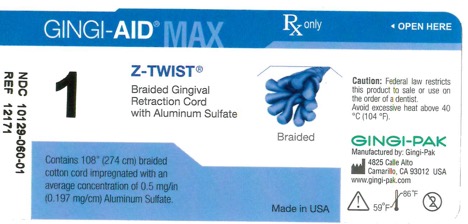
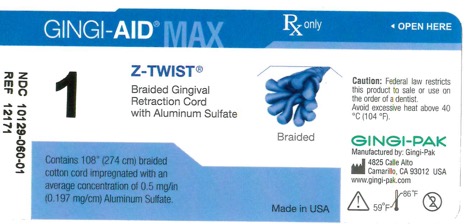
Label: GINGI-AID MAX Z-TWIST 1 solution
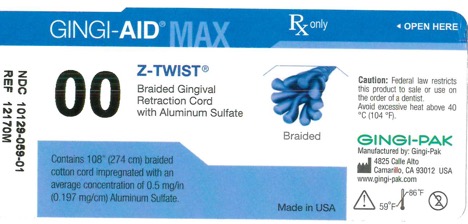
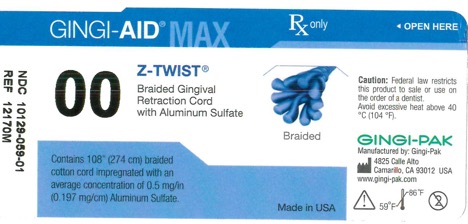
GINGI-AID MAX Z-TWIST 00 solution
GINGI-AID MAX Z-TWIST 3 solution
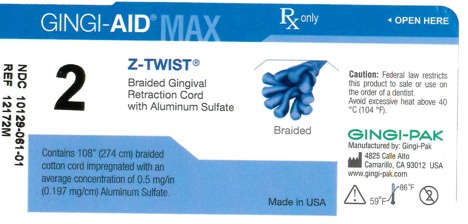
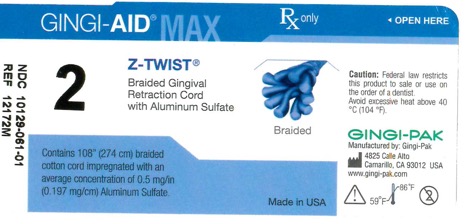
GINGI-AID MAX Z-TWIST 2 solution
- NDC Code(s): 10129-059-01, 10129-060-01, 10129-061-01, 10129-062-03
- Packager: Gingi-Pak a Division of the Belport
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 24, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Precautions
- Warnings
- Contraindications
- Purpose
- Keep out of reach of children
- Indication and Uses
- Avoid excessive heat
- Directions for use
- Contents
- Gingi-Aid Max Z-twist 00
- Gingi-Aid Max Z-twist 1
- Gingi-Aid Max Z-twist 2
- Gingi-Aid Max Z-twist 3
-
INGREDIENTS AND APPEARANCE
GINGI-AID MAX Z-TWIST 1
gingi-aid max z-twist 1 solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10129-060 Route of Administration DENTAL, PERIODONTAL, SUBGINGIVAL, ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM SULFATE (UNII: 34S289N54E) (ALUMINUM SULFATE ANHYDROUS - UNII:I7T908772F) ALUMINUM SULFATE 54 mg Inactive Ingredients Ingredient Name Strength COTTON FIBER (UNII: 70LDW53ROO) 2740 mm Product Characteristics Color blue (Teal) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10129-060-01 1 in 1 VIAL, PLASTIC; Type 0: Not a Combination Product 01/15/1995 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 01/13/1995 GINGI-AID MAX Z-TWIST 00
gingi-aid max z-twist 00 solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10129-059 Route of Administration ORAL, DENTAL, PERIODONTAL, SUBGINGIVAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM SULFATE (UNII: 34S289N54E) (ALUMINUM SULFATE ANHYDROUS - UNII:I7T908772F) ALUMINUM SULFATE 54 mg Inactive Ingredients Ingredient Name Strength COTTON FIBER (UNII: 70LDW53ROO) 2740 mm Product Characteristics Color blue (Teal) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10129-059-01 1 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 01/15/1995 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 01/13/1995 GINGI-AID MAX Z-TWIST 3
gingi-aid max z-twist 3 solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10129-062 Route of Administration ORAL, DENTAL, PERIODONTAL, SUBGINGIVAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM SULFATE (UNII: 34S289N54E) (ALUMINUM SULFATE ANHYDROUS - UNII:I7T908772F) ALUMINUM SULFATE 54 mg Inactive Ingredients Ingredient Name Strength COTTON FIBER (UNII: 70LDW53ROO) 2740 mm Product Characteristics Color blue (Teal) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10129-062-03 1 in 1 VIAL, PLASTIC; Type 0: Not a Combination Product 07/31/1998 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 07/31/1998 GINGI-AID MAX Z-TWIST 2
gingi-aid max z-twist 2 solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10129-061 Route of Administration ORAL, DENTAL, PERIODONTAL, SUBGINGIVAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM SULFATE (UNII: 34S289N54E) (ALUMINUM SULFATE ANHYDROUS - UNII:I7T908772F) ALUMINUM SULFATE 54 mg Inactive Ingredients Ingredient Name Strength COTTON FIBER (UNII: 70LDW53ROO) 2740 mm Product Characteristics Color blue (Teal) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10129-061-01 1 in 1 VIAL, PLASTIC; Type 0: Not a Combination Product 01/15/1995 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 01/13/1995 Labeler - Gingi-Pak a Division of the Belport (008480121)