DICLOFENAC SODIUM- diclofenac sodium tablet, delayed release
REMEDYREPACK INC.
----------
BOXED WARNING
Cardiovascular Risk
- NSAIDs may cause an increased risk of serious cardiovascular thrombotic events, myocardial infarction, and stroke, which can be fatal. This risk may increase with duration of use. Patients with cardiovascular disease or risk factors for cardiovascular disease may be at greater risk. (See WARNINGS).
- Diclofenac sodium delayed-release tablets are contraindicated for the treatment of perioperative pain in the setting of coronary artery bypass graft (CABG) surgery (see WARNINGS).
Gastrointestinal Risk
- NSAIDs cause an increased risk of serious gastrointestinal adverse events including inflammation, bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events (See WARNINGS).
DESCRIPTION
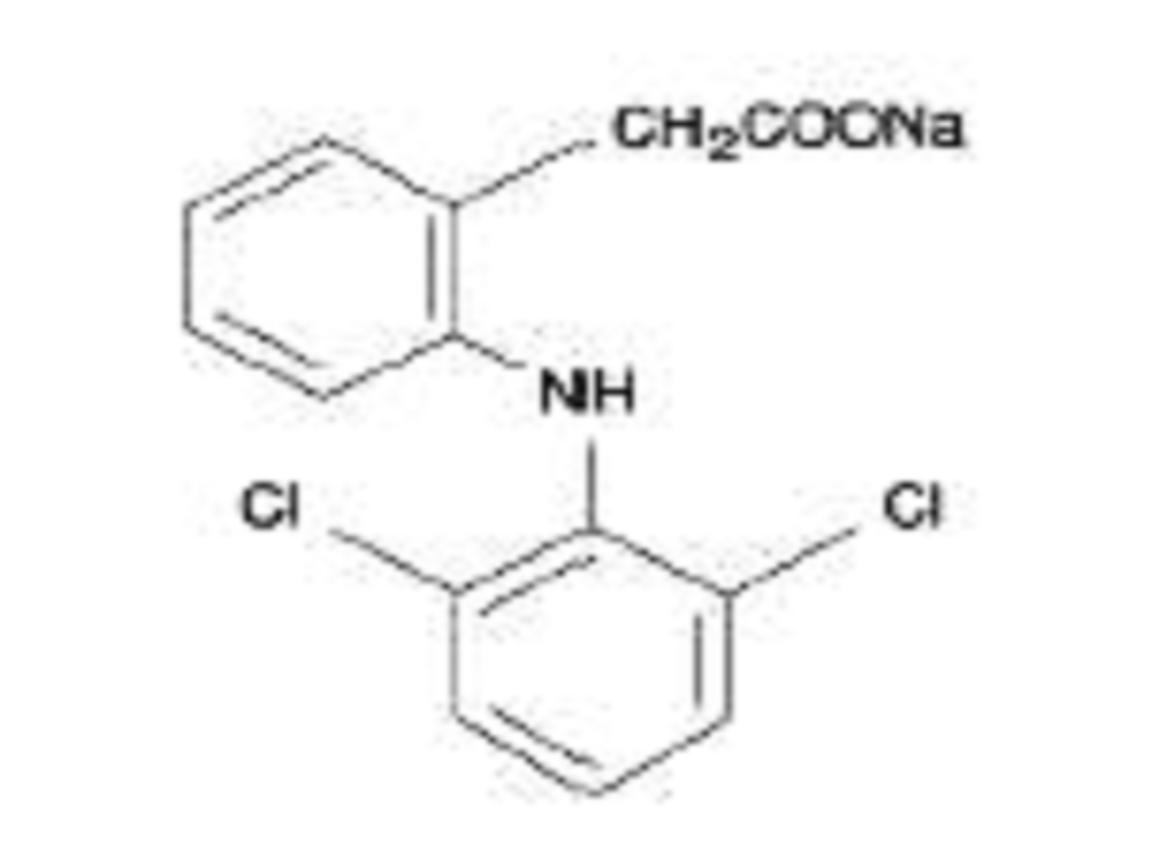
Diclofenac sodium delayed-release tablets are a benzeneacetic acid derivative. The chemical name is 2-[(2,6-dichlorophenyl)amino] benzeneacetic acid, monosodium salt. The molecular weight is 318.13. Its molecular formula is C14H10Cl2NNaO2, and it has the following structural formula
Each enteric-coated tablet for oral administration contains 50 mg or 75 mg of diclofenac sodium. In addition, each tablet contains the following inactive ingredients: aluminum hydrate, colloidal silicon dioxide, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, polyvinyl acetate phthalate, propylene glycol, silica, sodium alginate, sodium starch glycolate (Type A), stearic acid, synthetic black iron oxide, talc, and titanium dioxide.
