ISTODAX- romidepsin
Gloucester Pharmaceuticals, Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use ISTODAX safely and effectively. See full prescribing information for ISTODAX.
ISTODAX® (romidepsin) for injection For intravenous infusion only Initial U.S. Approval: 2009 INDICATIONS AND USAGEISTODAX is a histone deacetylase (HDAC) inhibitor indicated for:
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSISTODAX for injection, 10 mg, supplied with one Diluent vial containing 2 mL (deliverable volume) of solution (3) CONTRAINDICATIONSNone. (4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common adverse reactions in Study 1 were nausea, fatigue, infections, vomiting, and anorexia, and in Study 2 were nausea, fatigue, anemia, thrombocytopenia, ECG T-wave changes, neutropenia, and lymphopenia (6). To report SUSPECTED ADVERSE REACTIONS, contact Gloucester Pharmaceuticals, Inc. at 1-866-223-7145 or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 11/2009 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
ISTODAX is indicated for treatment of cutaneous T-cell lymphoma (CTCL) in patients who have received at least one prior systemic therapy.
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Information
The recommended dose of romidepsin is 14 mg/m2 administered intravenously over a 4-hour period on days 1, 8 and 15 of a 28-day cycle. Cycles should be repeated every 28 days provided that the patient continues to benefit from and tolerates the therapy.
2.2 Dose Modification
Nonhematologic toxicities except alopecia
- Grade 2 or 3 toxicity: Treatment with romidepsin should be delayed until toxicity returns to ≤Grade 1 or baseline, then therapy may be restarted at 14 mg/m2. If Grade 3 toxicity recurs, treatment with romidepsin should be delayed until toxicity returns to ≤Grade 1 or baseline and the dose should be permanently reduced to 10 mg/m2.
- Grade 4 toxicity: Treatment with romidepsin should be delayed until toxicity returns to ≤Grade 1 or baseline, then the dose should be permanently reduced to 10 mg/m2.
- Romidepsin should be discontinued if Grade 3 or 4 toxicities recur after dose reduction.
Hematologic toxicities
- Grade 3 or 4 neutropenia or thrombocytopenia: Treatment with romidepsin should be delayed until the specific cytopenia returns to ANC ≥1.5×109/L and/or platelet count ≥75×109/L or baseline, then therapy may be restarted at 14 mg/m2.
- Grade 4 febrile (≥38.5ºC) neutropenia or thrombocytopenia that requires platelet transfusion: Treatment with romidepsin should be delayed until the specific cytopenia returns to ≤Grade 1 or baseline, and then the dose should be permanently reduced to 10 mg/m2.
2.3 Instructions for Preparation and Intravenous Administration
ISTODAX should be handled in a manner consistent with recommended safe procedures for handling cytotoxic drugs.
ISTODAX must be reconstituted with the supplied diluent and further diluted with 0.9% Sodium Chloride Injection, USP before intravenous infusion.
- Each 10 mg single-use vial of ISTODAX (romidepsin) must be reconstituted with 2 mL of the supplied Diluent. With a suitable syringe, aseptically withdraw 2 mL from the supplied Diluent vial, and slowly inject it into the ISTODAX (romidepsin) for injection vial. Swirl the contents of the vial until there are no visible particles in the resulting solution. The reconstituted solution will contain ISTODAX 5 mg/mL. The reconstituted ISTODAX solution is chemically stable for at least 8 hours at room temperature.
- Extract the appropriate amount of ISTODAX from the vials to deliver the desired dose, using proper aseptic technique. Before intravenous infusion, further dilute ISTODAX in 500 mL 0.9% Sodium Chloride Injection, USP.
- Infuse over 4 hours.
The diluted solution is compatible with polyvinyl chloride (PVC), ethylene vinyl acetate (EVA), polyethylene (PE) infusion bags as well as glass bottles, and is chemically stable for at least 24 hours when stored at room temperature. However, it should be administered as soon after dilution as possible.
Parenteral drug products should be inspected visually for particulate matter and discoloration before administration, whenever solution and container permit.
