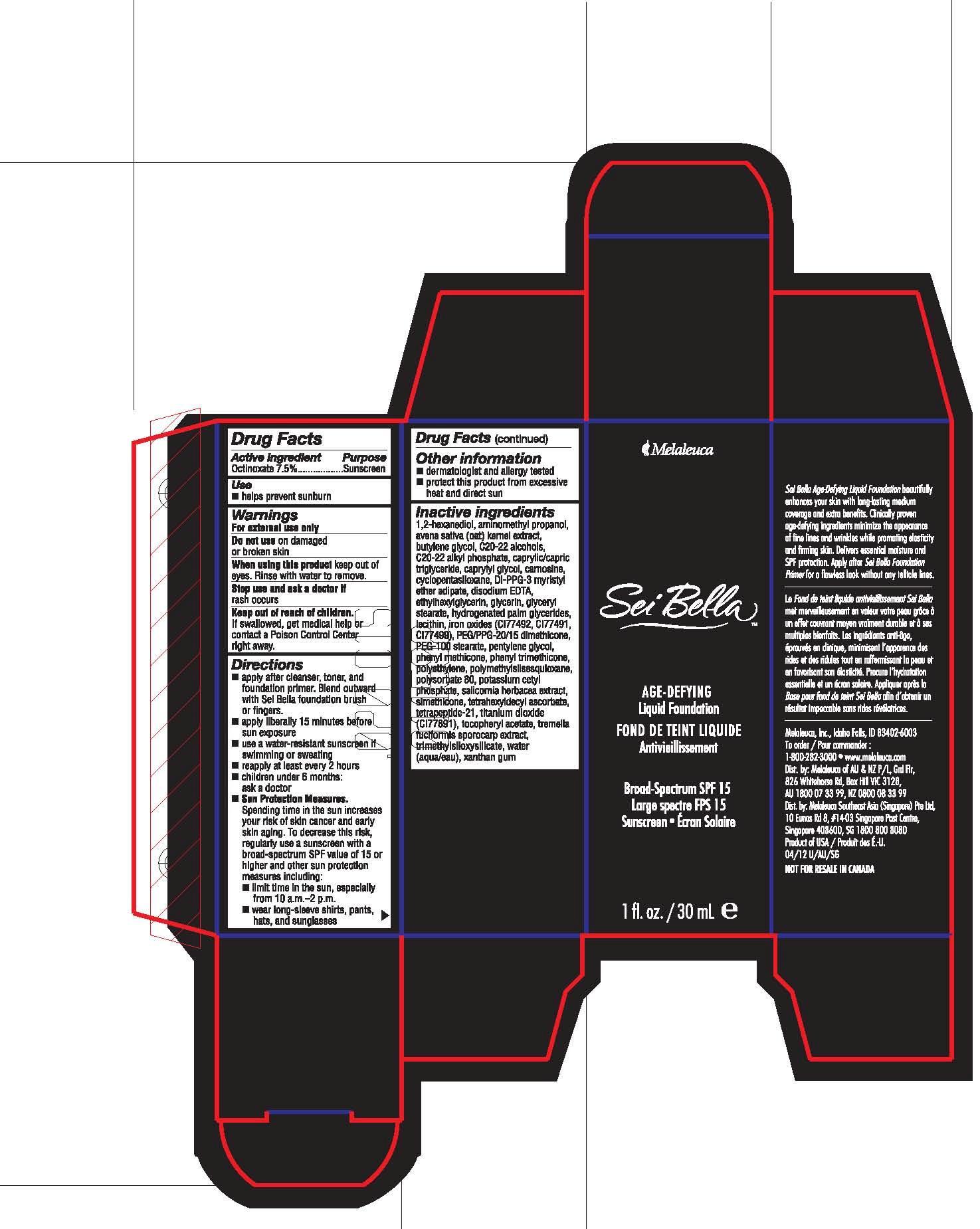
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
■ apply after cleanser, toner, and foundation primer. Blend outward with Sei Bella foundation brush or fingers.
■ apply liberally 15 minutes before sun exposure
■ use a water-resistant sunscreen if swimming or sweating
■ reapply at least every 2 hours
■ children under 6 months: ask a doctor
■ Sun Protection Measures. Spending time in the sun increases you risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad-spectrum SPF value of 15 or higher and other sun protection measures including:
■ limit time in the sun, especially from 10 a.m.-2 p.m.
■ wear long-sleeve shirts, pants, hats, and sunglasses
Inactive ingredients
1,2-hexanediol, aminomethyl propanol, avena sativa (oat) kernel extract, butylene glycol, C20-22 alcohols, C20-22 alkyl phosphate, caprylic/capric triglyceride, caprylyl glycol, carnosine, cyclopentasiloxane, DI-PPG-3 myristyl ether adipate, disodium EDTA, ethylhexylglycerin, glycerin, glyceryl stearate, hydrogenated palm glycerides, lecithin, iron oxides (CI77492, CI77491, CI77499), PEG/PPG-20/15 dimethicone, PEG-100 stearate, pentylene glycol, phenyl methicone, phenyl trimethicone, polyethylene, polymethylsilsesquioxane, polysorbate 80, potassium cetyl phosphate, salicornia herbacea extract, simethicone, tetrahexyldecyl ascorbate, tetrapeptide-21, titanium dioxide (CI77891), tocopheryl acetate, tremella fuciformis sporocarp extract, trimethylsiloxysilicate, water (aqua/eau), xanthan gum
| SEI BELLA AGE-DEFYING LIQUID FOUNDATION
BEIGE
octinoxate 7.5% lotion |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Melaleuca Inc. (139760102) |
| Registrant - Melaleuca Inc. (139760102) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Melaleuca Inc. | 079711683 | manufacture(54473-228) | |