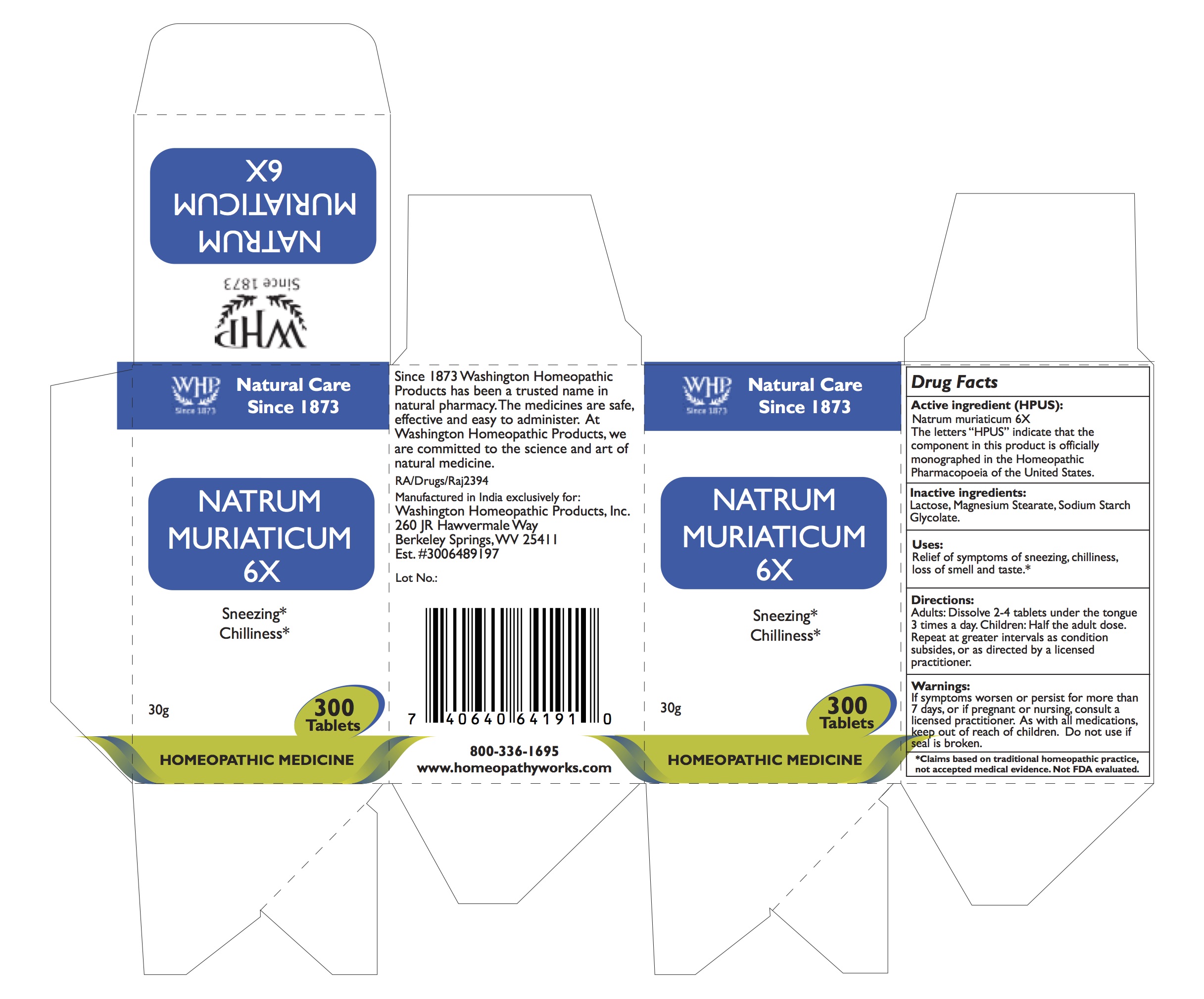
NATRUM MURIATICUM- sodium chloride tablet
Washington Homeopathic Products
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
NATRUM MURIATICUM X
ACTIVE INGREDIENTS
Active ingredient (HPUS):
Natrum muriaticum 6X
The letters “HPUS” indicate that the component in this product is officially monographed in the Homeopathic Pharmacopoeia of the United States.
INDICATIONS
Indications:
Relief of symptoms of sneezing, chilliness, loss of smell and taste.*
*Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
STOP USE AND ASK DOCTOR
If symptoms worsen or persist for more than 7 days, or if pregnant or nursing, consult a licensed practitioner.
| NATRUM MURIATICUM
sodium chloride tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Washington Homeopathic Products (084929389) |
| Registrant - Washington Homeopathic Products (084929389) |