3 DOSAGE FORMS AND STRENGTHS
ISTODAX is supplied as a kit which includes a sterile, lyophilized powder in a single-use vial containing 10 mg of romidepsin and 20 mg of the bulking agent, povidone, USP. In addition, each kit includes 1 sterile vial containing 2 mL (deliverable volume) of the Diluent composed of 80% propylene glycol, USP, and 20% dehydrated alcohol, USP.
5 WARNINGS AND PRECAUTIONS
5.1 Monitoring: Laboratory Tests
Due to the risk of QT prolongation, potassium and magnesium should be within the normal range before administration of ISTODAX [See Warnings and Precautions (5.3) and Adverse Reactions (6)].
5.2 Hematologic
Treatment with ISTODAX can cause thrombocytopenia, leukopenia (neutropenia and lymphopenia), and anemia; therefore, these hematological parameters should be monitored during treatment with ISTODAX, and the dose should be modified, as necessary [See Dosage and Administration (2.2) and Adverse Reactions (6)].
5.3 Electrocardiographic Changes
Several treatment-emergent morphological changes in ECGs (including T-wave and ST-segment changes) have been reported in clinical studies. The clinical significance of these changes is unknown [See Adverse Reactions (6)].
In patients with congenital long QT syndrome, patients with a history of significant cardiovascular disease, and patients taking anti-arrhythmic medicines or medicinal products that lead to significant QT prolongation, appropriate cardiovascular monitoring precautions should be considered, such as the monitoring of electrolytes and ECGs at baseline and periodically during treatment.
5.4 Use in Pregnancy
There are no adequate and well-controlled studies of ISTODAX in pregnant women. However, based on its mechanism of action, ISTODAX may cause fetal harm when administered to a pregnant woman. A study in rats did not expose pregnant animals to enough romidepsin to fully evaluate adverse outcomes.
If this drug is used during pregnancy, or if the patient becomes pregnant while taking ISTODAX, the patient should be apprised of the potential hazard to the fetus [See Use in Specific Populations (8.1)].
5.5 Use in Women of Childbearing Potential
Advise women of childbearing potential that ISTODAX may reduce the effectiveness of estrogen-containing contraceptives. An in vitro binding assay determined that romidepsin competes with β-estradiol for binding to estrogen receptors [See Nonclinical Toxicology (13.1)].
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ISTODAX was evaluated in 185 patients with CTCL in 2 single arm clinical studies in which patents received a starting dose of 14 mg/m2. The mean duration of treatment in these studies was 5.6 months (range: <1 to 83.4 months).
Common Adverse Reactions
Table 1 summarizes the most frequent adverse reactions (> 20%) regardless of causality using the National Cancer Institute-Common Terminology Criteria for Adverse Events (NCI-CTCAE, Version 3.0). Due to methodological differences between the studies, the AE data are presented separately for Study 1 and Study 2. Adverse reactions are ranked by their incidence in Study 1. Laboratory abnormalities commonly reported (>20%) as adverse reactions are included in Table 1.
