MENTHOLIX- menthol and lidocaine hydrochloride spray
1st Class Pharmaceuticals
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Mentholix
Warnings
For external use only. Use only as directed.
Avoid contact with eyes and mucous membranes.
Do not apply to open wounds or damaged skin.
Do not use in combination with any external
heat source.
Do not use on irritated or broken skin, or use
in combination with any bandage, wrap, stocking or
similar device or garment.
Consult physician for children under 12 years
of age.
Do not use if allergic to Camphor, Histamine,
Menthol or any other components.
Stop use and consult physician if symptoms
worsen or persist for more than 7 days.
Stop use if itching or excessive skin
irritation occurs.
If pregnant or breast feeding, contact
physician prior to use.
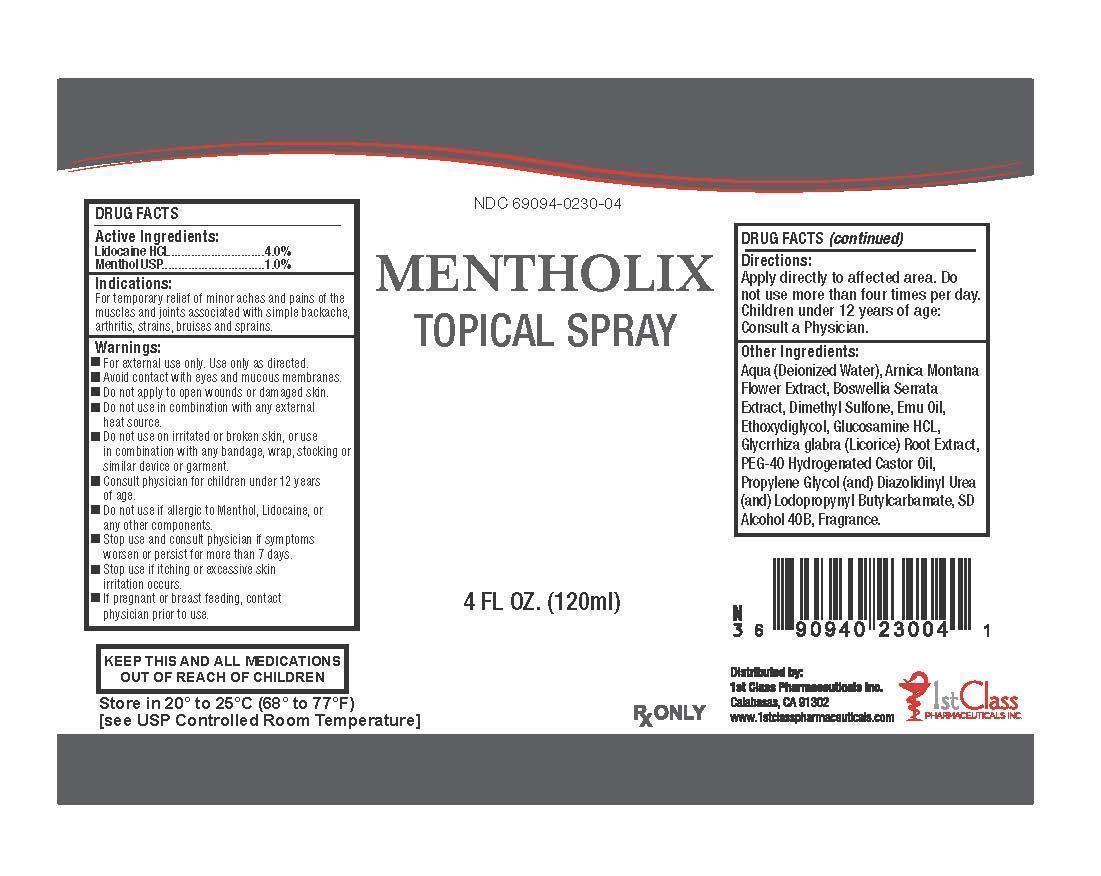
ACTIVE INGREDIENTS:
Lidocaine HCL 4%
Menthol 1%
INDICATIONS AND USAGE: For temporary relief of minor aches and pains of the muscles and joints associated with simple backache, arthritis, strains, bruises and sprains.
CLINICAL PHARMACOLOGY: Menthol instantly brings cool, soothing pain relief to aching muscles. Menthol also has a natural analgesic attribute when applied topically. It has molecules called ligands, which attach themselves to receptors (Kappa Opioid Receptors) in your cell, which produces a numbing effect. Using Menthol topically causes vasodilation, which increases blood flow to the affected area, allowing other medicinal ingredients found in the topical spray to work faster. Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, thereby effecting local anesthetic action.
CONTRAINDICATIONS: Mentholix is contraindicated in patients with known history of hypersensitivity to local anesthetics, Menthol and to other components of Mentholix topical spray. Directions:
Adults and children 12 years and over apply directly to affected area. Do not use more than four times per day. Children under 12 years of age, consult a physician prior to use.
Wash hands with soap before and after use. Avoid contact with eyes and mucous membranes.
WARNING: -For external use only. Use only as directed. -Avoid contact with eyes and mucous membranes. -Do not apply to open wounds or damaged skin. -Do not use in combination with any external heat source. -Do not use on irritated or broken skin, or use in combination with any bandage, wrap, stocking or similar device or garment. -KEEP OUT OF REACH OF CHILDREN. Consult physician for children under 12 years of age. -Do not use if allergic to Menthol, Lidocaine, or any other components. -Stop use and consult physician if symptoms worsen or persist for more than 7 days. -Stop use if itching or excessive skin irritation occurs. -If pregnant or breast feeding, contact physician prior to use.
Other Ingredients: Aqua (Deionized Water), Arnica Montana Flower Extract, Boswellia serrata extract, Dimethyl Sulfone, Emu Oil, Ethoxydiglycol, Glucosamine HCL, Glycrrhiza glabra (Licorice) Root Extract, PEG-40, Hydrogenated Castor Oil, Propylene Glycol (and) Diazolidinyl Urea (and) Iodopropynyl Butylcarbamate, SD Alcohol 40B.
How Supplied:
Mentholix is supplied in a 4 ounce (120ml) plastic bottle with a fine mist sprayer for topical application.
Storage:
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Avoid direct sunlight.
Aqua (Deionized Water), Arnica Montana
Flower Extract, Boswellia Serrata
Extract, Dimethyl Sulfone, Emu Oil,
Ethoxydiglycol, Glucosamine HCL,
Glycrrhiza glabra (Licorice) Root Extract,
PEG-40 Hydrogenated Castor Oil,
Propylene Glycol (and) Diazolidinyl Urea
(and) Lodopropynyl Butylcarbamate, SD
Alcohol 40B, Fragrance.
Apply directly to affected area. Do
not use more than four times per day.
Children under 12 years of age:
Consult a Physician.
| MENTHOLIX
lidocaine, menthol spray |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - 1st Class Pharmaceuticals (079448685) |