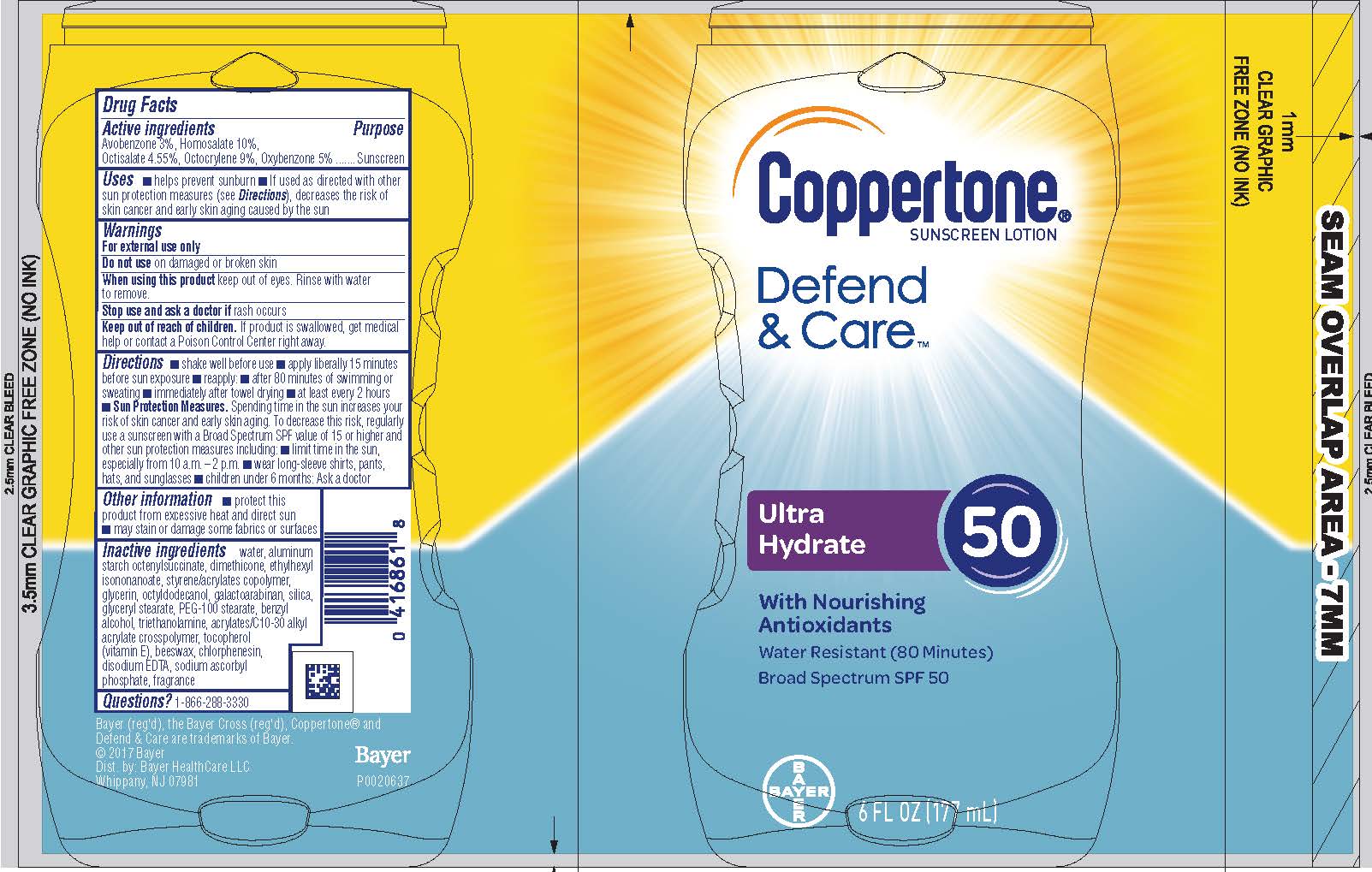
COPPERTONE DEFEND AND CARE ULTRA HYDRATING SPF 50- avobenzone, homosalate, octisalate, octocrylene, and oxybenzone lotion
Bayer HealthCare LLC.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Coppertone Defend & Care Ultra Hydrate Lotion SPF 50
Active ingredients
Active ingredients
Avobenzone 3%, Homosalate 10%, Octisalate 4.55%,
Octocrylene 9 %, Oxybenzone 5%
Uses
Uses
• helps prevent sunburn
• if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Directions
Directions
• Shake well before use.
• apply liberally 15 minutes before sun exposure
• reapply:
• after 80 minutes of swimming or sweating
• immediately after towel drying
• at least every 2 hours
• Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
• limit time in the sun, especially from 10 a.m. – 2 p.m.
• wear long-sleeve shirts, pants, hats, and sunglasses
• children under 6 months: Ask a doctor
Other information
Other information
• protect this product from excessive heat and direct sun
• may stain or damage some fabrics or surfaces
Inactive Ingredients
Water, aluminum starch octenylsuccinate, dimethicone, ethylhexyl isononanoate, styrene/acrylates copolymer, glycerin, octyldodecanol,
Galactoarabinan, silica, glyceryl stearate, peg-100 stearate, benzyl alcohol, triethanolamine, acrylates/C10-30 alkyl acrylate crosspolymer,tocopherol (vitamin E), Beeswax, Chlorphenesin, disodium EDTA, sodium ascorbyl phosphate, fragrance
| COPPERTONE DEFEND AND CARE
ULTRA HYDRATING SPF 50
avobenzone, homosalate, octisalate, octocrylene, and oxybenzone lotion |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - Bayer HealthCare LLC. (112117283) |