| Adverse Reactions n (%) | Study 1 (n=102) | Study 2 (n=83) |
||
|---|---|---|---|---|
| All | Grade 3 or 4 | All | Grade 3 or 4 | |
| Any adverse reaction | 99 (97) | 36 (35) | 83 (100) | 68 (82) |
| Nausea | 57 (56) | 3 (3) | 71 (86) | 5 (6) |
| Asthenia/fatigue | 54 (53) | 8 (8) | 64 (77) | 12 (14) |
| Infections | 47 (46) | 11 (11) | 45 (54) | 27 (33) |
| Vomiting | 35 (34) | 1 (<1) | 43 (52) | 8 (10) |
| Anorexia | 23 (23) | 1 (<1) | 45 (54) | 3 (4) |
| Hypomagnesemia | 22 (22) | 1 (<1) | 23 (28) | 0 |
| Diarrhea | 20 (20) | 1 (<1) | 22 (7) | 1 (1) |
| Pyrexia | 20 (20) | 4 (4) | 19 (23) | 1 (1) |
| Anemia | 19 (19) | 3 (3) | 60 (72) | 13 (16) |
| Thrombocytopenia | 17 (17) | 0 | 54 (65) | 12 (14) |
| Dysgeusia | 15 (15) | 0 | 33 (40) | 0 |
| Constipation | 12 (12) | 2 (2) | 32 (39) | 1 (1) |
| Neutropenia | 11 (11) | 4 (4) | 47 (57) | 22 (27) |
| Hypotension | 7 (7) | 3 (3) | 19 (23) | 3 (4) |
| Pruritus | 7 (7) | 0 | 26 (31) | 5 (6) |
| Hypokalemia | 6 (6) | 0 | 17 (20) | 2 (2) |
| Dermatitis/Exfoliative dermatitis | 4 (4) | 1 (<1) | 22 (27) | 7 (8) |
| Hypocalcemia | 4 (4) | 0 | 43 (52) | 5 (6) |
| Leukopenia | 4 (4) | 0 | 38 (46) | 18 (22) |
| Lymphopenia | 4 (4) | 0 | 47 (57) | 31 (37) |
| Alanine aminotransferase increased | 3 (3) | 0 | 18 (22) | 2 (2) |
| Aspartate aminotransferase increased | 3 (3) | 0 | 23 (28) | 3 (4) |
| Hypoalbuminemia | 3 (3) | 1 (<1) | 40 (48) | 3 (4) |
| Electrocardiogram ST-T wave changes | 2 (2) | 0 | 52 (63) | 0 |
| Hyperglycemia | 2 (2) | 2 (2) | 42 (51) | 1 (1) |
| Hyponatremia | 1 (<1) | 1 (<1) | 17 (20) | 2 (2) |
| Hypermagnesemia | 0 | 0 | 22 (27) | 7 (8) |
| Hypophosphatemia | 0 | 0 | 22 (27) | 8 (10) |
| Hyperuricemia | 0 | 0 | 27 (33) | 7 (8) |
Serious Adverse Reactions
Serious adverse reactions reported in > 2% of patients in Study 1 were infection, sepsis, and pyrexia. In Study 2, serious adverse reactions in > 2% of patients were infection, supraventricular arrhythmia, neutropenia, fatigue, edema, central line infection, ventricular arrhythmia, nausea, pyrexia, leukopenia, and thrombocytopenia.
Most deaths were due to disease progression. In Study 1, there were two deaths due to cardiopulmonary failure and acute renal failure. In Study 2, there were six deaths due to infection (4), myocardial ischemia, and acute respiratory distress syndrome.
7 DRUG INTERACTIONS
7.1 Coumadin or Coumadin Derivatives
Prolongation of PT and elevation of INR were observed in a patient receiving ISTODAX concomitantly with warfarin. Although the interaction potential between ISTODAX and Coumadin or Coumadin derivatives has not been formally studied, physicians should carefully monitor PT and INR in patients concurrently administered ISTODAX and Coumadin or Coumadin derivatives [See Clinical Pharmacology (12.3)].
7.2 Drugs that Inhibit or Induce Cytochrome P450 3A4 Enzymes
Romidepsin is metabolized by CYP3A4. Although there are no formal drug interaction studies for ISTODAX, strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, clarithromycin, atazanavir, indinavir, nefazodone, nelfinavir, ritonavir, saquinavir, telithromycin, voriconazole) may increase concentrations of romidepsin. Therefore, co-administration with strong CYP3A4 inhibitors should be avoided if possible. Caution should be exercised with concomitant use of moderate CYP3A4 inhibitors.
Co-administration of potent CYP3A4 inducers (e.g., dexamethasone, carbamazepine, phenytoin, rifampin, rifabutin, rifapentine, phenobarbital) may decrease concentrations of romidepsin and should be avoided if possible. Patients should also refrain from taking St. John's Wort.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category D [See 'Warnings and Precautions' section].
