TRIPROLIDINE HYDROCHLORIDE- triprolidine hydrochloride liquid
Strides Pharma Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Triprolidine HCl
Uses
- temporarily relieves:
- runny nose
- sneezing
- itching of nose and throat
- itchy, watery eyes due to hay fever or other upper respiratory allergies or allergic rhinitis
Do Not Use
- if you or your children are taking sedatives or tranquilizers, without first consulting your doctor
- with any other product containing triprolidine
Ask a doctor before use if your child has
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- difficulty in urination due to enlargement of the prostate gland
When using this product
- do not use more than directed
- may cause drowsiness
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
Stop use and ask a doctor if:
- nervousness, dizziness or sleeplessness occurs
- symptoms do not improve within 7 days or are accompanied by a fever
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- take every 6 hours
- do not take more than 4 doses in 24 hours or as directed by a doctor.
- do not give more than directed or exceed the recommended daily dosage
- use only enclosed dropper
- do not use enclosed dropper for any other drug product
| Adults and children 12 years of age and over | 4 mL (2.5 mg) |
| Children 6 to under 12 years of age | 2 mL (1.25 mg) |
| Children under 6 years of age | ask a doctor |
Other Information
- store at controlled room temperature 15°-30°C (59°-86°F)
- Tamper Evident: Do not use if seal is broken or missing.
Inactive Ingredients
Bubble Gum Flavor, Citric Acid, Glycerin, Propylene Glycol, Purified Water, Sodium Benzoate, Sodium Citrate, Sodium Saccharin, Sorbitol.
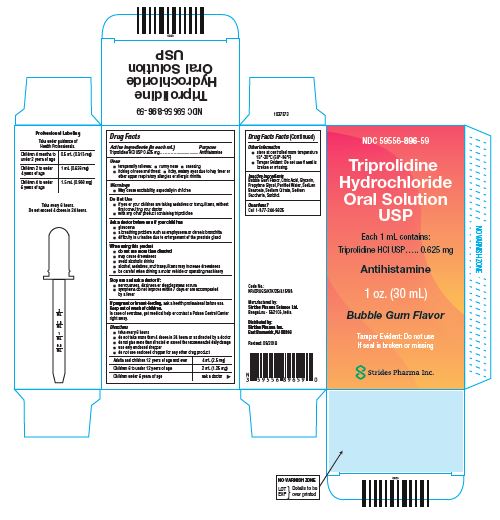
PRINCIPAL DISPLAY PANEL - 30 mL Bottle Carton
Triprolidine Hydrochloride Oral Solution USP
Each 1 mL contains:
Triprolidine HCl USP….. 0.625 mg
Antihistamine
1 oz. (30 mL)
Bubble Gum Flavor
Tamper Evident: Do not use If seal is broken or missing.
Strides Pharma Inc.
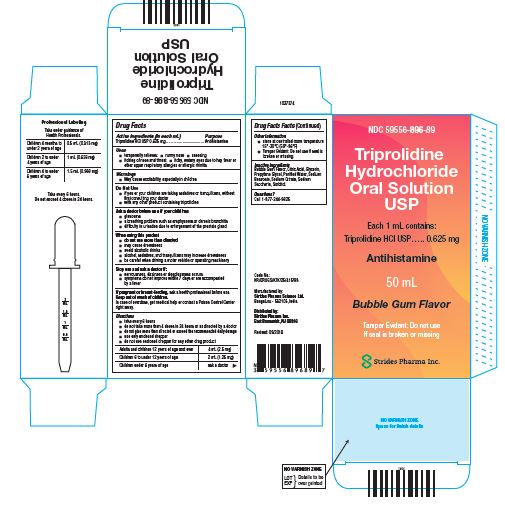
PRINCIPAL DISPLAY PANEL - 50 mL Bottle Carton
NDC 59556-896-89
Triprolidine Hydrochloride Oral Solution USP
Each 1 mL contains:
Triprolidine HCl USP….. 0.625 mg
Antihistamine
50 mL
Bubble Gum Flavor
Tamper Evident: Do not use If seal is broken or missing.
Strides Pharma Inc.
| TRIPROLIDINE HYDROCHLORIDE
triprolidine hydrochloride liquid |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Strides Pharma Inc (078868278) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Strides Pharma Science Limited | 918513263 | ANALYSIS(59556-896) , MANUFACTURE(59556-896) , PACK(59556-896) | |