FOLIKA-D- folic acid, cholecalciferol, folate tablet
Innovida Phamaceutique Corporation
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Innovida FOLIKA-D
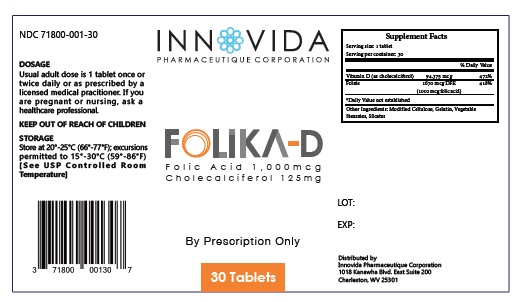
Usual adult dose is 1 tablet once or twice daily or as prescribed by a licensed medical practitioner. If you are pregnant or nursing, ask a healthcare professional.
This product is contraindicated in patients with a known hypersensitivity to any of the ingredients.
Folika-D tablets should only be used under the direction and supervision of a licensed medical practitioner. Use with caution in patients that may have a medical condition, are pregnant, lactating, trying to conceive, under the age of 18, or taking medications.
Pregnancy and Lactation
Folika-D is not intended for use in pregnant or lactating patients.
Folika-D Tablets is an orally administered prescription vitamin formulation for the clinical dietary management of suboptimal nutritional status in patients where advanced folate supplementation is required and nutritional supplementation in physiologically stressful conditions for maintenance of good health is needed.
Folika-D tablets are supplied as round shape biconvex plain tablet, natural color, mostly white with a light yellow in a child-resistant bottle of 30ct. (71800-001-30*)
Dispensed by Prescription†
*Innovida Pharmaceutique Corporation does not represent these product codes to be National Drug Codes (NDC). Product codes are formatted according to standard industry practice, to meet the formatting requirement by pedigree reporting and supply-chain control including pharmacies.
† This product is a prescription-folate with or without other dietary ingredients that – due to increased folate levels increased risk associated with masking of B12 deficiency (pernicious anemia) requires administration under the care of a licensed medical practitioner (61 FR 8760). 1-3 The most appropriate way to ensure pedigree reporting consistent with these regulatory guidelines and safety monitoring is to dispense this product only by prescription (Rx). This is not an Orange Book product. This product may be administered only under a physician’s supervision and all prescriptions using this product shall be pursuant to state statutes as applicable. The ingredients, indication or claims of this product are not to be construed to be drug claims.
- Federal Register Notice of August 2, 1973 (38 FR 20750)
- Federal Register Notice of October 17, 1980 (45 FR 69043, 69044)
- Federal Register Notice of March 5, 1996 (61 FR 8760)
| FOLIKA-D
folic acid, cholecalciferol, folate tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Innovida Phamaceutique Corporation (080892908) |
| Registrant - Innovida Phamaceutique Corporation (080892908) |