There are no adequate and well-controlled studies of ISTODAX in pregnant women. However, based on its mechanism of action, ISTODAX may cause fetal harm when administered to a pregnant woman. A study in rats did not expose pregnant animals to enough romidepsin to fully evaluate adverse developmental outcomes. If this drug is used during pregnancy, or if the patient becomes pregnant while taking ISTODAX, the patient should be apprised of the potential harm to the fetus.
In an animal reproductive study, pregnant rats received daily intravenous romidepsin during the period of organogenesis up to a dose of 0.06 mg/kg/day (0.36 mg/m2/day). This dose in rats is approximately equivalent to 18% the estimated human daily dose based on body surface area and resulted in 5% reduction in fetal weight. Embryofetal toxicities associated with the use of ISTODAX were not adequately assessed in this study.
8.3 Nursing Mothers
It is not known whether romidepsin is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from ISTODAX, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
The safety and effectiveness of ISTODAX in pediatric patients has not been established.
8.5 Geriatric Use
Of the 167 patients with CTCL in trials, 23% were >65 years old. No overall differences in safety or effectiveness were observed between these subjects and younger subjects; however, greater sensitivity of some older individuals cannot be ruled out.
8.6 Hepatic Impairment
No dedicated hepatic impairment study for ISTODAX has been conducted. Mild hepatic impairment does not alter pharmacokinetics of romidepsin based on a population pharmacokinetic analysis. Patients with moderate and severe hepatic impairment should be treated with caution [See Clinical Pharmacology (12.3)].
8.7 Renal Impairment
No dedicated renal impairment study for ISTODAX has been conducted. Based upon the population pharmacokinetic analysis, renal impairment is not expected to significantly influence drug exposure. The effect of end-stage renal disease on romidepsin pharmacokinetics has not been studied. Thus, patients with end-stage renal disease should be treated with caution [See Clinical Pharmacology (12.3)].
10 OVERDOSAGE
No specific information is available on the treatment of overdosage of ISTODAX.
Toxicities in a single-dose study in rats or dogs, at intravenous romidepsin doses up to 2.2 fold the recommended human dose based on the body surface area, included irregular respiration, irregular heart beat, staggering gait, tremor, and tonic convulsions.
In the event of an overdose, it is reasonable to employ the usual supportive measures, e.g., clinical monitoring and supportive therapy, if required. There is no known antidote for ISTODAX and it is not known if ISTODAX is dialyzable.
11 DESCRIPTION
Romidepsin, a histone deacetylase (HDAC) inhibitor, is a bicyclic depsipeptide. At room temperature, romidepsin is a white powder and is described chemically as (1S,4S,7Z,10S,16E,21R)-7-ethylidene-4,21-bis(1-methylethyl)-2-oxa-12,13-dithia-5,8,20,23-tetraazabicyclo[8.7.6]tricos-16-ene-3,6,9,19,22-pentone. The empirical formula is C24H36N4O6S2.
The molecular weight is 540.71 and the structural formula is:
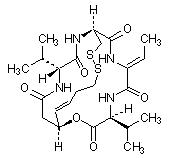
ISTODAX (romidepsin) for injection is intended for intravenous infusion only after reconstitution with the supplied Diluent and after further dilution with 0.9% Sodium Chloride, USP.
ISTODAX is supplied as a kit containing two vials.
ISTODAX (romidepsin) for injection is a sterile lyophilized white powder and is supplied in a single-use vial containing 10 mg romidepsin and 20 mg povidone, USP.
Diluent for ISTODAX is a sterile clear solution and is supplied in a single-use vial containing a 2-mL deliverable volume. Diluent for ISTODAX contains 80% (v/v) propylene glycol, USP and 20% (v/v) dehydrated alcohol, USP.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Romidepsin is a histone deacetylase (HDAC) inhibitor. HDACs catalyze the removal of acetyl groups from acetylated lysine residues in histones, resulting in the modulation of gene expression. HDACs also deacetylate non-histone proteins, such as transcription factors. In vitro, romidepsin causes the accumulation of acetylated histones, and induces cell cycle arrest and apoptosis of some cancer cell lines with IC50 values in the nanomolar range. The mechanism of the antineoplastic effect of romidepsin observed in nonclinical and clinical studies has not been fully characterized.
