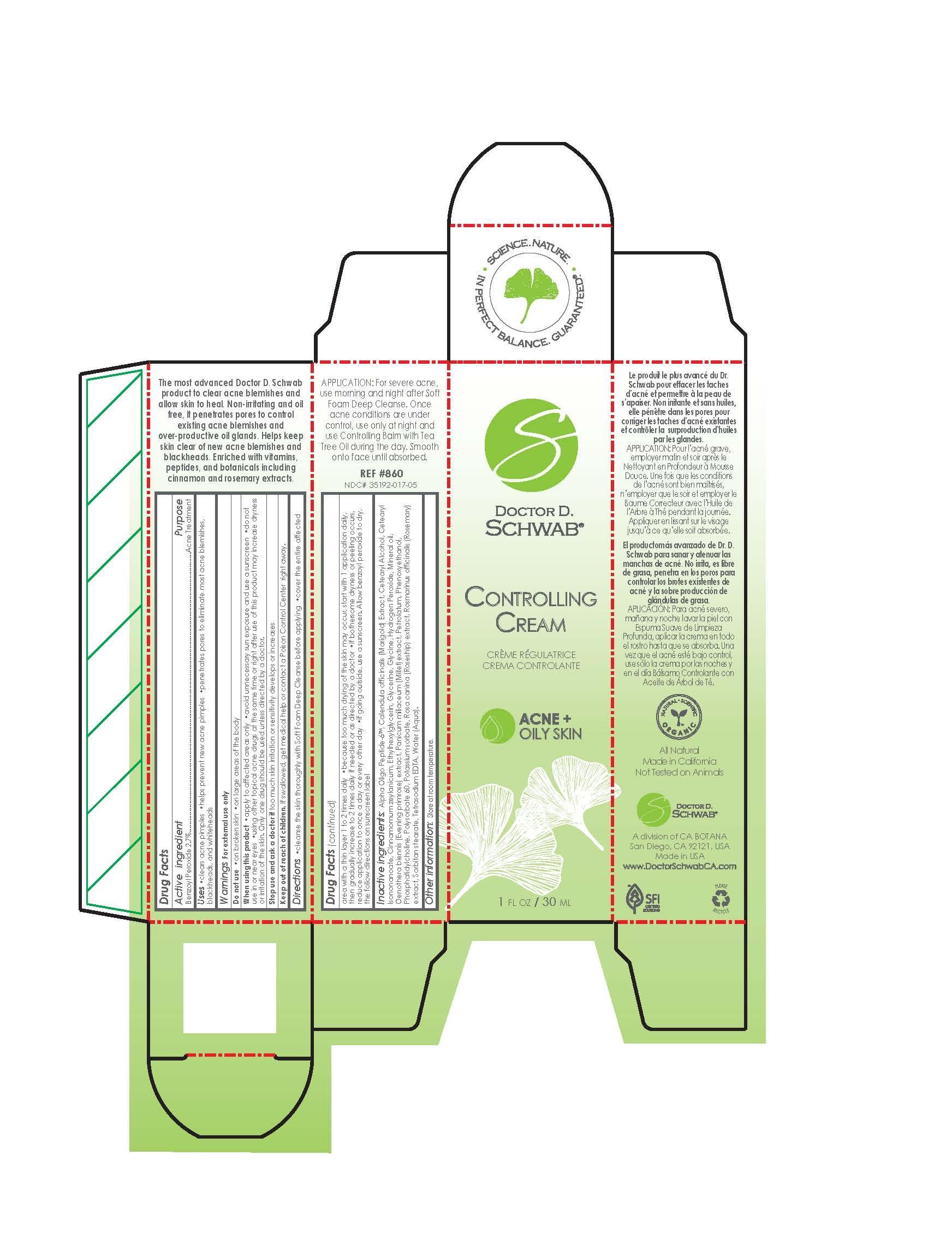
Label: CONTROLLING CREAM- benzoyl peroxide cream
- NDC Code(s): 35192-017-05
- Packager: CA-BOTANA INTERNATIONAL
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
WARNINGS
Warnings:
For external use only.
Do not use on wounds or damaged skin
When using this product: use only as directed. Avoid contact with eyes. Do no bandage tightly
Stop use and ask a doctor if: redness is present. Irritation develops. Condition worsens or symptoms persist more than 7 days. Symptoms clear up and occur again within a few days.
Store at room temperature. Lot number and expiration date see crimp or see box. -
DOSAGE & ADMINISTRATION
cleanse the skin thoroughly with soft foam deep cleanse before applying. cover the entire affected aread with a thin layer 1 to 2 times daily. because too much drying of the skin may occure, start with 1 application daily, then gradually increase to 2 times daily if needed or as directed by a doctor. if bothersome dryness or peeling occurs, reduce application to once a day or every other day. Allow benzoyl peroxide to dry the follow directions on sunscreen label.
-
INACTIVE INGREDIENT
Alpha Oligo Peptide-6 TM Calendula officinalis (Marigold) Extract Cetearyl Alcohol Cetearyl Isononanoate Cynamomum Zeylanicum Ethylhexylglycerin Glycerine Glycine Hydrogen Peroxide Mineral oil Oenothera biennis (Evening Primrose) extract Panicum Miliaceum (Millet)extract Petrolatum Phenoxyethanol Phosphatidycholine Polysorbate 60 Potassium Sorbate Rosa canina (Roseship) extract Rosmarinus officinalis (Rosemary) extract Sorbitan Stearate Tetra Sodium Water (Aqua) - ACTIVE INGREDIENT
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- PURPOSE
- PRINCIPAL DISPLAY PANEL
- Package label display panel
-
INGREDIENTS AND APPEARANCE
CONTROLLING CREAM
benzoyl peroxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:35192-017 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOYL PEROXIDE (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) BENZOYL PEROXIDE 0.8 mg in 29.6 mg Inactive Ingredients Ingredient Name Strength CINNAMON (UNII: 5S29HWU6QB) CINNAMON LEAF OIL (UNII: S92U8SQ71V) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLYCERIN (UNII: PDC6A3C0OX) GLYCINE (UNII: TE7660XO1C) HYDROGEN PEROXIDE (UNII: BBX060AN9V) LIGHT MINERAL OIL (UNII: N6K5787QVP) OENOTHERA BIENNIS (UNII: 76UI55V071) MILLET (UNII: TJR6B3R47P) PETROLATUM (UNII: 4T6H12BN9U) PHENOXYETHANOL (UNII: HIE492ZZ3T) HYDROGENATED SOYBEAN LECITHIN (UNII: H1109Z9J4N) POLYSORBATE 60 (UNII: CAL22UVI4M) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) ROSA CANINA LEAF (UNII: J3N2Z889QP) ROSEMARY (UNII: IJ67X351P9) SORBITAN MONOSTEARATE (UNII: NVZ4I0H58X) EDETATE SODIUM (UNII: MP1J8420LU) WATER (UNII: 059QF0KO0R) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) CETEARYL ISONONANOATE (UNII: P5O01U99NI) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:35192-017-05 29.6 mg in 1 TUBE; Type 0: Not a Combination Product 11/02/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 11/09/2014 Labeler - CA-BOTANA INTERNATIONAL (106276728) Registrant - RODOLFO UGELSTAD (106276728) Establishment Name Address ID/FEI Business Operations CA-BOTANA INTERNATIONAL 106276728 manufacture(35192-017)