INDICATIONS & USAGE
Carefully consider the potential benefits and risks of diclofenac sodium delayed-release tablets and other treatment options before deciding to use diclofenac sodium delayed-release. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see
WARNINGS
).
Diclofenac sodium delayed-release tablets are indicated:
For relief of the signs and symptoms of osteoarthritis
For relief of the signs and symptoms of rheumatoid arthritis
For acute or long-term use in the relief of signs and symptoms of ankylosing spondylitis
CONTRAINDICATIONS
Diclofenac sodium delayed-release tablets are contraindicated in patients with known hypersensitivity to diclofenac.
Diclofenac sodium delayed-release should not be given to patients who have experienced asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see
WARNINGS, Anaphylactoid Reactions
, and
PRECAUTIONS, Preexisting Asthma
Diclofenac sodium delayed-release is contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery (see
WARNINGS
INFORMATION FOR PATIENTS
Patients should be informed of the following information before initiating therapy with an NSAID and periodically during the course of ongoing therapy. Patients should also be encouraged to read the NSAID Medication Guide that accompanies each prescription dispensed.
1. Diclofenac sodium delayed-release, like other NSAIDs, may cause serious CV side effects, such as MI or stroke, which may result in hospitalization and even death. Although serious CV events can occur without warning symptoms, patients should be alert for the signs and symptoms of chest pain, shortness of breath, weakness, slurring of speech, and should ask for medical advice when observing any indicative sign or symptoms. Patients should be apprised of the importance of this follow-up (see
WARNINGS, Cardiovascular Effects
).
2. Diclofenac sodium delayed-release, like other NSAIDs, can cause GI discomfort and, rarely, more serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow-up (see
WARNINGS, Gastrointestinal (GI) Effects: Risk of GI Ulceration, Bleeding, and Perforation
).
3. Diclofenac sodium delayed-release, like other NSAIDs, can cause serious skin side effects such as exfoliative dermatitis, SJS, and TEN, which may result in hospitalizations and even death. Although serious skin reactions may occur without warning, patients should be alert for the signs and symptoms of skin rash and blisters, fever, or other signs of hypersensitivity such as itching, and should ask for medical advice when observing any indicative signs or symptoms. Patients should be advised to stop the drug immediately if they develop any type of rash and contact their physicians as soon as possible.
4. Patients should promptly report signs or symptoms of unexplained weight gain or edema to their physicians.
5. Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, andflu-likesymptoms). If these occur, patients should be instructed to stop therapy and seek immediate medical therapy. (see
WARNINGS, Hepatic Effects
).
6. Patients should be informed of the signs of an anaphylactic reaction (e.g., difficulty breathing, swelling of the face or throat). If these occur, patients should be instructed to seek immediate emergency help (see
WARNINGS, Anaphylactic Reactions
).
7. In late pregnancy, as with other NSAIDs, diclofenac sodium delayed-release should be avoided because it will cause premature closure of the ductus arteriosus.
LABORATORY TESTS
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. In patients on long-term treatment with NSAIDs, including diclofenac sodium delayed-release, the CBC and a chemistry profile (including transaminase levels) should be checked periodically. If clinical signs and symptoms consistent with liver or renal disease develop, systemic manifestations occur (e.g., eosinophilia, rash, etc.) or if abnormal liver tests persist or worsen, diclofenac sodium delayed-release should be discontinued.
DRUG INTERACTIONS
Methotrexate: NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. This may indicate that they could enhance the toxicity of methotrexate. Caution should be used when NSAIDs are administered concomitantly with methotrexate.
Cyclosporine: Diclofenac sodium delayed-release, like other NSAIDs, may affect renal prostaglandins and increase the toxicity of certain drugs. Therefore, concomitant therapy with diclofenac sodium delayed-release may increase cyclosporine's nephrotoxicity. Caution should be used when diclofenac is administered concomitantly with cyclosporine.
ACE-Inhibitors: Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE inhibitors. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE inhibitors.