12.3 Pharmacokinetics
Absorption
Romidepsin exhibited linear pharmacokinetics across doses ranging from 1.0 to 24.9 mg/m2 when administered intravenously over 4 hours in patients with advanced cancers.
In patients with T cell lymphomas who received 14 mg/m2 of romidepsin intravenously over a 4-hour period on days 1, 8 and 15 of a 28-day cycle, geometric mean values of the maximum plasma concentration (Cmax) and the area under the plasma concentration versus time curve (AUC0-inf) were 377 ng/mL and 1549 ng*hr/mL, respectively.
Distribution
Romidepsin is highly protein bound in plasma (92% to 94%) over the concentration range of 50 ng/mL to 1000 ng/mL with α1-acid-glycoprotein (AAG) being the principal binding protein.
Metabolism
Romidepsin undergoes extensive metabolism in vitro primarily by CYP3A4 with minor contribution from CYP3A5, CYP1A1, CYP2B6, and CYP2C19. At therapeutic concentrations, romidepsin did not competitively inhibit CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP2E1, or CYP3A4 in vitro.
Excretion
Following 4-hour intravenous administration of romidepsin at 14 mg/m2 on days 1, 8 and 15 of a 28-day cycle in patients with T cell lymphomas, the terminal half-life (t1/2) was approximately 3 hours. No accumulation of plasma concentration of romidepsin was observed after repeated dosing.
Effect of Age, Gender or Race
The population pharmacokinetic analysis of romidepsin showed that age, gender, or race (white vs black) did not appear to influence the pharmacokinetics of romidepsin.
Effect of Hepatic Impairment
No dedicated hepatic impairment study has been conducted for ISTODAX. The population pharmacokinetic analysis indicates that mild hepatic impairment [total bilirubin (TB) ≤ upper limit of normal (ULN) and aspartate aminotransferase (AST) > ULN; or TB > 1.0x - 1.5x ULN and any AST] had no significant influence on romidepsin pharmacokinetics. As the effect of moderate (TB > 1.5x - 3x ULN and any AST) and severe (TB > 3x ULN and any AST) hepatic impairment on the pharmacokinetics of romidepsin is unknown, patients with moderate and severe hepatic impairment should be treated with caution [See Use in Specific Populations (8.6)].
Effect of Renal Impairment
No dedicated renal impairment study has been conducted for ISTODAX. The population pharmacokinetic analysis showed that romidepsin pharmacokinetics were not affected by mild (estimated creatinine clearance 50 - 80 mL/min), moderate (estimated creatinine clearance 30 - 50 mL/min), or severe (estimated creatinine clearance < 30 mL/min) renal impairment. Nonetheless, the effect of end-stage renal disease on romidepsin pharmacokinetics has not been studied. Thus, patients with end-stage renal disease should be treated with caution [See Use in Specific Populations (8.7)].
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been performed with romidepsin. Romidepsin was not mutagenic in vitro in the bacterial reverse mutation assay (Ames test) or the mouse lymphoma assay. Romidepsin was not clastogenic in an in vivo rat bone marrow micronucleus assay when tested to the maximum tolerated dose (MTD) of 1 mg/kg in males and 3 mg/kg in females (6 and 18 mg/m2 in males and females, respectively). These doses were up to 1.3-fold the recommended human dose, based on body surface area.
