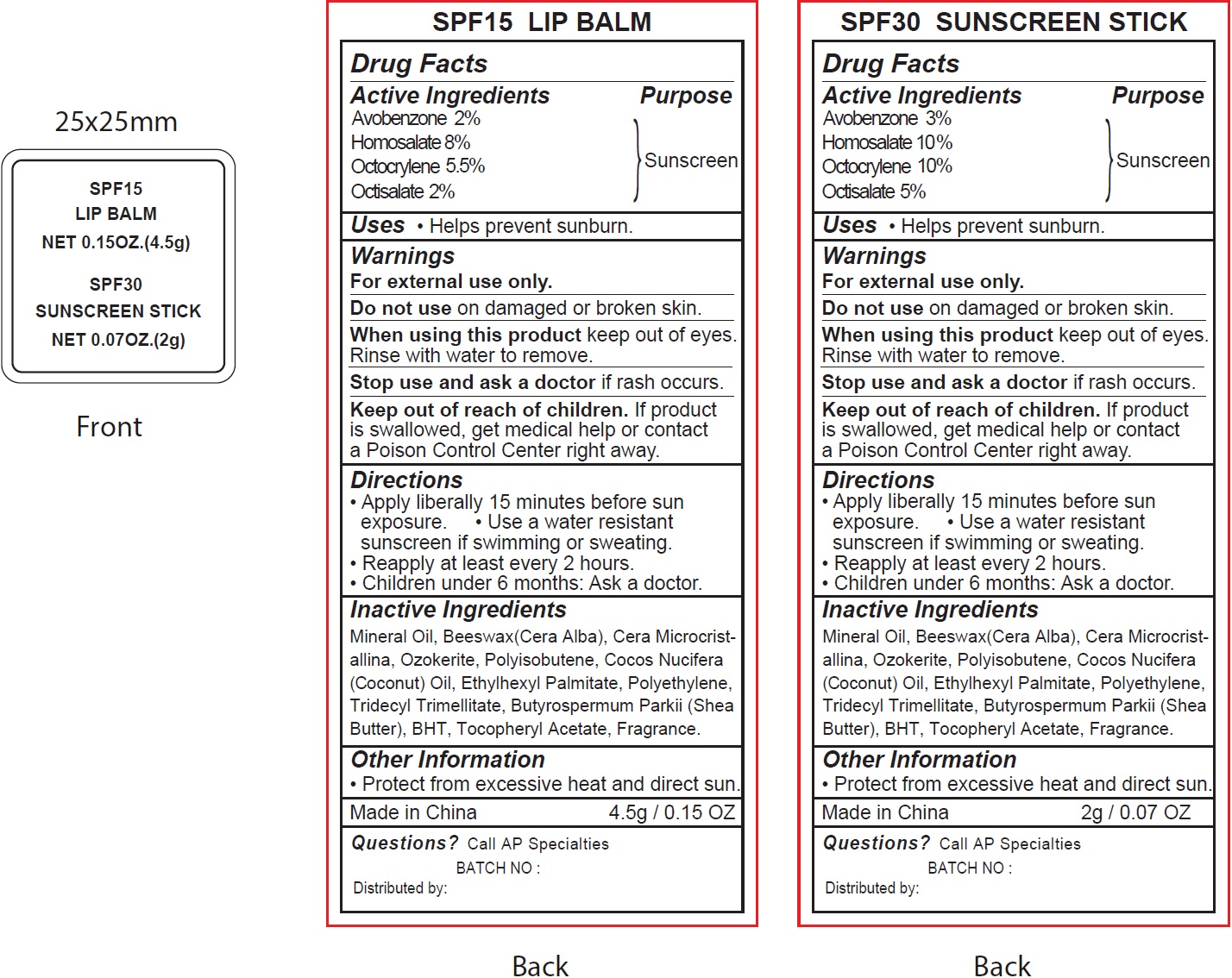
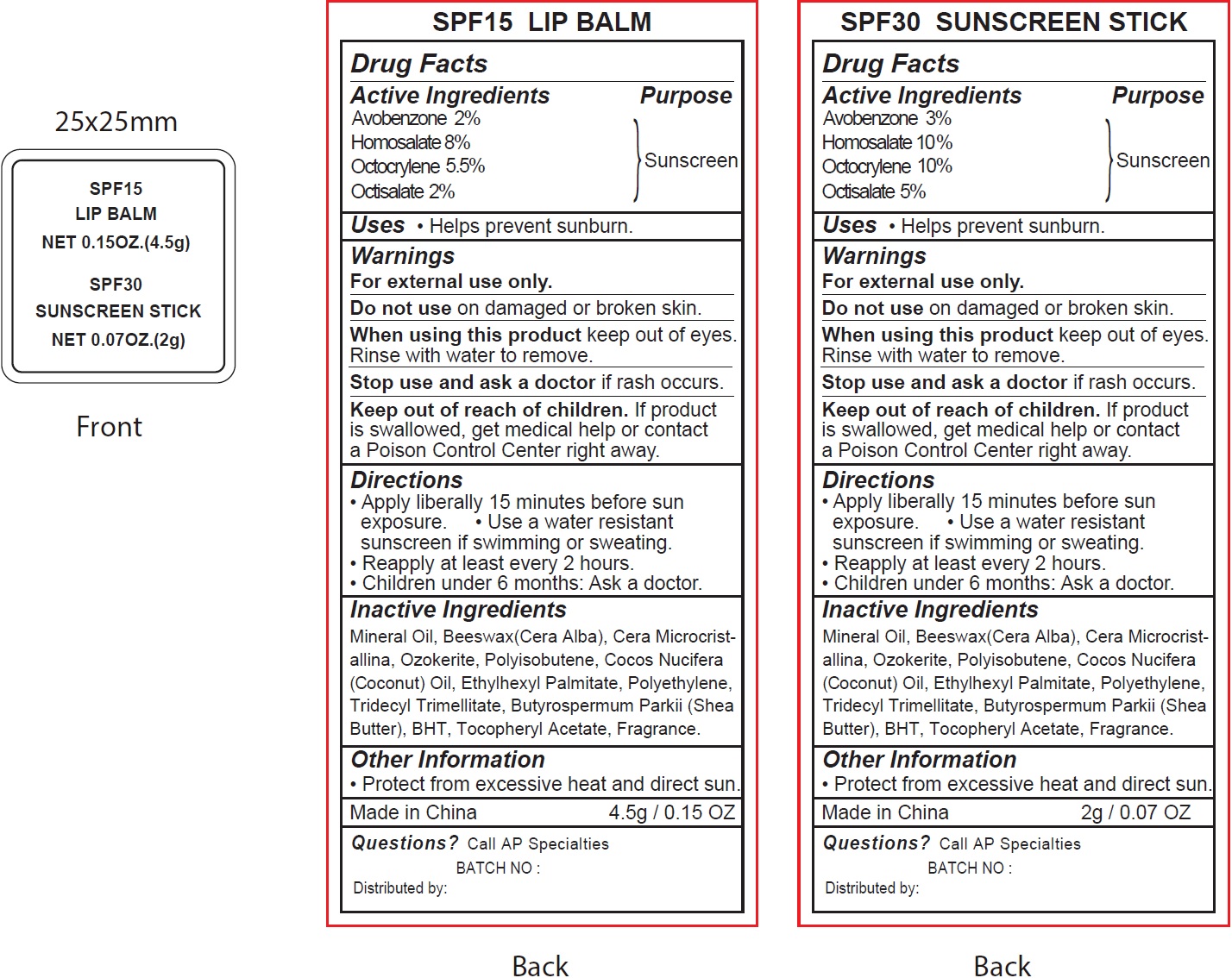
Label: SPF 15 LIP AND SPF 30 SUNSCREEN- avobenzone, homosalate, octocrylene, octisalate kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 70412-211-15, 70412-316-15, 70412-816-10 - Packager: Zhejiang Ayan Biotech Co.,Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 24, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients
- Uses
- Warnings
- Directions
- Inactive Ingredients
- Other Information
- Questions?
- Drug Facts
- Active Ingredients
- Uses
- Warnings
- Directions
- Inactive Ingredients
- Other Information
- Questions?
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
SPF 15 LIP AND SPF 30 SUNSCREEN
avobenzone, homosalate, octocrylene, octisalate kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70412-816 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70412-816-10 1 in 1 KIT 06/01/2018 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 4.5 g Part 2 1 TUBE 2 g Part 1 of 2 SPF15
avobenzone, homosalate, octocrylene, octisalate lipstickProduct Information Item Code (Source) NDC:70412-316 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 20 mg in 1 g HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 80 mg in 1 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 55 mg in 1 g OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 20 mg in 1 g Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) YELLOW WAX (UNII: 2ZA36H0S2V) CERESIN (UNII: Q1LS2UJO3A) COCONUT OIL (UNII: Q9L0O73W7L) ETHYLHEXYL PALMITATE (UNII: 2865993309) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) SHEA BUTTER (UNII: K49155WL9Y) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70412-316-15 4.5 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 06/01/2018 Part 2 of 2 SPF30 SUNSCREEN
avobenzone, homosalate, octocrylene, octisalate stickProduct Information Item Code (Source) NDC:70412-211 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 g HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 100 mg in 1 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 100 mg in 1 g OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 g Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) YELLOW WAX (UNII: 2ZA36H0S2V) CERESIN (UNII: Q1LS2UJO3A) COCONUT OIL (UNII: Q9L0O73W7L) ETHYLHEXYL PALMITATE (UNII: 2865993309) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) SHEA BUTTER (UNII: K49155WL9Y) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70412-211-15 2 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 06/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 06/01/2018 Labeler - Zhejiang Ayan Biotech Co.,Ltd. (544377996)