Furosemide: Clinical studies, as well as post-marketing observations, have shown that diclofenac sodium delayed-release can reduce the natriuretic effect of furosemide and thiazides in some patients. This response has been attributed to inhibition of renal prostaglandin synthesis. During concomitant therapy with NSAIDs, the patient should be observed closely for signs of renal failure (see
WARNINGS, Renal Effects
), as well as to assure diuretic efficacy.
Lithium: NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance was decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity.
Warfarin: The effects of warfarin and NSAIDs on GI bleeding are synergistic, such that users of both drugs together have a risk of serious GI bleeding higher than users of either drug alone.
CYP2C9 Inhibitors or Inducers: Diclofenac is metabolized by cytochrome P450 enzymes, predominantly by CYP2C9. Co-administration of diclofenac with CYP2C9 inhibitors (e.g. voriconazole) may enhance the exposure and toxicity of diclofenac whereas co-administration with CYP2C9 inducers (e.g. rifampin) may lead to compromised efficacy of diclofenac. Use caution when dosing diclofenac with CYP2C9 inhibitors or inducers; a dosage adjustment may be warranted (see
CLINICAL PHARMACOLOGY, Pharmacokinetics, Drug Interactions
LABOR & DELIVERY
In rat studies with NSAIDs, as with other drugs known to inhibit prostaglandin synthesis, an increased incidence of dystocia, delayed parturition, and decreased pup survival occurred. The effects of diclofenac sodium delayed-release on labor and delivery in pregnant women are unknown.
NURSING MOTHERS
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from diclofenac sodium delayed-release, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
GERIATRIC USE
As with any NSAIDs, caution should be exercised in treating the elderly (65 years and older).
OVERDOSAGE
Symptoms following acute NSAID overdoses are usually limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which are generally reversible with supportive care. Gastrointestinal bleeding can occur. Hypertension, acute renal failure, respiratory depression and coma may occur, but are rare. Anaphylactoid reactions have been reported with therapeutic ingestion of NSAIDs, and may occur following an overdose.
Patients should be managed by symptomatic and supportive care following a NSAID overdose. There are no specific antidotes. Emesis and/or activated charcoal (60 to 100 g in adults, 1 to 2 g/kg in children) and/or osmotic cathartic may be indicated in patients seen within 4 hours of ingestion with symptoms or following a large overdose (5 to 10 times the usual dose). Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
DOSAGE & ADMINISTRATION
Carefully consider the potential benefits and risks of diclofenac sodium delayed-release tablets and other treatment options before deciding to use diclofenac sodium delayed-release. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see
WARNINGS
).
After observing the response to initial therapy with diclofenac sodium delayed-release, the dose and frequency should be adjusted to suit an individual patient's needs.
For the relief of osteoarthritis, the recommended dosage is 100-150 mg/day in divided doses (50 mg b.i.d. or t.i.d., or 75 mg b.i.d.).
For the relief of rheumatoid arthritis, the recommended dosage is 150-200 mg/day in divided doses (50 mg t.i.d. or q.i.d., or 75 mg b.i.d.).
For the relief of ankylosing spondylitis, the recommended dosage is 100-125 mg/day, administered as 25 mg q.i.d., with an extra 25-mg dose at bedtime if necessary.
Different formulations of diclofenac (diclofenac sodium delayed-release tablets; diclofenac sodium extended-release tablets; diclofenac potassium immediate-release tablets) are not necessarily bioequivalent even if the milligram strength is the same.
HOW SUPPLIED
Diclofenac Sodium Delayed-Release Tablets, USP are available as follows:
50 mgEach white, round, enteric-coated tablet printed with "R" on one side and 550 on the other side with black ink contains 50 mg of Diclofenac Sodium, USP. Tablets are supplied in bottles of 60 (NDC 0228-2550-06), 100 (NDC 0228-2550-11) and 1000 (NDC 0228-2550-96).
75 mgEach white, round, enteric-coated tablet printed with "R" on one side and 551 on the other side with black ink contains 75 mg of Diclofenac Sodium, USP. Tablets are supplied in bottles of 60 (NDC 0228-2551-06), 100 (NDC 0228-2551-11), and 1000 (NDC 0228-2551-96).
| DICLOFENAC SODIUM
diclofenac sodium tablet, delayed release |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - REMEDYREPACK INC. (829572556) |