Based on non-clinical findings, male and female fertility may be compromised by treatment with ISTODAX. In a 26-week toxicology study, romidepsin administration resulted in testicular degeneration in rats at 0.33 mg/kg/dose (2 mg/m2/dose) following the clinical dosing schedule. This dose resulted in AUC0-inf. values that were approximately 2% the exposure level in patients receiving the recommended dose of 14 mg/m2/dose. A similar effect was seen in mice after 4 weeks of drug administration at higher doses. Seminal vesicle and prostate organ weights were decreased in a separate study in rats after 4 weeks of daily drug administration at 0.1 mg/kg/day (0.6 mg/m2/day), approximately 30% the estimated human daily dose based on body surface area. Romidepsin showed high affinity for binding to estrogen receptors in pharmacology studies. In a 26-week toxicology study in rats, atrophy was seen in the ovary, uterus, vagina and mammary gland of females administered doses as low as 0.1 mg/kg/dose (0.6 mg/m2/dose) following the clinical dosing schedule. This dose resulted in AUC0-inf. values that were 0.3% of those in patients receiving the recommended dose of 14 mg/m2/dose. Maturation arrest of ovarian follicles and decreased weight of ovaries were observed in a separate study in rats after four weeks of daily drug administration at 0.1 mg/kg/day (0.6 mg/m2/day). This dose is approximately 30% the estimated human daily dose based on body surface area.
14 CLINICAL STUDIES
ISTODAX was evaluated in 2 multicenter, single-arm clinical studies in patients with CTCL. Overall, 167 patients with CTCL were treated in the US, Europe, and Australia. Study 1 included 96 patients with confirmed CTCL after failure of at least 1 prior systemic therapy. Study 2 included 71 patients with a primary diagnosis of CTCL who received at least 2 prior skin directed therapies or one or more systemic therapies. Patients were treated with ISTODAX at a starting dose of 14 mg/m2 infused over 4 hours on days 1, 8, and 15 every 28 days.
In both studies, patients could be treated until disease progression at the discretion of the investigator and local regulators. Objective disease response was evaluated according to a composite endpoint that included assessments of skin involvement, lymph node and visceral involvement, and abnormal circulating T-cells ("Sézary cells.").
The primary efficacy endpoint for both studies was overall objective disease response rate (ORR) based on the investigator assessments, and defined as the proportion of patients with confirmed complete response (CR) or partial response (PR). CR was defined as no evidence of disease and PR as ≥50% improvement in disease. Secondary endpoints in both studies included duration of response and time to response.
14.1 Baseline Patient Characteristics
Demographic and disease characteristics of the patients in Study 1 and Study 2 are provided in Table 2.
| Characteristic | Study 1 (N=96) | Study 2 (N=71) |
|---|---|---|
| Age | ||
| N | 96 | 71 |
| Mean (SD) | 57 (12) | 56 (13) |
| Median (Range) | 57 (21, 89) | 57 (28, 84) |
| Sex, n (%) | ||
| Men | 59 (61) | 48 (68) |
| Women | 37 (39) | 23 (32) |
| Race, n (%) | ||
| White | 90 (94) | 55 (77) |
| Black | 5 ( 5) | 15 (21) |
| Other/Not Reported | 1 ( 1) | 1 ( 1) |
| Stage of Disease at Study Entry, n (%) | ||
| IA | 0 ( 0) | 1 ( 1) |
| IB | 15 (16) | 6 ( 9) |
| IIA | 13 (14) | 2 ( 3) |
| IIB | 21 (22) | 14 (20) |
| III | 23 (24) | 9 (13) |
| IVA | 24 (25) | 27 (38) |
| IVB | 0 ( 0) | 12 (17) |
| Number of Prior Skin-Directed Therapies | ||
| Median (Range) | 2 (0,6) | 1 (0,3) |
| Number of Prior Systemic Therapies | ||
| Median (Range) | 2 (1, 8) | 2 (0, 7) |
14.2 Clinical Results
Efficacy outcomes are provided in Table 3. Median time to first response was 2 months (range 1 to 6) in both studies. Median time to CR was 6 months in Study 1 and 4 months in Study 2 (range 2 to 9).
| Response Rate | Study 1 (N=96) | Study 2 (N=71) |
|---|---|---|
|
||
| ORR (CR + PR), n (%) [95% Confidence Interval] | 33 (34) [25, 45] | 25 (35) [25, 49] |
| CR, n (%) [95% Confidence Interval] | 6 (6) [2, 13] | 4 (6) [2, 14] |
| PR, n (%) [95% Confidence Interval] | 27 (28) [19, 38] | 21 (30) [20, 43] |
| Duration of Response (months) | ||
| N | 33 | 25 |
| Median (range) | 15 (1, 20*) | 11 (1, 66*) |
15 REFERENCES
- NIOSH Alert: Preventing occupational exposures to antineoplastic and other hazardous drugs in healthcare settings. 2004. U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 2004-165.
