DECONEX IR- guaifenesin, phenylephrine hydrochloride tablet
Poly Pharmaceuticals, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
DECONEX IR TABLETS
Uses
Temporarily relieves these symptoms due to the common cold, hay fever (allergic rhinitis) or other upper respiratory allergies:
- helps loosen phlegm (mucus) and thin bronchial secretions to drain bronchial tubes and make coughs more productive
- nasal congestion
- reduces swelling of nasal passage
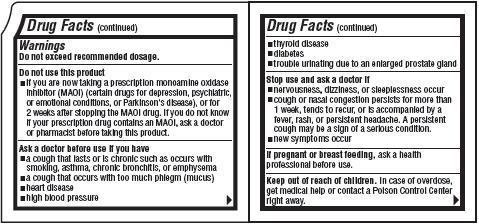
Warnings
Do not exceed recommended dosage.
Do not use this product
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- a cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- a cough that occurs with too much phlegm (mucus)
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to an enlarged prostate gland
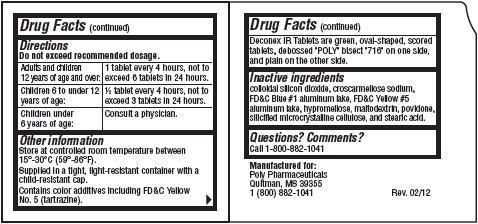
Directions
Do not exceed recommended dosage.
| Adults and children
12 years of age and over: | 1 tablet every 4 hours, not to
exceed 6 tablets in 24 hours. |
| Children 6 to under 12
years of age: | 1/2 tablet every 4 hours, not to
exceed 3 tablets in 24 hours. |
| Children under
6 years of age: | Consult a physician.
|
Other information
Store at controlled room temperature between 15°- 30°C (59°- 86°F).
Supplied in a tight, light-resistant container with a child-resistant cap.
Contains color additives including FD&C Yellow No. 5 (tartrazine).
Deconex IR Tablets are green, oval-shaped, scored tablets, debossed "POLY" bisect "716" on one side, and plain on the other side.
Inactive ingredients
colloidal silicon dioxide, croscarmellose sodium, FD&C Blue #1 aluminum lake, FD&C Yellow #5 aluminum lake, hypromellose, maltodextrin, povidone, silicified microcrystalline cellulose, and stearic acid.
Questions? Comments?
Call 1-800-882-1041
Manufactured for:
Poly Pharmaceuticals
Quitman, MS 39355
1 (800) 882-1041
Rev. 02/12
Product Packaging
The packaging below represents the labeling currently used.
Principal display panel and side panel for 60 tablets label:
NDC 50991-716-60
DECONEX IR
TABLETS
EXPECTORANT · NASAL DECONGESTANT
Each tablet contains:
Guaifenesin..............................380 mg
Phenylephrine HCl......................10 mg
Tamper evident by foil seal under cap.
Do not use if foil seal is broken or missing.
Distributed by:
Poly Pharmaceuticals
Quitman, MS 39355
60 tablets
Rev. 02/12

| DECONEX
IR
guaifenesin, phenylephrine hydrochloride tablet |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Poly Pharmaceuticals, Inc. (198449894) |