- OSHA Technical Manual, TED 1-0.15A, Section VI: Chapter 2. Controlling Occupational Exposure to Hazardous Drugs. OSHA, 1999. http://www.osha.gov/dts/osta/otm/otm_vi/otm_vi_2.html
- American Society of Health-System Pharmacists. ASHP Guidelines on Handling Hazardous Drugs: Am J Health-Syst Pharm. 2006;63:1172-1193.
- Polovich M., White JM, Kelleher LO (eds). Chemotherapy and biotherapy guidelines and recommendations for practice (2nd ed.) 2005. Pittsburgh, PA: Oncology Nursing Society.
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
ISTODAX is supplied as a kit including a sterile, lyophilized powder in a single-use vial containing 10 mg of romidepsin and 20 mg of the bulking agent, povidone, USP. In addition, each kit includes one sterile Diluent vial containing 2 mL (deliverable volume) of 80% propylene glycol, USP, and 20% dehydrated alcohol, USP.
NDC 46026-983-01: ISTODAX® KIT containing 1 vial of romidepsin, 10 mg and 1 vial of diluent for romidepsin, 2 mL per carton
Storage
ISTODAX (romidepsin) for injection is supplied as a kit containing two vials in a single carton. The carton must be stored at 20° to 25°C, excursions permitted between 15° to 30°C. (See USP Controlled Room Temperature.)
Keep out of reach of children.
Procedures for proper handling and disposal of anticancer drugs should be considered. Several guidelines on this subject have been published [See References (15)].
17 PATIENT COUNSELING INFORMATION
See FDA-approved patient labeling.
17.1 Instructions
- Patients should be instructed to report excessive nausea or vomiting, abnormal heartbeat, chest pain, or shortness of breath to their physician. Patients receiving ISTODAX® should seek immediate medical attention if unusual bleeding occurs.
- ISTODAX binds to estrogen receptors. Advise women of childbearing potential that ISTODAX may reduce the effectiveness of estrogen-containing contraceptives [See Warnings and Precautions (5.5)].
- Patients should be instructed to read the patient insert carefully.
Manufactured for:
Gloucester Pharmaceuticals, Inc.
One Broadway 14th Floor
Cambridge MA 02142 USA
Manufactured by:
Ben Venue Laboratories, Inc.
300 Northfield Road
Bedford, OH 44146 USA
FDA-Approved Patient Labeling
ISTODAX (ISS toe dax) (romidepsin) for injection
Read the patient information that comes with ISTODAX before you receive your first treatment and each time before you are treated. There may be new information. This leaflet does not take the place of talking with your doctor about your medical condition or your treatment.
What is ISTODAX?
ISTODAX is a prescription medicine used to treat people with a type of cancer called cutaneous T-cell lymphoma (CTCL) after at least one other type of medicine by mouth or injection has been tried.
It is not known if ISTODAX is safe and effective in children under 18 years of age.
What should I tell my doctor before I receive ISTODAX?
Before taking ISTODAX, tell your doctor if you:
- have any heart problems, including an irregular or fast heartbeat, or a condition called QT prolongation.
- have kidney problems
- have liver problems
- have problems with the amount of potassium or magnesium in your blood
- have nausea, vomiting, or diarrhea
- have any other medical conditions
- are pregnant or plan to become pregnant. ISTODAX may harm your unborn baby. Talk to your doctor about the best way to prevent pregnancy while taking ISTODAX. Tell your doctor right away if you become pregnant while taking ISTODAX.
- are breastfeeding or plan to breastfeed. It is not known if ISTODAX passes into your breast milk. You and your doctor should decide if you will take ISTODAX or breast-feed. Talk to your doctor about the best way to feed your baby while you are being treated with ISTODAX.
Tell your doctor about all of the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
Some medicines may affect how ISTODAX works, or ISTODAX may affect how your other medicines work. Especially tell your doctor if you take or use:
- warfarin sodium (Coumadin, Jantoven) or any other blood thinner medicine. Ask your doctor if you are not sure if you are taking a blood thinner. Your doctor may want to test your blood more often.
- a medicine to treat abnormal heart beats
- a type of birth control that contains estrogen, such as birth control pills, patches, implants, or IUDs. ISTODAX may reduce the effectiveness of estrogen-containing contraceptives. You may become pregnant.
- St. John's Wort (Hypericum perforatum)
- Dexamethasone (a steroid)
- Medicine for:
- tuberculosis (TB)
- seizures (epilepsy)
- bacterial infections (antibiotics)
- fungal infections (antifungals)
- HIV (AIDS)
- depression
Ask your doctor if you are not sure if your medicine is one that is listed above. Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist when you get a new medicine.
How should I take ISTODAX?
- ISTODAX will be given to you by a healthcare provider as an intravenous (IV) injection into your vein usually over 4 hours.
- ISTODAX is usually given on Day 1, Day 8, and Day 15 of a 28 day cycle of treatment.
- Your doctor will decide how long you will receive treatment with ISTODAX.
- Your doctor will check your blood cell counts and other blood tests regularly during your treatment with ISTODAX to check for side effects of ISTODAX. Your doctor may decide to do other tests to check your health as needed.
- Your doctor may stop your treatment, change when you get your treatment, or change the dose of your treatment if you have certain side effects while taking ISTODAX.
What are the possible side effects of ISTODAX?
ISTODAX may cause serious side effects, including:
-
Low blood cell counts: Your doctor will regularly do blood tests to check your blood counts.
- Low platelets: can cause unusual bleeding, or bruising under the skin. Talk to your doctor right away if this happens.
- Low red blood cells: may make you feel tired and you may get tired easily. You may look pale, and feel short of breath. Tell your doctor if you have these symptoms.
- Low white blood cells: can cause you to get infections, which may be serious.
- Infection can also happen with normal white blood cells counts.
- Tell your doctor right away if you have any of these symptoms: fever, cough, flu-like symptoms, burning on urination, muscle aches, or worsening of skin problems.
- Changes in the electrical activity of your heart seen on ECG (electrocardiogram). Your doctor may check your ECG as needed for this problem. Tell your doctor if your feel an abnormal heart beat, chest pain or shortness of breath.
Common side effects of ISTODAX include:
- nausea, vomiting, diarrhea, and loss of appetite.
- tiredness
Tell your doctor if you have any side effect that bothers you or does not go away.
These are not all the possible side effects of ISTODAX. For more information, ask your doctor or pharmacist.
Ask your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about ISTODAX
Medicines are sometimes prescribed for purposes other than those listed in patient information leaflets.
This patient information leaflet summarizes the most important information about ISTODAX. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about ISTODAX that is written for health professionals. For more information, go to www.ISTODAX.com or call 1-866-223-7145.
What are the ingredients in ISTODAX?
Active ingredient: romidepsin
Inactive ingredients: povidone. The diluent contains 80% propylene glycol and 20% dehydrated alcohol.
Issued November 2009
Gloucester Pharmaceuticals, Inc.
One Broadway, 14th floor; Cambridge, MA 02142
U.S. Patents: 4,977,138; 7,056,884
PRINCIPAL DISPLAY PANEL - Kit Carton
NDC XXXX-XXXX-XX
ISTODAX® Kit
(romidepsin)
for
injection
10 mg
per single-use vial
Rx Only
Reconstitution and dilution required.
After reconstitution with 2 mL of supplied Diluent,
the concentration of ISTODAX® is 5 mg/mL.
Discard unused portion.
Each kit contains:
- –
- One single-use vial with 10 mg of ISTODAX®
- –
- One single-use vial with 2 mL of Diluent
For Intravenous Use Only

| ISTODAX
romidepsin kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Gloucester Pharmaceuticals, Inc. (153288035) |